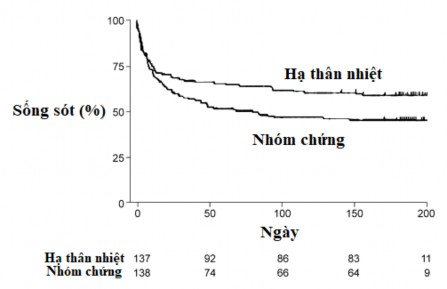
Figure 1.6. Survival curve in HACA study [11].
The results of two randomized controlled clinical studies showed that mild hypothermia (32°C - 34°C) for 12 - 24 hours was effective in improving survival and increasing neurological outcomes in comatose patients after out-of-hospital ventricular tachycardia with initial ventricular fibrillation/ventricular tachycardia [11],[12].
Several studies from different centers around the world have been published since 2002 showing the benefit of hypothermia in patients after ventricular fibrillation, possibly effective in ventricular fibrillation due to asystole or pulseless electrical activity. These studies were all retrospective interventional studies with placebo-controlled groups [76], [77], [78], [79], [80], [81], [82], [83], [84], [85], [86], [87].
(Table 1.4).
29
Table 1.4. Studies on hypothermia in comatose patients after CPR
Study
Number of Patients | Initial heart rate when NTH | Mortality rate (HTN vs. control group) | Good neurological outcome (HTN vs control group) | |
Busch (2006) | 61 | All types | 41% - 68%, p = 0.05 | 41% - 26%, p = 0.21 |
Knafeij (2007) | 72 | ST elevation myocardial infarction with ventricular fibrillation | 25% - 86%, p = 0.001 | 55% - 16%, p = 0.001 |
Bellard (2007) | 68 | Ventricular fibrillation | 44% - 64%, p = 0.04 | 72% - 46%, p = 0.02 |
Sunde (2007) | 119 | All types | 44% - 69%, p = 0.007 | 56% - 26%, p < 0.001 |
Oddo (2008) | 109 | All types | VF: 40% - 56%, p = 0.28 | VF: 56% - 26%, p = 0.004 |
Storm (2008) | 126 | All types | 29% - 42%, p = 0.19 | 62% - 23%, p < 0.001 |
Don (2009) | 491 | All types | VF/VT: 46% - 61%, p = 0.04 | VF/VT: 35% - 15%, p < 0.01 |
Bro-Jeppesen (2009) | 156 | All types | VF/VT: 33% - 32%, p = 0.79 | VF/VT: 97% - 71%, p = 0.003 |
Castrejon (2009) | 69 | Ventricular fibrillation/ventricular tachycardia | 44% - 61%, p = 0.17 | 44% - 18%, p = 0.029 |
Dumas (2011) | 1145 | All types | VF/VT 56% - 71%, p < 0.001 | No analysis |
Testori (2011) | 374 | Asystole/pulseless electrical activity | 65% - 77%, p = 0.024 | 39% - 25%, p = 0.025 |
Lyndbye (2012) | 100 | Asystole/pulseless electrical activity | 61% - 81%, p = 0.03 | 29% - 13%, p = 0.02 |
Maybe you are interested!
-
 Evaluation of the effectiveness of brain protection in comatose patients after circulatory arrest by command hypothermia - 25
Evaluation of the effectiveness of brain protection in comatose patients after circulatory arrest by command hypothermia - 25 -
 Evaluation of the effectiveness of brain protection in comatose patients after circulatory arrest by command hypothermia - 2
Evaluation of the effectiveness of brain protection in comatose patients after circulatory arrest by command hypothermia - 2 -
 Model of Some Related Studies.
Model of Some Related Studies. -
 Foreign Studies on Improving Competitiveness for Tourism Development
Foreign Studies on Improving Competitiveness for Tourism Development -
 Comparison of Distribution by Number of Cesarean Sections Between Studies
Comparison of Distribution by Number of Cesarean Sections Between Studies
Several later studies tested the efficacy of directed hypothermia with other temperature targets. Niklas Nielsen et al. published the results of the TTM trial [13]. (in The New England Journal of Medicine in 2013), comparing the efficacy of directed hypothermia with a target of 33°C versus 36°C. This was a multicenter randomized clinical trial (36 centers in Europe and Australia) [13]. A study of 950 comatose patients after out-of-hospital sepsis, including patients with initial shockable or non-shockable heart rates, was randomized to 2 groups, one group with hypothermia with a target temperature of 33°C, the other group maintained at a target temperature of 36°C. Both surface and intravascular cooling were used, depending on the study site. At 6 months, all-cause mortality was similar between patients randomized to 33°C versus 36°C (50% versus 48%; p = 0.51). The rate of death or survival with poor neurological function at 6 months was also similar (54% versus 52%; p = 0.78) [13]. There was no difference in mortality or neurological outcome between the 2 groups at day 180.
The FROST-I trial (2018) [88] was a randomized controlled trial that studied 150 comatose patients after witnessed out-of-hospital septic shock with initial cardioversion at three different target temperatures of 32°C, 33°C, and 34°C, respectively. The study results found no difference in the rate of survival to discharge with good neurological outcome at 90 days at each target temperature [88]. However, the trial had a relatively small sample size. To date, there have been no further studies large enough to select the optimal target temperature. A target temperature of 36°C may be selected in some cases of coagulopathy, severe acidosis, or hemodynamic disturbances.
The HYPERION trial [89] published in The New England Journal of Medicine (2019) was a large randomized controlled trial evaluating the efficacy of hypothermia in patients with STH with an initial heart rate of
nonshockable rhythm. A study of 581 patients, including both out-of-hospital and in-hospital NTH, with an initial nonshockable rhythm, was randomized to a target temperature of 33°C versus a target temperature of 37°C. The proportion of patients with a good neurological outcome (CPC 1.2) at 90 days was significantly higher in the 33°C target hypothermia group (10.2%) compared with 5.7% in the 37°C group (95% CI 0.1 - 8.9, P = 0.04) but
There was no significant difference in overall mortality between groups (81.3% vs. 83.2%) [89]. This is the first randomized controlled study to demonstrate the benefit of targeted hypothermia of 33°C in patients with nonshockable initial rhythm [89].
Kim et al. [90] published a study on the effectiveness of prehospital hypothermia with cold saline infusion. They randomized 1359 out-of-hospital patients with sepsis to receive 4°C intravenous saline before admission. Patients receiving cold saline achieved their target temperature on average 1 hour earlier than those in the control group, with no significant difference in survival to hospital discharge or good neurological outcome. However, the rate of on-scene cardiac arrest, pulmonary edema, and vasopressor use was higher in the intervention group [90]. This result is consistent with several other randomized studies that also found no improvement in clinical outcomes when starting cold saline intravenously before admission, such as Bernard's study (RICH trial) [91]. Current guidelines from the American Heart Association recommend against starting prehospital hypothermia with cold saline intravenously [14],[15].
PRINCESS trial in witnessed out-of-hospital septic patients undergoing prehospital cooling by nasal spray. This was a multicenter study in 7 European countries, over 8 years from 2010 to 2018. The study was randomized, including 343 prehospital cooling patients and 334 control patients [92]. The median time to reach target temperature below
34°C was shorter with nasal cooling, but survival with favorable neurological outcome did not differ significantly between groups (16.6% vs. 13.5%; P = 0.25) [92]. The PRINCESS trial was negative, but it should be noted that in this study, patients with septic shock had a witness, the rate of bystander CPR was higher than 60%, and the mean time to hospital was 51 minutes. The time to target temperature was 105 minutes in the intervention group and 182 minutes in the control group. The time to hospital and target temperature was also very short in the control group. This may be why the study did not find a difference. To date, the evidence from randomized trials supporting prehospital cooling to achieve target temperature more quickly is lacking.
Kirkegaard et al. [93] published a clinical trial comparing the efficacy of targeted hypothermia of 33°C for 24 and 48 hours. The multicenter randomized controlled trial was conducted in 10 intensive care units of 10 university hospitals in 6 European countries from February 2013 to December 2016 and enrolled 355 patients. Patients were divided into 2 groups, 176 patients in the 33°C target hypothermia group for 48 hours and 179 patients in the 33°C target hypothermia group for 24 hours. The trial found no significant difference in good neurological outcome between the two groups (69% vs. 64%; p = 0.33), nor in mortality between the two groups. The rate of adverse events was higher in the 48-hour group than in the 24-hour group (p = 0.03), and patients had a longer stay in the intensive care unit (151 hours vs. 117 hours; p < 0.001). The 24-hour target temperature maintenance period was still preferred.
The ICECAP trial, which is expected to enroll 1,800 patients over five years (2020-2025) to study whether hypothermia at various time points (6 hours - 12 hours - 18 hours - 24 hours - 36 hours - 48 hours) leads to better outcomes, is currently underway.
In 2015 guidelines, the American Heart Association recommends targeted hypothermia (32°C - 36°C) for comatose patients after VT at level IB for out-of-hospital VT due to ventricular fibrillation, and level IC for other arrhythmias and in-hospital VT [14].
Based on the results of published studies on hypothermia, the 2020 American Heart Association recommendations were enhanced for in-hospital CPR and nonshockable initial rhythm. Target temperature control (32°C - 36°C) is recommended for patients who do not fully respond to commands after out-of-hospital CPR, in-hospital CPR regardless of rhythm, and class IB [15].
Based on the brain protection mechanism when performing hypothermia, as well as clinical research evidence, many researchers in the world still lean towards 33°C if patients have no contraindications [94],[95],[96].
1.3.5. Methods of command hypothermia
Refrigeration devices are all operated on a closed-loop, automatic principle. The system relies on temperature sensors, automatically calculating to adjust the temperature according to the set modes. There are 2 main groups including the external cooling group (surface cooling) and the internal cooling group (intra-circuit cooling) [97]
Extracorporeal cooling systems (surface cooling systems) typically consist of several sheets or patches wrapped around the limbs or upper body that cool directly to the skin. This is a noninvasive method, thus minimizing the risk of bleeding or infection [97].
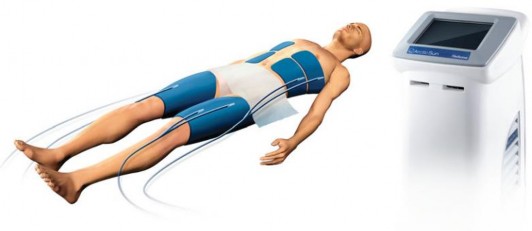
Figure 1.7. Arctic Sun cooling system using adhesive pads [97].
Figure 1.9. Cold blanket system InnerCool STx [97]. |
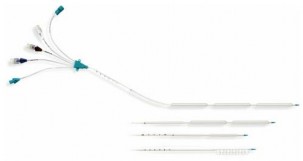
Intravascular cooling systems use a catheter with a closed circuit to circulate cold saline in balloons on the catheter body to directly cool the blood stream. The intravascular method allows for good temperature control and easy patient care. Intravascular catheters, if maintained for a long time, can increase the risk of infection, bleeding, and local injury [97].

| |
Figure 1.10. Cooling catheter InnerCool RTx [97]. | Figure 1.11. Cooling catheter ThermoGuard by ZOLL [97]. |
The ICEREA trial compared surface hypothermia with the ZOLL endovascular hypothermia device in out-of-hospital septic shock patients [98]. The randomized, double-blind trial included 203 patients treated with ZOLL endovascular therapy versus 197 patients treated with surface hypothermia. Results showed no significant difference in favorable neurological outcome at 28 days (OR 1.41; 95% CI 0.93–2.16; P = 0.107), and favorable neurological outcome at 90 days in the endovascular group compared with the surface group with OR 1.51; 95% CI 0.96–2.35 (P = 0.07). However, the group using the intravascular hypothermia device had a significantly shorter time to reach target temperature than the surface group (p < 0.001), and the target temperature was maintained more strictly (p < 0.001) [98]. Nursing care for the intravascular hypothermia device was easier and more convenient [98].
In our study, we used the Thermogard XP endovascular hypothermia device from ZOLL [99].