Patient compliance with treatment:
Treatment adherence is considered the most important component of successful ARV treatment at both the individual and program levels. Poor treatment adherence is a predictor of virological treatment failure [23], increased emergence of drug-resistant HIV mutations, increased disease progression, and mortality [24].
At the individual level, studies suggest that depression, addiction, poor understanding of the disease and ARV treatment, adolescents or young adults, and use of multiple single-pill, multiple-dose medications may be associated with adherence [67]. Adherence to treatment may be particularly difficult in children for a variety of reasons, including drug formulation and palatability. In addition, children are at higher risk of developing drug-resistant HIV mutations because their treatment often depends on their caregivers. Adherence is also negatively affected by HIV stigma and discrimination [88]. People living with HIV may fear taking medication in front of others, which may lead to inadvertent disclosure of their HIV status, thus preventing them from following their prescribed regimen [92].
Factors related to HIV/AIDS treatment programs
Overcrowded or understaffed HIV/AIDS treatment facilities may not have enough time to provide counseling and reinforce adherence messages. Reduced quality of counseling and patient follow-up in ARV treatment facilities may increase loss to follow-up, leading to higher rates of treatment discontinuation and more people with unknown treatment outcomes [33].
In addition, weak drug procurement and supply management systems can lead to drug stockouts and ARV shortages [78]. In resource-limited countries, difficulties in accessing continuous ARV treatment and interruptions in receiving medication are major barriers to increasing the risk of developing drug-resistant HIV mutations [66]. Other factors such as food insecurity, high travel costs, and patients having to pay for treatment monitoring tests themselves can lead to treatment interruptions, reduced adherence, and increased HIV drug resistance [120].
1.3. The situation of HIV drug resistance prevention and surveillance in the world
1.3.1. Early warning indicators for HIV drug resistance (EWI)
The HIV early warning index (EWI) reflects the status related to the possibility of emergence of HIV drug resistance mutations during ARV treatment at the treatment facility level and ARV treatment program level [42].
These indicators include:
EWI 1 – Guidelines-based ARV prescribing: This indicator assesses the prescribing practices of physicians. If physicians prescribe ineffective single-drug or dual-drug regimens or combinations of ARVs, this will lead to the rapid emergence of selected drug-resistant HIV mutations during ARV treatment.
EWI 2: Lost to follow-up within 12 months of ARV treatment: When a person on ARV treatment is lost to follow-up, it causes treatment interruption. Treatment interruption leads to failure to suppress HIV replication, which over time leads to the emergence of drug-resistant HIV mutations. Treatment interruptions for NNRTI ARVs of ≥ 48 hours have been associated with the emergence of drug-resistant HIV mutations in the patient population receiving regimens containing this class of drugs [78].
EWI 3: Maintaining first-line ARV regimen beyond 12 months: In many developing countries, alternative regimens are very limited, including second-line ARV regimens. Therefore, maintaining an effective first-line ARV regimen for HIV is an essential requirement for maintaining the success of an ARV treatment program.
EWI 4 - timely follow-up visits and EWI 5 - timely medication collection are indicators to assess patient adherence to ARV treatment. Failure to follow-up visits and medication collection on time will lead to treatment interruption. Adherence to timely medication collection is predicted to be associated with unsuppressed HIV viral load and the emergence of drug-resistant HIV mutations [30].
EWI 6: Continuous ARV drug supply: When ARV drug supply is not continuous, it leads to interruption of ARV treatment, which in turn leads to treatment failure and promotes the emergence of drug-resistant HIV mutations.
EWI 7: Count the number of ARV pills remaining. This is an indicator of adherence.
treatment adherence of patients. However, this index is very difficult to implement.
EWI 8: HIV viral load after 12 months of treatment is below 1000 copies/ml. This is the suppression threshold and if the patient reaches this threshold, it is considered to have achieved HIV drug resistance prevention [103]. This is a direct indicator of the success of the ARV treatment regimen. The EWI indicators and targets for each indicator recommended by WHO are presented in Table 1.5 [103].
Table 1.5. WHO recommended early warning indicators for HIV drug resistance
TT
Index | Target | |
1 | Practice standard prescribing according to guidelines nation | 100% of patients prescribed as directed national lead |
2 | Lost to follow-up at 12 months post-treatment ARV treatment | ≤20% of patients lost to follow-up |
3 | Maintain first-line ARV regimen at 12 months post-treatment | ≥70% of patients still maintain the regimen level 1 |
4 | Get your medication on time | ≥ 90% of patients receive the correct medication appointment |
5 | Re-examination on time | ≥ 80% of patients return for check-ups on time |
6 | Continuous supply of ARV drugs | 100% of treatment facilities ensure adequate stock of drugs at any given time |
7 | Treatment adherence through counting remaining pill | ≥ 90% of patients took the full dose pills given |
8 | HIV viral load below 1000 copies stars/ml | ≥ 70% of patients have HIV viral load at <1000 copies/ml |
Maybe you are interested!
-
 Description of the Current Situation of Early Warning of HIV Drug Resistance at Treatment Facilities
Description of the Current Situation of Early Warning of HIV Drug Resistance at Treatment Facilities -
 Study on HIV drug resistance in patients receiving first-line ARV treatment in some provinces and cities - 2
Study on HIV drug resistance in patients receiving first-line ARV treatment in some provinces and cities - 2 -
 Current status of HIV infection, AFB + tuberculosis and evaluation of preventive intervention effectiveness among drug addicts in Dak Lak province - 1
Current status of HIV infection, AFB + tuberculosis and evaluation of preventive intervention effectiveness among drug addicts in Dak Lak province - 1 -
 Risk of HIV infection and effectiveness of preventive intervention among drug addicts in Quang Nam - 1
Risk of HIV infection and effectiveness of preventive intervention among drug addicts in Quang Nam - 1 -
 Draw the Frame or General Formula of Each Drug Group (If Any); Structural Formula of Substances in the Chapter.
Draw the Frame or General Formula of Each Drug Group (If Any); Structural Formula of Substances in the Chapter.
Source: The WHO's global strategy for prevention and assessment of HIV
drug resistance, 2008, [117].
Since 2004, 50 countries have collected early warning indicators of HIV drug resistance.
to assess HIV drug resistance prevention in treatment facilities [42], [101].
Table 1.6. Distribution of HIV treatment facilities according to the results of warning indicators
WHO-recommended HIV drug resistance by region, 2004-2009
Index
EWI 1: Standard Prescribing Practice | EWI 2: Lost Tracking | EWI 3: Maintain first-line ARV regimen after 12 month | EWI 4: Get medicine on time | EWI 5: Re-examination on time | EWI 6: Providing ARV drugs continuous | EWI 8: HIV viral load suppression after 12 months | ||
Target | 100% | ≤20% | ≥70% | ≥90% | ≥80% | 100% | ≥70% | |
Africa region (all) all years) | Number of establishments treatment | 907 | 794 | 863 | 321 | 309 | 537 | 24 |
% base achieve the goal | 74% | 59% | 61% | 15% | 43% | 63% | 96% | |
Asia (all years) | Number of establishments treatment | 1048 | 1043 | 1045 | 10 | 1037 | 100 | — |
% of establishments achieving the target pepper | 80% | 75% | 72% | 0% | 64% | 89% | — | |
Latin America and the Caribbean (all) year) | Number of establishments treatment | 141 | 116 | 132 | 21 | 20 | 86 | 22 |
% of establishments achieving the target | 46% | 85% | 71% | 57% | 15% | 51% | 73% | |
Total (all regions, all year) | Number of establishments treatment | 2096 | 1953 | 2040 | 352 | 1366 | 723 | 46 |
% of establishments achieving the target | 75% | 68% | 67% | 17% | 57% | 65% | 85% | |
Source: WHO HIV Drug Resistance Report 2012, [101]
Assessments were conducted on 131,686 patients at 2107 treatment facilities.
ARVs are mainly in Africa and Asia.
EWI 1, 2, and 3 (standard prescribing practices, loss to follow-up, and retention on first-line ARV regimen at 12 months, respectively) were the three most commonly monitored indicators[101]. Despite their strong association with HIV-related morbidity, only a few treatment facilities reported on-time follow-up and timely medication refilling.
appointments (EWI 4 and EWI 5). Half of the countries reported continuous ARV supply (EWI6). EWI 7 (assessing adherence using pill counts) was rarely implemented in practice. Few treatment facilities reported EWI 8 because most countries had limited access to routine HIV viral load testing for clinical monitoring purposes.
According to the table above, it can be seen that 75% of the facilities achieved the WHO target for standard prescribing indicators. Facilities in the Asian region had the highest rate of facilities achieving the WHO target (80%), while the lowest was in Latin America and the Caribbean. This is explained by the fact that in the Caribbean region, treatment facilities have a more individualized approach to treatment than a community approach [131]. 68% of treatment facilities achieved the target of having less than 20% of patients on ARV treatment lost to follow-up after 12 months of ARV treatment, with the highest in the Caribbean (85%) and the lowest in Africa (59%). 67% of treatment facilities achieved the recommended level for EWI 3 (maintaining first-line ARV treatment), ranging from 60%-70% of patients still maintaining first-line ARV treatment. The index of timely follow-up visits and timely medication collection are two indicators that very few facilities reported, and for facilities that reported on this index, only 17% of treatment facilities achieved the WHO target. Notably, 35% of treatment facilities did not continuously supply ARV drugs. EWI 8 is an indicator that directly monitors the ability to suppress HIV of the ARV regimen at 12 months after treatment. However, very few treatment facilities (46 facilities) achieved this indicator due to limited resources in most countries. However, one point to note is that at facilities reporting on this indicator, the rate of facilities achieving the WHO target is quite high (85%). This shows the effectiveness of the ARV treatment program in those facilities.
EWI results in some countries have shown the limitations of countries in program management in some specific indicators such as EWI 4, EWI 6. Reports show that the main limitations of the current data monitoring system are incomplete medical records, lack of data, non-standard data recording, and different data recording between facilities [42]. Some specific cases show that the limitations discovered from EWI results show that there are issues that need to be addressed.
need to be improved immediately, such as the management and monitoring of ARV drug supply [85]. Based on the results of EWI, including rates of loss to follow-up, missed follow-up appointments, and missed drug refills, countries need to implement measures to improve the quality of patient management and monitoring, and reduce loss to follow-up.
Several countries have undertaken follow-up to assess factors influencing EWI outcomes such as adherence factors including treatment or medication costs [95], distance and lack of transport [95], [64], long waiting times for medication [64], and stigma and discrimination [85]. All of these factors are important barriers to continued ARV treatment in resource-poor countries.
In China, the EWI results suggest that further research is needed to understand the reasons why patients are late in receiving their medications, and the issues that affect patient adherence to treatment [110]. In Papua New Guinea, based on the EWI results, health care workers at treatment facilities discussed and agreed on key measures to improve ARV treatment services: (1) Establish a formal referral system to ensure the successful transfer of patient treatment information between treatment facilities; (2) Regularly review patients' timely follow-up visits and timely medication collection; (3) identify ways to reduce barriers to medication collection through transportation and food assistance for those in need [37]. The EWI results in the Caribbean have provided evidence to inform decision-making and policy related to reducing HIV drug resistance. Proper practice of combining first- and second-line ARV drugs makes an important contribution to preventing HIV drug resistance right from the start of ARV treatment, protecting cost-effective second-line ARV regimens in the future [131].
Testing for drug resistance is not required, but EWIs provide information that informs the interpretation of results from drug resistance surveys of transmitted and acquired HIV. EWI results from representative samples
or all domestic facilities can help detect major program problems.
treatment and reduce the emergence of HIV drug resistance[107].
1.3.2. Acquired drug-resistant HIV in patients on ARV treatment
1.3.2.1 HIV drug resistance before starting ARV treatment
Results from a pooled study conducted by WHO on 6370 individuals recruited in 40 acquired drug resistance surveys from 2007 to 2010 [101], showed that 5.0% of individuals had one or more mutations to any ARV class before starting treatment, 4.5% had one or more resistance mutations to NRTI or NNRTI (3.7% NNRTI, 1.4% NRTI, 0.6% to both NNRTI and NRTI), and 0.6% had one
or multiple PI-resistant mutations .
Distribution of HIVKT mutations in the HIV-infected population before ARV treatment,
2007 – 2010 is presented in chart 1.1
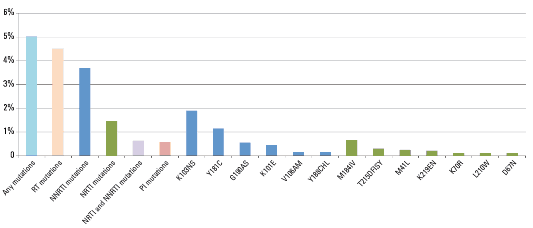
Figure 1.1. Distribution of HIV drug-resistant mutations in the HIV-infected population before starting ARV treatment, 2007 - 2010
Source: WHO HIV drug resistance report 2012, [101].
There has been an increase in the prevalence of drug-resistant HIV mutations in the pre-ART population[101]. The differences in the prevalence of mutations with ARV drug classes are detailed in Table 1.7.
Table 1.7. Prevalence of HIV drug resistance at the start of ARV treatment in the WHO survey (n = 36), by survey year and drug class, 2007-2010
Punch type
variable
Prevalence of HIV drug-resistant mutations% (95% CI) | |||||
2007 | 2008 | 2009 | 2010 | p-value | |
Mutation any | 4.8 (3.8–6.0) | 3.9 (3.0–4.9) | 4.6 (2.2–7.8) | 6.8 (4.8–9.0) | 0.06 |
NRTI | 1.2 (0.7–2.0) | 1.3 (0.8–2.0) | 1.1 (0.3–2.2) | 1.0 (0.3–2.1) | 0.70 |
NNRTI | 3.7 (2.5–4.9) | 2.4 (1.6–3.3) | 3.3 (1.8–5.1) | 5.5 (3.8–7.4) | 0.06 |
PI | 0.3 (0.0–0.7) | 0.4 (0.1–0.8) | 0.5 (0.0–1.7) | 0.0 (0.0–0.4) | 0.97 |
Source: WHO HIV drug resistance report 2012, [101]
The table above shows an increase in the prevalence of any mutation at pre-ART from 2008 to 2010, reaching 6.8% (95% CI 4.8%-9.0%) in 2010, mainly for NNRTIs and NRTIs. The mutation rate for PIs - which are mainly used in second-line regimens - is very low.
Among the African study sites, NNRTI resistance at baseline increased from 3.4% (95% CI 2.4%-4.5%) to 5.4% (95% CI 3.7%-7.4%) over the same period, a statistically significant increase (p = 0.03). This may be due to previous exposure to ARVs (prevention of mother-to-child transmission or previous ARV treatment) or to acquired resistance. Resistance mutations found were K103NS, Y181C, G190AS, K101E, V106AM, Y188CHL (NNRTI resistance), M184IV, T215DFISY, M41L, K219EN, K70R, L210W, D67N (NRTI resistance) [142].
In Cambodia, a 2009 study of 67 cases showed that the prevalence of HIV drug resistance at the start of ARV treatment was 1.49%[74]. In 2010, in China, Liao et al. reported the results of a survey on HIVKT in 1194 HIV-infected people not yet on ARV treatment in 28 provinces of China[43]. 26/676 cases (3.8%) had at least one HIVKT mutation,