Parallel Opposing Inductors
Then the total inductance, L T for two parallel opposing inductors is given as:
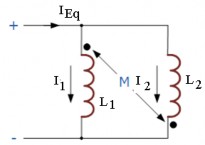
Figure 2.0-31: Parallel inductor with left polarity induction
2.3.6. Inductive Reactance
Since the component we are concerned with is an inductor, the reactance of an inductor is called “Inductive Reactance”. In other words, an electrically inductive resistor when used in an AC signal source circuit is called inductive reactance.
Inductive reactance is given the symbol XL , which is the parameter in an AC circuit that opposes changes in current.
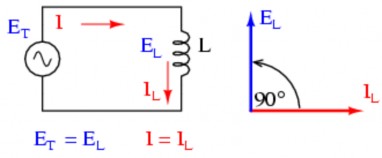
Figure 2.0-32: Current and voltage relationship of coil
In an AC circuit, the current and voltage are out of phase by 90 o , or (π / 2 rads). X L = V L / I L = L ( ) = 2 fL
Example : A 150mH inductance coil is connected across a 100V, 50Hz supply. Calculate the induced resistance of the coil and the current flowing through it.
Tutorial Apply: X L = 2 fL = 2 x 50 x 0.15 = 47.12 I = V / X L = 100 / 47.12 = 2.12 A |
Maybe you are interested!
-
 Basic electronic engineering - City College of Construction. HCM Part 1 - 1
Basic electronic engineering - City College of Construction. HCM Part 1 - 1 -
 Measurement and Remote Control – Electrical Engineering Industry - 8
Measurement and Remote Control – Electrical Engineering Industry - 8 -
 Mechanical Engineering - Automobile Technology - Lao Cai College - 7
Mechanical Engineering - Automobile Technology - Lao Cai College - 7 -
 Basic Refrigeration Refrigeration and Air Conditioning Engineering - Intermediate - Ha Nam Vocational College 2020 - 1
Basic Refrigeration Refrigeration and Air Conditioning Engineering - Intermediate - Ha Nam Vocational College 2020 - 1 -
 Overview of Thai Nguyen University of Industry and Mechanical Engineering
Overview of Thai Nguyen University of Industry and Mechanical Engineering
REVIEW QUESTIONS
Question 1: A resistor has four colors: green, blue, brown, yellow. Find its impedance and tolerance.
Example 2: A resistor has five color bands: red, red, green, yellow, brown. What is its resistance and tolerance?
Question 3: Calculate the total inductance of the following circuit, knowing:
L 1 = 2H, L 2 = 2.5H, L 3 = 5H and L 4 = 1H
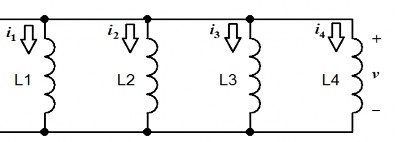
Question 4: Calculate the capacitance C of the following circuit.
The above circuit is equivalent to the following circuit (True / False)
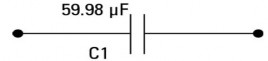
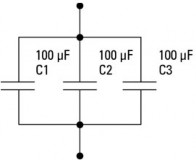
Question 5: Calculate the equivalent capacitance
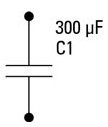
Is the above circuit equivalent to the following?
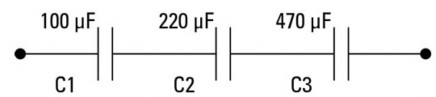
Question 6: Calculate the capacitance of the following circuits.
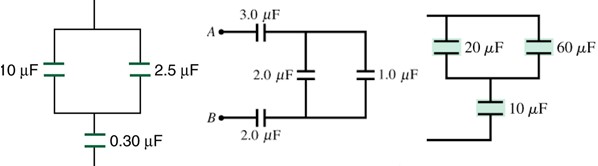
Question 7: Four-band resistor: gray, black, black, no fourth band. What is its resistance and tolerance?
Question 8: Read the resistor values: 103, 392, 563, 105, 390, 470
Hint: 103 = 10 × 1,000 ohms = 10 kiloΩ
392 = 39 × 100 ohms = 3.9 kiloΩ
563 = 56 × 1,000 ohms = 56 kiloΩ
105 = 10 × 100,000 ohms = 1 MegaΩ
390 = 39 × 1Ω = 39Ω or 39RΩ
470 = 47 × 1Ω = 47Ω or 47RΩ
Question 9: What values does the 5-bar resistor: Green Gray Black Red Brown correspond to? Instructions: The colors correspond to: 6 8 0 2 => 680×10 2 =68000Ω or 68kΩ
Question 10: If a resistor is blue-red-yellow-silver then its value is 5.2 Ω and tolerance is +/- 10% true or false? Why?
Question 11: A brown-blue-gray-silver-red resistor would be 1.58 Ω with a tolerance of +/- 2% true or false? Why?
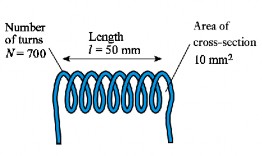
Question 12: In the following circuit, calculate the self-inductance coefficient L
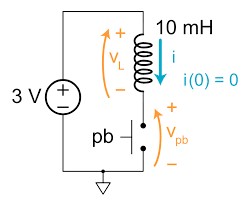
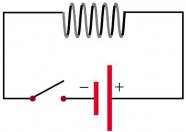
Question 13: Explain the working principle of the following two circuits.
Chapter 3: SEMICONDUCTORS – DIODES
1. Objective
+ Present the concept of semiconductor
+ Describe the structure of a semiconductor diode.
+ Analyze the problem of P - N semiconductor junction.
+ Analyze the operating principle of semiconductor diodes
+ Classification of semiconductor diodes.
2. Main content
2.1. Semiconductor
2.2. Semiconductor diode
2.3. Diode Classification
2.4. Application
3.1. Semiconductor
3.1.1 Pure semiconducting
Semiconductor materials, such as silicon (Si), germanium (Ge), and gallium arsenide (GaAs), have electrical properties that are both “conductors” and “insulators”. They are neither good conductors nor good insulators (hence their name “semi” conductors). They have very few “free electrons” because their atoms are grouped together in a crystalline pattern called a “lattice”, but electrons can still flow only under special conditions.
The ability of a semiconductor to conduct electricity can be greatly improved by replacing or adding certain atoms or accepting atoms into this crystal structure, creating more free electrons or conversely more holes. That is by adding a small percentage of another element to the base material, either silicon or germanium.
Silicon and Germanium are chemically pure semiconductors, containing nothing but the semiconductor material. But by controlling the amount of impurities added to this intrinsic semiconductor material, its conductivity can be controlled. Depending on the impurities added, electrons or holes can be created, respectively.
The process of adding impurities to semiconductor atoms (1 impurity atom per 10 million atoms of semiconductor) is called Doping. When silicon is doped it is no longer pure, these atoms are collectively called “impurities”, and by doping these silicon materials with impurities, we can turn it into a semiconductor.
The most commonly used semiconductor base material today is silicon. Silicon has four valence electrons in its outermost shell which it shares
shares with its neighboring silicon atoms to form eight electrons. The bond structure between two silicon atoms is such that each atom shares one electron with its neighbor making the bond very stable.
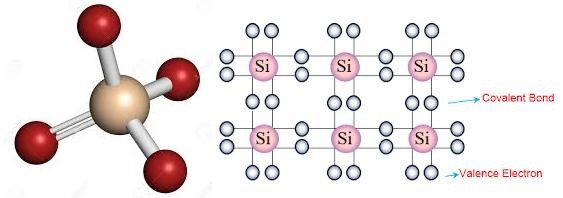
Figure 3.1: Structure and bonding of Silicon atoms
Since there are very few free electrons available to move around the silicon crystal, pure silicon (or germanium) crystals will either be good insulators, or very large value resistors.
The silicon atoms are arranged in a specific symmetrical pattern that makes them a solid crystalline structure. A pure silicon crystal (silicon dioxide or glass) is often said to be a crystal that has no impurities and therefore no free electrons.
But simply connecting a silicon crystal to a power source (like a battery) is not enough to get current flowing from it. To do that, we need to create an anode and a cathode in the silicon that will allow electrons, and thus current, to flow out of the silicon. These are created by doping the silicon with certain impurities.
3.1.2 N-type Semiconductor Basics
In order for our silicon crystal to conduct electricity, we need to introduce an impurity atom such as Arsenic, Antimony or Phosphorus from outside into the structure.