Water-soluble vitamins are less toxic to the body when in excess. However, fat-soluble vitamins such as vitamin A when in excess cause increased pressure in the skull, vitamin C can cause kidney stones, B6 causes sensory nerve disorders, B12 causes excess Co, causing hyperplasia of the thyroid gland, and excessive increase in red blood cells.
1.8. Drug interactions
The effect of vitamins on the effects of drugs
Vitamin D increases blood calcium and can cause heart rhythm disturbances if used with digitalis.
Vitamin D and vitamin K are metabolized by phenytoin and phenobarbital when used long-term for the treatment of epilepsy.
Vitamin K reduces the effect of anticoagulants by antagonizing their effects. Vitamin PP has a peripheral vasodilating effect and can cause orthostatic hypotension when used with antihypertensive drugs of the adrenergic inhibitor group.
Vitamin B6 is a coenzyme that transports amino groups and decarboxylates amino acids, so when combined with levodopa, a Parkinson's drug, it will quickly inactivate this drug.
Vitamin C is an acid, so it quickly reduces the activity of antibiotics that are unstable in an acidic environment such as PNC, ampicillin, erythromycin, increases the absorption of acidic drugs and increases the excretion of alkaline drugs (alkaloids) by making the urine acidic.
Effects of drugs on vitamins
Broad-spectrum antibiotics unbalance the intestinal flora, reducing the amount of vitamins formed in the intestine.
Metal ions of divalent and trivalent ions form complexes with vitamin A.
Laxatives that irritate the digestive mucosa such as phenolphthalein when used for a long time cause changes in the structure of the intestinal mucosa, reducing absorption capacity.
Paraffin laxatives are a barrier that prevents water-soluble vitamins from coming into contact with the intestinal mucosa, hindering the emulsification and dissolution of fat-soluble vitamins.
Colchicine cures gout, fights inflammation that damages the small intestinal mucosa, inhibits enzymes in the intestinal wall, reducing the absorption of vitamin B12 .
Sulfasalazine for colitis inhibits intestinal folate absorption.
Cholinergic agents increase intestinal motility, causing vitamins to move too quickly away from the absorption site, reducing absorption.
Interactions between vitamins
Too much vitamin A causes osteoporosis in sheep despite adequate vitamin D intake. Vitamin C is required for the conversion of vitamin B9 to its active form, and a lack of C causes B9 deficiency .
Vitamin B 6 is needed for the synthesis of vitamin B 3 from tryptophan, a lack of B 6 will cause a lack of B 3 .
2. Common vitamins
VITAMIN A
Vitamin A is the generic name for a number of substances with similar chemical structures and activities. Vitamin A contains a suitable form of retinol (C 20 H 30 O, vitamin A alcohol) and has vitamin A activity corresponding to not less than 95% of the substance stated on the label.
254
It may consist of retinol or retinol esters formed from edible fatty acids mainly acetic or palmitic acid.
Retinal is an aldehyde corresponding to retinol, 11-cis-retinal combines with opsine (protein) to form rhodopsin, a light-sensitive substance of rod cells in the retina, helping to see clearly in low light intensity.
Retinoic acid : has effective activity in skin treatment indications, these substances are more easily tolerated than vitamin A, used to treat keratinization disorders.
Tretinoin or etinonic acid: treats acne.
Isotretinoin : is the 13-cis isomer of fully trans retinoic acid used to treat severe acne such as nodular, cystic, and resistant acne to other treatments.
Recipe
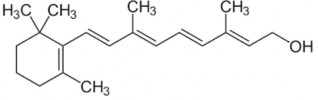
Aromatic retinoids: the hexenyl ring of fully trans retinoic acid is replaced by a benzene ring. Aromatic retinoids are used to treat psoriasis and have the potential to cause birth defects, so contraception is required when using the drug.
RETINOL
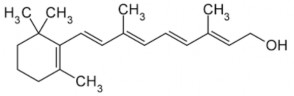
C 20 H 30 O ptl: 286.5
Scientific name: 9,13-dimethy 1-1-(2,16,17-trimethylcyclohex-1-enyl)nona-2',4',6',8'- tetraen-1-ol.
Source
In nature, it is often in the form of fatty acid esters in fish liver, eggs, butter, milk... Provitamin A (pre-vitamin A) or carotenoids are often found in the green parts of plants, orange peels of fruits: carrots, tomatoes, gac...
Carotene exists in α, β, γ forms, β-carotene decomposes into two vitamin A molecules.
Structure
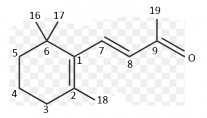
Vitamin A has a diterpene structure consisting of 4 isopen units (-C5H8) joined together, in the molecule there are 4 conjugated double bonds in the branch chain and one in the β-ionone nucleus.
Structure of β-ionone nucleus.
255
Vitamin A1 or retinol is found in the liver of saltwater fish.
Vitamin A2 or 3-dehydroretinol is found in the liver of fish living in brackish water, and is about 1/3 as active as vitamin A1.
Structure-function relationship
β-ionone nucleus : is a necessary component for its action.
Methyl group in β-ionone nucleus : if H is replaced for methyl group, the activity is significantly reduced.
Branch circuit :
Number of C atoms: must be at least 9, attached to position 1 of the β-ionone nucleus. At positions 9 and 13, there is a –CH3 group.
The conjugated double bonds of the branch chain are also conjugated with the double bonds of the nucleus. Displacement of the double bonds (still conjugated) results in inactivation. Removal of double bonds in the branch chain: no more activity.
Conversion of double bonds to triple bonds also inactivates them.
Primary alcohol group: when oxidized to -COOH, the molecule still retains activity.
Nature
Slightly yellow crystals, practically insoluble in water, easily soluble in alcohol, chloroform, ether, petroleum ether, fatty oils.
- Primary alcohol group : can be etherified, esterified, in the form of ester the preparation is more stable. When oxidized to aldehyde, retinal participates in visual function or to retinoic acid participates in the process of cell differentiation.
Conjugated double bond system : makes the molecule very susceptible to oxidation and unstable, especially when exposed to light and air.
Conjugated system attached to primary alcohol group : easy to react with metal hallogennides, electrophiles to form colored substances.
Carr and Price reaction (1926): retinol in chloroform reacts with stibi chloride (antimoin chloride) in chloroform without traces of ethanol to form a blue product. This reaction is used to qualitatively determine vitamin A by spectrophotometry at 587 nm, however, the quantification will not be specific because vitamin D and the degradation products of vitamin A also give this reaction.
Testing
Qualitative
UV spectrum, color reaction.
Test purity
pH:>2, peroxide (volume method).
Quantitative
UV spectrometry method.
256
Assign
Prevention and treatment of vitamin A deficiency due to malnutrition or malabsorption.
Side effects
Acute : doses above 150,000 IU in children and 1,000,000 IU in adults may cause digestive disorders, insomnia, increased intracranial pressure, convulsions, dry skin, itching, hair loss. The drug should be discontinued immediately and corticosteroids used if necessary.
Chronic : infants: dose>10,000 IU/day, 1-3 months; children: 10,000-30,000 IU/day, 2-6 months, adults: 50,000-100,000 IU/day, 6-12 months causes growth arrest, cirrhosis.
Use with caution
Do not exceed the prescribed dose and duration of treatment.
Pregnant women: can cause birth defects at doses above 20,000 IU/day and long-term use. Vitamin supply during this period is about 6,000 IU/day, can be through food. Breastfeeding women: risk of poisoning for children when using high doses.
Form of use
Soft capsules: vitamin supplements, dry eye treatment. Hard capsules, cream, gel: acne treatment. Dosage
Oral: to treat dry eyes: 25,000-50,000 IU/day for 5 days, to make vitamin deficiency symptoms disappear: 10,000-25,000 IU/day for 2-3 weeks, can use up to 200,000 IU. Topical application: in case of psoriasis, acne.
VITAMIN D
Define
Vitamin D is a name used to refer to substances with similar structures, including:
Provitamin D : ergosterol; dehydro-7-cholesterol; dihydro-22,23-ergosterol and dehydro-7- stigmasterol.
Vitamin D : vitamin D2 (ergocalciferol); vitamin D3 (cholescalciferol); vitamin D4 (dihydroergocalciferol) and vitamin D5 (sitocalciferol).
Source
Plants : vitamin D2 or ergocalciferol is prepared by irradiating ultraviolet rays on ergosterol of plant origin such as yeast and mushrooms.
Animals : vitamin D5 or cholecalciferol is found in fish liver oil. In humans, this substance is formed by exposing 7-dehydrocholesterol in the skin to ultraviolet rays from sunlight. This substance is then hydroxylated at position 25 in the liver and then hydroxylated again at position 1 in the kidney to form 1,25-dihydroxycalciferol, the active form.
Structure
Provitamin D has a sterol structure consisting of
257
Table 13.2 Structure of vitamin D
Vitamin D precursor
Vitamin D | R | |
Ergosterol | Vitamin D2 (ergocalciferol) |
|
Dehydro-7-cholesterol | Vitamin D3 (cholecalciferol) |
|
Dihydro-22,23-ergosterol | Vitamin D4 (dihydroergocalciferol) |
|
Dehydro-7-stigmaterol | Vitamin D5 (sitocalciferol) |
|
Maybe you are interested!
-
 Pharmaceutical Chemistry, Danang Oriental College - 34
Pharmaceutical Chemistry, Danang Oriental College - 34 -
 Petition to Danang City People's Committee
Petition to Danang City People's Committee -
 Danang International Parachute Competition From 2012-2013
Danang International Parachute Competition From 2012-2013 -
 Oriental Medicine - West Saigon College Used to train Traditional Medicine Doctors - 2
Oriental Medicine - West Saigon College Used to train Traditional Medicine Doctors - 2 -
 Opportunities and Challenges for the Development of Danang Beach Tourism
Opportunities and Challenges for the Development of Danang Beach Tourism

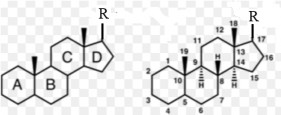
Cyclopentanoperhydrophenatrene nucleus: has 17 carbons divided into 4 rings A, B, C, D.
- The nucleus can be saturated or carry double bonds at 5-6 and 7-8, the –OH group is attached at position 3; 2 methyl groups (-CH3) are attached at positions 10 and 13.
For sterols to become provitamins, the B nucleus needs a conjugated double bond system at 5-6, 7-8.
Vitamin D no longer has a sterol structure because the B ring is opened and has additional double bonds at 10-19, so it becomes a system of 3 conjugated double bonds (triene) at 5-6, 7-8, 10-19.
The branch chain at carbon 17 is different.
Structure-function relationship
Sterol nucleus: the effects of vitamins D 2 and D 3 depend on the special steroid structure of the nucleus:
- Ring B must be open.
- The –OH group at position 3 must be in the β position (above the plane) and free.
- Must have a methylene group at position 10.
5-6, 7-8, 10-19 conjugated triple bond system
Branched chain – R: affects the intensity of the reaction, if the carbon chain is shorter or longer, the loss of double bond will reduce the activity.
Preparation
Vitamin D 3 ; extracted from natural ingredients Vitamin D 3 ; semi-synthetic from provitamin D 2 Properties:
- Colorless needle-shaped crystals, insoluble in water, soluble in alcohol, acetone, ether, chloroform.
- Slow melting temperature: 115 – 118 0 C, instantaneous 120 0 C.
- Stable in alkaline environment, neutral even when heated to 100 0 C. Not stable in acidic environment, light.
258
- The –OH group at position 3: can participate in the esterification reaction.
- Liebermann reaction due to –OH at β position: reacts with concentrated sulfuric acid to give red color, when acetic anhydride in chloroform is added, the red color gradually changes to purple and then blue.
- Conjugated double bond system: reacts with SbCl 3 in chloroform to create a pink product that absorbs UV light, so it can be qualitatively and quantitatively identified.
- Characteristic reaction of sterol nucleus: Pesez reaction is used to distinguish vitamin D from other sterols: dissolve in trichloroacetic acid and add furfurol solution:
Calciferol: gives pink color
Vitamin D 2 : gives purple color
Vitamin D 3 : gives orange color
Other sterols: no reaction
Vitamin D 3 Test
Qualitative
Melting point (82 – 87 0 C), IR spectrum, thin layer chromatography.
Test purity
Ultraviolet rotational power, UV absorbance, related impurities (7-dehydrocholesterol).
Quantitative
Liquid chromatography method.
Effect
Vitamin D maintains blood calcium ion levels by:
Facilitates the absorption of calcium ions in the intestine as well as their fixation on bone tissue, thus preventing rickets in children.
Assign
Prevention of vitamin D deficiency: for the elderly, pregnant women, breastfeeding women, people with poor intestinal absorption of vitamin D, biliary obstruction, liver cell failure, taking anticonvulsants. Prevention of rickets: up to 18 months of age for children who drink no more than 500ml of milk per day. Do not expose to the sun.
Treatment of rickets, osteomalacia, osteoporosis.
Side effects
Overdose : Symptoms include hypercalcemia, calciuria, calcuria, dehydration, anxiety, vomiting. Immediately stop all sources of vitamin D and calcium, drink plenty of water to compensate for dehydration, if necessary, lower blood calcium with diuretics furosemide, corticosteroids, calcitonin, dialysis.
Use with caution
Because it is often combined with calcium to treat rickets in children, it is necessary to monitor blood and urine calcium periodically every week.
Do not use high doses for people who are immobile for a long time.
259
For pregnant women: the usual dose is about 400 - 600 IU, because if used in higher doses it can cause fetal malformations.
Dosage
Prevention of rickets
Daily use: 1200 – 2400 IU for infants and children; 600 – 1800 IU for adults; 600 IU for pregnant and lactating women.
Use every 6 months: 200,000 IU for children and adults.
People tend to take it periodically rather than daily to avoid overdose or not taking the medicine continuously due to forgetting.
Treatment of rickets, osteoporosis, osteomalacia
Dose 4000-8000IU for children for 3 weeks, dose 4000 - 20000IU for adults until cured
Preparations
- Vitamin D 2 (ergocalciferol).
- Vitamin D 3 (cholecalciferol), combined with vitamin A.
- Alfacalcidol (1α – OH – D 3 ): is the form used for people with kidney failure.
- Calcifediol (25 –OH – D 3 ): is the form used for people with liver failure.
- Calcitriol (1,25 – (OH) 2 – D 3 ): is the form used for people with kidney and liver failure.
VITAMIN E
Vitamin E is a group of (4) tocol derivatives, in which the α-tocopherol form (C29H50O2 ) has the strongest activity .
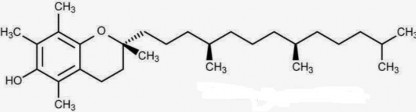
Vitamin E (α-tocopherol) C 29 H 50 O 2 ptl: 430
Scientific name : (2R) – 2,5,7,8 – tetramethyl – 2 – [(4R, 8R)] – 4,8,12 – trimethyl – tridecyl] chroman – 6-ol.
Source
Plants : wheat germ, soybeans, corn, lettuce, sunflower seeds, vegetable oil…
Animals : milk, eggs, beef, pork, fish… Structure
Structure-function relationship
Chroman core :
260
- Replacing oxygen with sulfur in the chroman nucleus forms thiotocopherol, which is a substance that has no vitamin activity but is also not an anti-vitamin.
- The –OH group at position 6 is indispensable. The –OH group can be in free or esterified form, the acetate ester has 3 times higher activity than tocopherol.
- Methyl groups are essential for vitamin activity; the more methyl groups, the higher the activity:
The three groups attached at positions 5, 7, 8 have the strongest activity (α form).
The two groups attached at positions 5, 7 or 5, 8 remain active (β and γ forms)
A group attached at position 5 is no longer active ( δ form)
- Replacing the chroman nucleus with a coumarin or thiochroman nucleus has an insignificant effect.
Branch line :
- Number of carbon atoms in the main chain
From 5 – 9 carbon: inactive
13 carbon: most active
17 carbon: activity reduced 10 times
Add one or two isoperene groups: no longer effective.
- Unbranched chains consisting entirely of –CH 2 - groups and ending in a CH 3 group are inactive.
- If the label chain is replaced by a ring or a –CH 3 group , the molecule will not be active.
Preparation of α-tocopherol
Extraction : From the tips of germinating seeds but difficult due to low yield.
Synthesis : since 1938 by Karrer method (from trimethyl hydroquinone and phytyl bromide)
Properties of α-tocopherol
Lubricant, bright yellow, insoluble in water, dilute acids and dilute alkalis, soluble in ether, absolute alcohol, benzene, chloroform.
Resistant to acid, alkali, heat resistant up to 40 0 C
Destroyed by UV rays, easily oxidized and inactive.
- Chroman nucleus: absorbs UV so it can be qualitatively and quantitatively identified based on this spectrum.
261