- –OH group at position 6:
Can be esterified with acetic acid, palmitic acid (most stable form)
Oxidation reaction
Reaction with FeCl 3 / HCl: forms p-benzoquinone or α-tocopherylquinone ( dark yellow)
Reacts with HNO 3 to form ortho benzoquinone or red tocopherol, which reacts with o-phenylenediamine to form an azo compound
Maybe you are interested!
-
 Pharmaceutical Chemistry, Danang Oriental College - 33
Pharmaceutical Chemistry, Danang Oriental College - 33 -
 Petition to Danang City People's Committee
Petition to Danang City People's Committee -
 Danang International Parachute Competition From 2012-2013
Danang International Parachute Competition From 2012-2013 -
 Oriental Medicine - West Saigon College Used to train Traditional Medicine Doctors - 2
Oriental Medicine - West Saigon College Used to train Traditional Medicine Doctors - 2 -
 Opportunities and Challenges for the Development of Danang Beach Tourism
Opportunities and Challenges for the Development of Danang Beach Tourism
Reducing properties: Vitamin E is a strong reducing agent, protecting other oxidized substances well.
Testing :

Qualitative
IR spectrum, UV absorption spectrum, thin layer chromatography
Test purity
UV absorption, pH, heavy metals, sulfate ash.
Quantitative
Gas chromatography
Effect
Prevents oxidation of cell components, acts as an antioxidant, plays an important role in protecting the integrity of cell membranes, especially red blood cells. If there is a lack of vitamin E, the red blood cell membrane is easily ruptured.
Anti-aging (reduces atherosclerosis, poor vision), heals wounds and burns quickly. Symptoms of vitamin E deficiency
Ataxia, muscle weakness, nystagmus, miscarriage. Anemia or hemolysis in premature infants.
Daily requirement: 12-15mg for children and adults; 3-7mg for infants.
Assign
Treatment and prevention of vitamin E deficiency due to poor absorption through the digestive system.
Side effects
262
When injected, there may be anaphylactic reactions due to castor oil excipients.
High doses may cause gastrointestinal upset and vitamin K resistance (increased blood clotting time).
Dosage
100 – 500mg/day
Form of use
Oral: tablets, capsules 100mg; 200mg; 500mg Injection: 100mg/tube
VITAMIN K
Natural Vitamin K
Plant origin: vitamin K1 is phytomenadione or α – phylloquinone
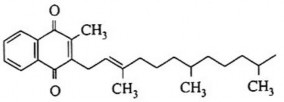
Animal source: vitamin K2 is fernoquinone

Synthetic vitamin K
Vitamin K3 ( menadione)

Vitamin K4
263

Source
Plants : broccoli, watercress, green vegetables... contain vitamin K 1
Animals : fish meal, liver, milk, meat contain vitamin K2
Structure-function relationship
If there are 3 rings, it becomes an inactive anthraquinone.
Nucleus A cannot be substituted with any group (-OH, -CH 3 , -Cl, Br)
Nucleus B: oxygen must be polar
Replace heteroatom O with S or N or 5-membered ring: no activity
Position 2 must have a CH 3 group to be active. If replaced by -H, -Cl, or a chain with many carbons, it will have anti-vitamin K activity.
Branch wire at position 3
No branch wire still works
Long branching lines make fat-soluble vitamins
The branch chain must be long enough, at least 8 carbons, geranyl (C 10 H 17 ) and farnesyl (C 15 H 25 ) have the same activity as natural vitamin K, but when it reaches C 30 H 44 it is less active than vitamin K 1
Saturated and unmethylated side chains make the phylyl chain less active.
When vitamin K 3 carries the -SO 3 Na group, it is Vikasol, it is soluble in water (also has a hemostatic effect), so it can be mixed into a solution for subcutaneous injection.
Preparation of vitamin K3 and vitamin K1
- Synthesis of menadione (vitamin K3 ) and menadiol (from β-methyl naphthalene to create menadione, from menadione to create menadiol)
- From menadione, bisulfite to create vitamin K3 , bisulfite form.
- From menadiol, attach branched chains to create vitamin K1 ( phytomenadione).
264
Nature
- Vitamin K 1 : liquid, viscous, bright yellow oil, d=0.976, crystallizes at -20 degrees C
- Vitamin K 2 : bright yellow crystals, melting point 53 - 54 degrees Celsius
- Vitamin K 3 : bright yellow crystalline powder, strong odor, hot taste, melting point 105 – 107 0 C
- Reactions due to quinone structure : redox reactions
- Reduction reaction : Hydrogen can reduce colored vitamin K3 (naphtoquinone) to colorless vitamin K4 ( naphtohydroquinone ), in humid air this substance is converted back to colored quinone.
- Oxidation reaction : vitamin K3 reacts with oxidation under the influence of light and air and condenses into a dimer, losing its hemostatic effect. The reaction occurs due to the double bond between the two ketone groups, so it is very flexible. Therefore, vitamin K must be preserved away from light and air.
Vitamin K Test 1
Qualitative
UV spectrum, thin layer chromatography, color reaction
Test purity
Solution appearance, pH, menadione and related impurities, sulfate ash.
Quantitative
Liquid chromatography method
Effect
Acts as a coenzyme in the biosynthesis of the elements of the prothrombin complex (factors II, VII, IX and X), ensuring prothrombin concentration in the blood.
Pharmacokinetics
Completely absorbed from the gastrointestinal tract in the presence of bile salts.
Metabolism is unknown, excreted in inactive form in bile and urine.
Duration of action is approximately 6-8 hours after intravenous or intramuscular injection and 12 hours by oral route.
265
Assign
Treatment and prevention of bleeding due to vitamin K deficiency in newborns, premature infants whose mothers use vitamin K antagonists, anticoagulants: phenytoin, cephalosporins containing the tetazonnyl group in the structure such as lactamocef, cefamandol, cefoperazone.
Provide vitamin in case of poor absorption through the digestive tract due to biliary obstruction, hepatitis, deficiency due to use of vitamin K antagonists, antiepileptic drugs. A single dose is sufficient but can also be repeated if using many vitamin K antagonists, poisoning by rat poison with vitamin K antagonists
Side effects
Anaphylactic reaction: Intramuscular injection has the risk of local hematoma formation in people who have used vitamin K antagonists, so intravenous injection is better. Hardening of the skin at the injection site is very rare.
Use with caution
Use intravenously only when intramuscular injection is contraindicated (people who have used antihistamines, hypoprothrombinemia)
People who have used vitamin K antagonists should wait about 8 days before using vitamin K.
Form of use
Tablets, oral solution, intramuscular and intravenous injection
Dosage
Treatment and prevention of bleeding
Take 1-2mg
Intramuscular injection: 5mg or 1-2mg if treatment is prolonged
Provide vitamins
Oral: children 5-10mg, adults 10-20mg
Injection: 5-20mg by intramuscular injection, can be injected slowly intravenously if bleeding occurs.
VITAMIN B 1
266
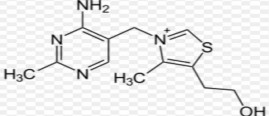
Scientific name : 3-[4-amino-2-methylpyrimidin-5-yl]-5-2(2-hydroxyethyl)-4 methyl-thiazolium hydrochloride.
Other name : thiamine
Source
Plants : yeast, barley germ, rice bran, soybeans, peanuts…
Animals : liver, kidney, pork, egg yolk, milk...
Structure
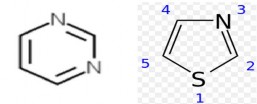
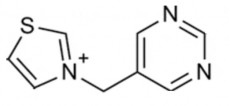
Vitamin B1 molecule has a pyrimidine nucleus and a thiazole nucleus linked by a methylene bridge at C5 of the pyrimidine and C3 of the thiazole.
Structure-function relationship
Pyrimidine nucleus:
- Position 2: the –CH 3 or C 2 H 5 group is active, the butyl group gives antagonistic activity .
- Position 4: free –NH 2 group is necessary, if the substituted form (methylamino – NHCH 3 ) then the activity is almost eliminated, if NH 2 is replaced by –OH group then the anti-vitamin activity is lost.
Thiazole nucleus
- Position 2: carbon must be in a free state, if attached –CH 3, -C 2 H 5 : the molecule will lose its activity .
- Position 4: must have a short carbon chain, when there are more than 3C, the molecule loses its activity but does not turn into an anti-vitamin.
267
- Position 5: Must be in the form of β-hydroxyethyl, primary alcohol for easy esterification or esterification to form pamitate stearate... but most importantly, phosphate or also known as cocarboxylase is a coenzyme of many enzymes that catalyze metabolic processes in the body. If it is in the form of α and secondary alcohol, it forms an inactive compound.
- Replacing the thiazole nucleus with a pyridine nucleus results in pyrithiamine or pyrianeurin, which is an anti-vitamin B1 substance .
- Hydrogenation of the thiazol nucleus to thiazolin makes the molecule inactive.
- Esters are easily hydrolyzed and have the same effect as thiamine but are more stable and longer lasting.
- Methylene bridge: if the two thiazole and pyrimidine groups are directly linked together without going through this group or the bridge consists of two methylene groups, the molecule will not be effective.
Nature
Colorless crystals or white crystalline powder, slightly bitter. Easily soluble in water, soluble in glycerol, slightly soluble in alcohol, practically insoluble in ether.
- Thiamine has the properties of alcohol because it carries β-hydroxyethyl groups in the thiazole nucleus, so it can be esterified with palmitic and stearic acids.
- Reaction with alkaline agents: thiamine is converted to the base form with pseudo-base isomer and then opens the inactive thiazole ring.
- Oxidation in alkaline environment: converted to pseudo-base form, then dehydrated and cyclized to form thiochrome which is no longer biologically active.
Thiochrome fluoresces so this reaction is used for qualitative and quantitative purposes.
Effect of reducing agent: The reaction occurs because the thiazole nitrogen in thiamine is in the form of quaternary ammonium (the thiazole nucleus has a trivalent nitrogen that does not react).
Reducing agents can convert thiamine to dihydrothiamine, which is no longer active as a vitamin. This explains why canned and preserved foods with reducing agents lose their vitamin content very quickly. Therefore, care should be taken when choosing a preservative for vitamin B1 test tubes to be reducing agents similar to sodium hydrosulfite.
Testing
Qualitative
268
IR spectrum, Cl- ion determination reaction , thiochrome formation reaction
Test purity
Clarity – color, pH, nitrates, sulfates, heavy metals, water, sulfated ash.
Quantitative
Anhydrous method.
Physical and chemical incompatibility
Thiamine. HCl is stable in acid solutions (pH 2.5 – 4) but not in alkaline solutions. This explains the incompatibility of thiamine in solution with alkaline substances: Na phenobarbital, bicarbonate, quaternary ammonium.
Effect
Vitamin B1 plays a role in carbohydrate metabolism, nerve conduction, and is a coenzyme in the decarboxylation of α-ketones such as pyruvic acid and α-ketoglutaric acid. Pyruvic acid is toxic to nerve cells and causes polyneuropathy.
Vitamin B 1 decarboxylate acids to form acetaldehyde and CO 2 .
Symptoms of vitamin B1 deficiency
Beriberi occurs in alcoholics who eat grains that are so highly milled that most of the vitamins in the bran are lost.
Daily requirement: 0.3 – 1mg in children; 1.3 – 1.5mg in adults; 1.5 – 1.8mg in pregnant and lactating women
Assign
Treatment of vitamin B1 deficiency Polyneuropathy
Symptoms of vitamin B1 deficiency due to malnutrition, poor diet, and poor absorption.
Contraindications
Intolerance to vitamin B1 often causes shock when injected intravenously, so intramuscular injection is required if necessary, dose 100 - 500mg/day.
Dosage
Prevention: 250 – 500mg/day.
269





