Infusion emulsions should show no evidence of separation. The diameter of the majority (80%) of dispersed droplets should be less than 1µm and no droplet should be greater than 5µm in diameter, unless otherwise specified.
4.2. Volume
The permissible limits for volume differences (%) of infusion drugs are as follows:
- The volume stated on the label is not greater than 50ml, the allowable difference is 10%.
- The volume stated on the label is greater than 50ml, the allowable difference is 5%.
Maybe you are interested!
-
 Some Key Solutions to Promote Positive Changes and Limit Negative Changes of Socialist Production Relations in
Some Key Solutions to Promote Positive Changes and Limit Negative Changes of Socialist Production Relations in -
 Some solutions to limit risks in international goods trading activities of Vietnamese traders - 10
Some solutions to limit risks in international goods trading activities of Vietnamese traders - 10 -
 Basic electronic engineering - City College of Construction. HCM Part 1 - 1
Basic electronic engineering - City College of Construction. HCM Part 1 - 1 -
 Organizing physical education teaching activities at People's Security College I in the current reform period - 14
Organizing physical education teaching activities at People's Security College I in the current reform period - 14 -
 Solutions for developing competitive strategies of Vietnam Airlines - 2
Solutions for developing competitive strategies of Vietnam Airlines - 2
4.3. Pyrogenic substances
Intravenous drugs must be pyrogen-free. Pyrogen testing is not required unless bacterial endotoxin testing is indicated, unless otherwise specified.
5. Some intravenous drug formulas
5.1. Infusion solutions for hydration
5% glucose solution:
Formula: Anhydrous glucose 50g
Distilled water for injection v.v. 1000ml
Glucose used as a preparation material must meet the standards specified in the Pharmacopoeia for preparing injectable drugs, be free of fungi and contain no pyrogens.
Glucose solutions, especially those with high glucose concentrations, may change color when sterilized at high temperatures. This discoloration is due to caramelization of glucose under the influence of heat and alkaline environment. To limit the degree of caramelization of sugar, the Russian Pharmacopoeia lists the formula of glucose solutions as follows:
Anhydrous glucose 50g or 100g or 250g or 400g 0.1N HCl acid solution to pH = 3 – 4
Sodium chloride 0.26g
Distilled water 1000ml
It is believed that sodium chloride forms a complex with glucose and this complex is less likely to caramelize and this salty-sweet solution is suitable for the body's physiology.
Hydrolyzed sucrose solution
Formula: Sucrose 47.5g 0.1N HCl solution 4.0ml Distilled water for injection q.s. 1000ml
Dissolve sucrose in water, add acid, filter, steam in a steamer at 121 o C for 30 minutes. Under the influence of acid and heat, sucrose will be hydrolyzed into glucose and fructose. The solution has a pH of about 3.5 - 6.
5.2. Infusion solutions providing electrolytes
Living cells, the basic units that make up living organisms, are always “bathed” in physiological fluids, which are kept stable and regulated by many complex physiological processes. Any change in the quantity or composition of these fluids can cause physiological disorders.
Body fluids are generally divided into: plasma (fluid within blood vessels), intracellular fluid (fluid within cells) and intercellular fluid (fluid between cells).
The composition of these fluids is water containing a mixture of electrolytes and neutral solutes. The composition of electrolytes in plasma, intracellular fluid and intercellular fluid is recorded in Table 4.5.
Table 4.5. Electrolyte concentrations in normal human body fluids
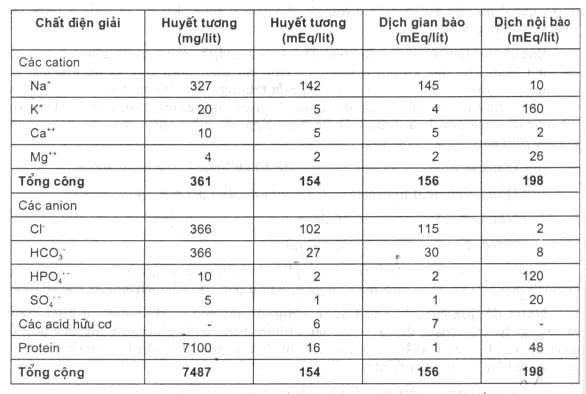
When a patient loses electrolytes, the proportion of electrolytes lost is not the same. Therefore, testing is needed to determine which electrolytes the patient has lost and how much needs to be supplemented, based on which appropriate electrolyte solution can be prescribed. In this case, the electrolyte loss solution that needs to be infused to the patient can be prescribed in the form of:
Rp .
Na + 147 mEq
K + 4 mEq
Ca 2+ 4 mEq
Cl - 155 mEq
Distilled water 1000 ml
Mf infusion
With this prescription method, when preparing the mixture, the pharmacist must choose a salt with the appropriate anion and must calculate the amount of salt to be used for the prescription. In case the solution is not isotonic, add glucose to make the solution isotonic.
Some electrolyte solutions are widely used in treatment and are mass produced:
0.9% sodium chloride solution:
Formula: Sodium chloride 9 g
Distilled water for injection v.v. 1000 ml
Sodium chloride solution is an isotonic solution, with a pH of 4.5 - 7.0, injected to restore the electrolyte balance of the extracellular fluid when the patient has lost electrolytes due to burns, vomiting, diarrhea, or prolonged high fever. The solution is also used in large quantities for peritoneal dialysis for patients with severe renal failure.
Ringer's solution :
Formula: Sodium chloride 8.6 g
Potassium chloride 0.3 g
Calcium chloride 0.33 g
Distilled water for injection v.v. 1000 ml
Ringer's solution has a pH of 5.0 - 7.5, used for infusion to provide water and electrolytes.
Ringer-lactate solution:
Formula: Sodium chloride 6.0 g
Potassium chloride 0.3 g
Calcium chloride 0.2 g
Sodium lactate 3.1 g
Water for injection v.v. 1000 ml
Also known as Hartmann's solution, the solution has a pH of 6.0 - 7.5. Ringer's lactate solution is a polyelectrolyte solution that is widely used in treatment because when the body loses electrolytes, it often loses many electrolytes at the same time. The lactate ion in this solution is converted in the liver into glycogen, creating CO2 and water, consuming H + ions , so it has the effect of alkalizing the blood.
5.3. Infusion solutions to restore acid-base balance
Normal human plasma has a pH of 7.35 - 7.45 and is maintained stable within this pH range thanks to many physiological buffer systems available in the body. However, for some reason, plasma pH < 7.35 means the blood is acidic; conversely, if pH > 7.45 means the blood is alkaline. In these cases, it is necessary to infuse solutions that can restore the acid-base balance of the blood.
5.3.1. Some infusion solutions used when blood is contaminated with acid 1.4% sodium bicarbonate solution:
Formula: Sodium bicarbonate 14 g Distilled water for injection q.s. 1000ml
Sodium bicarbonate in aqueous solution, under the influence of heat during sterilization, decomposes to produce CO2 gas and reduces the concentration of HCO3- , increasing the alkalinity of the preparation . Therefore, when preparing this infusion solution , it is necessary to apply many synchronous measures to maintain the correct content of HCO3- ions in the preparation.
To keep the above reaction from right to left (i.e. keeping the substance in the form of NaHCO 3 ), the CO 2 concentration in the solution must be increased by bubbling CO 2 gas before bottling or by creating endogenous CO 2 by reacting HCl acid with an excess amount of NaHCO 3 calculated in the formula.
At the same time, CO2 must not be lost during the sterilization process as well as during the preservation of the preparation. To do this, the medicine must be sealed in a glass bottle with a stopper.
Close tightly, turn the bottle upside down when sterilizing, after sterilization, let the medicine cool before taking it out, shake the bottle vigorously to dissolve CO2 back into the solution .
Sodium lactate solution:
Formula: Lactic acid 14 ml
Sodium hydroxide 6.7 g
Dilute hydrochloric acid qs. Distilled water for injection qs. 1000 ml
Because sodium lactate is unstable, easily decomposed and discolored during storage, to prepare sodium lactate injection solution, people often start from lactic acid and sodium hydroxide. Dissolve sodium hydroxide in 400ml of water, add lactic acid and steam at 115 o C for 1 hour, let cool, add enough diluted HCl acid to pH 5.0 - 7.0, add enough water to make 1 liter, filter, bottle and sterilize at 121 o C for 30 minutes.
The solution contains 1.75–1.95% sodium lactate. The infusion of this solution is intended to provide a direct source of sodium ions to increase the concentration of hydrocarbonate ions in cases of severe acidosis of the blood. Lactate ions are rapidly converted in the liver to hydrogen.
5.3.2. Infusion solution used when blood is alkaline 2.14% ammonium chloride solution:
Formula: Ammonium chloride 21.4 g Distilled water for injection q.s. 1000 ml Sterilize the preparation at 115 o C for 30 minutes
The solution contains 400 mEq/liter of ammonium ion and chloride ion, with a pH of 4.5 - 6.0, and is infused slowly intravenously (500ml in 3 hours) to restore balance when the blood is alkaline due to metabolism. When this solution is infused into the blood, ammonium chloride passes through red blood cells, easily causing hemolysis. To ensure safety, glucose is often added to this solution, because glucose has the effect of stabilizing red blood cell membranes.
5.4. Infusion solutions providing nutrients
In cases where the patient cannot receive the body's nutritional needs from food through the digestive tract due to certain medical reasons. At that time, the need for essential nutrients such as carbohydrates, amino acids, fatty acids, minerals, trace elements and vitamins is provided by injecting the patient with intravenous solutions containing the necessary nutritional ingredients.
To provide energy (calories), 10%, 20%, 30% glucose solutions or 5% glucose solution combined with 5% ethanol can be injected or D/N fat emulsion containing:
Soybean oil 100 g
Egg yolk phospholipid 12 g
Glycerin 22.5 g
Water for injection v.v. 1000 ml
Soybean oil contains many fatty acids such as linoleic acid (50%), oleic acid (26%) and palmitic, linolenic, stearic acids, etc. These fatty acids are the main components that provide energy for the body. Egg yolk phospholipid is a natural emulsifier, it does not change the permeability of the vascular wall as well as the blood cell membrane. Glycerin and water are necessary components to form an emulsion. This is a D/N infusion emulsion, so the size of the dispersed droplets of the oil phase must be controlled <0.5µm to avoid vascular accidents during infusion.
5.5. Blood volume replenishment solutions
In cases where the patient has lost blood due to trauma, surgery or internal bleeding. Depending on the amount of blood lost, the patient may experience hypotension, heart failure and possibly death if the lost blood volume is not replaced promptly. In cases where there is no blood to transfuse, blood volume can be temporarily compensated by injecting some solutions of substances with high molecular weight. When infusing these solutions, because the solutes have large molecular size, it is difficult to diffuse through the vascular wall, so they remain in the vascular lumen longer than when infusing solutions with small molecular weight solutes such as glucose.
Some commonly used solutions:
- Dextran solution.
- Dextran 40 infusion solution.
- Dextran 70 infusion solution.
5.6. Osmotic diuretic infusion solutions
Mannitol solutions with concentrations: 10, 15, 20 and 25% in distilled water for injection, pH 4.5 - 7.0 or mannitol solutions combined with glucose or sodium chloride, are infusion solutions with osmotic diuretic effects, used for infusion in the treatment of cerebral edema and diuresis.
Mannitol solutions with concentrations of 20% or more are saturated solutions, so the preparation must be stored at a temperature of 20 - 30 o C. If the room temperature drops below 20 o C, the mannitol in the solution will crystallize. When infusing, if the solution contains crystalline mannitol, the solution must be heated so that the mannitol dissolves in the solution before being infused into the patient, and the solution must be passed through a blood filter (included in the blood transfusion set) to completely remove crystalline mannitol.
5.7. Anticoagulant and blood preservation solutions
When the body loses blood, injecting high molecular solutions such as dextran can only temporarily replenish the body's missing plasma volume, but cannot replenish the number of lost cells, especially when the body loses a large amount of blood. In such cases, it is best to transfuse blood for the patient.
To perform a blood transfusion, anticoagulant and preservative solutions are required.
blood.
ACDP solution (USP26):
Anhydrous citric acid 2.99 g
Sodium citrate dihydrate 26.3 g
Monosodium phosphate monohydrate 2.22 g
Dextrose monohydrate 25.5 g
Distilled water for injection 1000 ml Or mix according to the formula:
Citric acid monohydrate 3.27 g
Sodium citrate anhydrous 23.06 g
Monosodium phosphate anhydrous 1.93 g
Anhydrous Dextrose 23.2 g
Distilled water for injection v.v. 1000 ml
The solution has pH = 5.0 - 6.0; 14ml of solution is obtained from 100ml of blood.
5.8. Some other solutions
There are some solutions such as surgical irrigation solutions, peritoneal dialysis or hemodialysis solutions. Although these solutions are not intended for infusion, they are used in very large volumes and are in prolonged direct contact with internal body tissues (e.g. in the abdomen). Therefore, these solutions must also be isotonic, sterile, pyrogen-free, antiseptic-free and must be prepared, manufactured and quality-controlled like parenteral drugs.
VALUATION
I. Fill in the blanks:
1. Injectable drugs are preparations...(A)...., which may be in ..(B).... form, which may be in powder form.
2. Intravenous drugs are ...(A).... or oil-in-water emulsion ....(B)...., free of pyrogens, ...(C).... bacteria, free of antiseptics, isotonic with blood, used for intravenous infusion with .....(D).......
3. Based on the dispersion system, injectable drugs can be divided into:
a. Injection solution B. ...............
C. Powder injection D. ...............
4. The injection must......(A)...., if the injection is not...(B).... will cause serious consequences.
5. Pyrogens are.....(A)....produced by microorganisms in their ...(B)....
6. Plastic injection packaging has the advantage of...(A)... the impact of many types of...(B)...
7. Benzalkonium chloride is a...(A)... with properties....(B)....so in addition to its effect...(C).... it also has the effect of increasing the solubility of drugs that are difficult to dissolve in solvents.
8. If the room temperature drops to ..(A)..., the mannitol in the solution will ..(B)....
9. If the volume of an infusion drug labeled is ..(A)... 50ml, then the allowable difference is ..(B)...
10. When using intravenous drugs as a “carrier” to deliver other drugs into the body, attention must be paid to the ............
II. Choose true or false:
1. Using drugs by injection cannot localize the drug's effect at the injection site. Answer:
2. Infusion solutions are usually hypertonic solutions compared to blood. Answer:
3. Hypotonic injection solution can be injected subcutaneously or intramuscularly in small doses. Answer:
4. The injectable suspension is not sterilized after preparation.
5. The composition of the injectable suspension always contains an additional antiseptic.
6. Pyrogenic substances can be removed from injection preparation equipment by drying the equipment at a high temperature above 250 0 C for 30 minutes. Answer
7. The injectable container is not a component of a complete injectable preparation.
8. Intravenous emulsions can only be oil-in-water emulsions. Answer
9. When preparing Barbituric injections, distilled water without dissolved oxygen must be used. Answer:
10. Benzylic alcohol is both an antiseptic and anesthetic, suitable for oily injections such as Vitamin A and E.
III. Choose the best answer:
1. Intravenous injections must be prepared in the form of
A. Aqueous solution. B. Oil solution
C. Suspension D. Emulsion N/D
2. The route of injection with 100% bioavailability is:
A. Intramuscular injection B. Intradermal injection
C. Intravenous injection D. Subcutaneous injection
3. Infusion solution with osmotic diuretic effect that can be given to patients with high blood pressure and cerebral edema is:
A. Dextran 70 solution B. Fructose 10% solution
C. 10% Mannitol solution D. Ringer's solution
4. The method of sterilizing plastic injection containers is:
A. Dry in the cabinet B. Use Ethylene oxide gas
C. Steaming in an autoclave D. UV irradiation
5. The appropriate sterilization method for sterilizing oils used as solvents for injections is:
A. Steam in an autoclave at 120 0 C for 30 minutes
B. Filter out bacteria using a filter with 0.22 micrometer pores
C. Using Ethylene oxide gas
D. Dry at 160 0 C for 1 hour
6. Which of the following substances is not an antioxidant for oil injections:
A. Tocoferol B. Rongalid
C. Butylhydroxytoluene D. Butylhydroxyanisone
7. Which of the following substances is not an antioxidant for aqueous injections:
A. Tocoferol B. Ascorbic Acid
C. Cysteine D. Thiourea
8. Buffer systems not used to adjust pH in injectable formulations are buffer systems:
A. Acetic/acetate B. Boric/borate
C. Citric/ citrate D. Glutamic/ Glutamate
9. Which PEG can be used as a solvent for preparing injectable drugs:
A. PEG 400 B. PEG 4000
C. PEG 1540 D. PEG 10000
10. According to Vietnamese Pharmacopoeia III, water used to mix injections is:
A. Demineralized water B. Demineralized water
C. Distilled water D. Distilled water within 24 hours
IV. Answer the following questions:
1. Analyze the formula, method and process of preparing the following injectable drugs:
a. Amikacin sulfate 50mg/ml, 2ml vial (Elkins – Sinn, USP 24):
Amikacin sulfate 100ng
Sodium bisulfite 0.13%
Sodium citrate 0.5%
Sulfuric acid to adjust pH = 3.5 – 5.5 (best is 4.5)
Sterile water for injection q.s. 2ml
b. Ergonovine maleate, 1ml syringe (Lilly):
Ergonovine maleate 0.2mg
Ethyl lactate 0.1%
Lactic acid 0.1%
Phenol 0.25%
Sterile water for injection for 1ml Injection has pH = 2.7 - 3.5.
2. Analyze the formula, method and process of preparing the following injectable drugs:
a. ACDP solution (USP26):
Anhydrous citric acid 2.99 g
Sodium citrate dihydrate 26.3 g Monosodium phosphate monohydrate 2.22 g Dextrose monohydrate 25.5 g
Distilled water for injection v.v. 1000 ml
b. Fat emulsion:
Soybean oil 100 g
Egg yolk phospholipid 12 g
Glycerin 22.5 g
Water for injection v.v. 1000 ml
3. Describe the process of preparing an injectable solution? Give an example.
4. Describe the methods of sterilization of injectable and infusion drugs?
5. Describe the isotonic substances used in the preparation of injectable and infusion drugs?
6. Describe how to sterilize plastic and glass packaging in the preparation of intravenous drugs?





