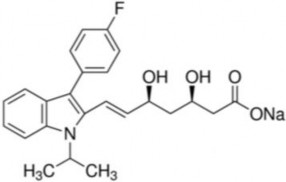
C 24 H 25 FNNaO 4 ptl: 433.40
Scientific name: Sodium (±)7-[3-(p-fluorophenyl)-1-isopropylindol-2-yl]-3,5-dihydroxy-6- heptanoate.
Nature:
Maybe you are interested!
-
 Car body electrical practice - 8
zt2i3t4l5ee
zt2a3gs
zt2a3ge
zc2o3n4t5e6n7ts
If the voltage is out of specification, replace the wire or connector.
If the voltage is within specification, install the front fog light relay and follow step 5.
Step 5 Check the front fog light switch
- Remove the D4 connector of the fog light switch
- Use a multimeter to measure the resistance of the front fog light switch.
Measurement location
Condition
Standard
D4-3 (BFG) -D4-4 (LFG)
Light switchFront Fog OFF
>10kΩ
D4-3 (BFG) -D4-4 (LFG)
Front fog light switchON
<1 Ω
- Standard resistor
D4 connector is located on the combination switch assembly.
If the resistance is out of specification, replace the combination switch (the fog light switch is located in the combination switch).
If the resistance is within specification, follow step 6.
Step 6 Check wiring and connectors (front fog light relay-light selector switch)
- Disconnect connector D4 of the combination switch assembly
- Use a voltmeter to measure the voltage value of jack D4 on the wire side.
Measurement location
Control modecontrol
Standard
D4-3 (BFG) - (-) AQ
TAIL
11 to 14 V
D4 connector for the wiring of the combination switch assembly
If the voltage does not meet the standard, replace the wire or connector.
If the voltage is within standard, there may have been an error in the previous measurements.
Step 7 Check the front fog lights
- Remove the front fog light electrical connector.
- Supply battery voltage to the fog lamp terminals
Jack 8, B9 of front fog lamp on the electrical side
blind first.
Power supply location
Terms and Conditions
Battery positive terminal - Terminal 2Battery negative terminal - Terminal 1
Fog lightsbefore morning
- If the light does not come on, replace the bulb.
If the light is on, re-plug the jack and continue to step 8.
Step 8 Check wiring and connectors (relay and front fog lights)
- Disconnect the B8 and B9 connectors of the front fog lights.
- Use a voltmeter to measure voltage at the following locations:
Measurement location
Switch location
Terms and Conditions
B8-2 - (-) AQ
Electric lock ON TAIL size switchFog switch ON
11 to 14 V
B9-2 - (-) AQ
Electric lock ONTAIL size switch Fog switch ON
11 to 14 V
B8 and B9 connectors on the front fog lamp wiring side
Voltage is not up to standard, repair or replace the jack. If up to standard, there may have been an error in the measurement process.
2.2.4. Procedure for removing, installing and adjusting fog lights 1. Procedure for removing
- Remove the front inner ear pads
Use a screwdriver to remove the 3 screws and remove the front part of the front inner ear liner
-Remove the fog light assembly
+ Disconnect the connector.
+ Use a screwdriver to remove 3 screws to remove the fog light cover
2. Installation sequence
-Rotate the fog lamp bulb in the direction indicated by the arrow as shown in the figure and remove the fog lamp from the fog lamp assembly.
-Rotate the fog light bulb in the direction indicated by the arrow as shown in the figure and install the light into the fog light assembly.
- Use a screwdriver to install the fog light cover
-Install the electrical connector
Attention: Be careful not to damage the plastic thread on the lamp assembly.
- Install the front inner ear pads
Use a screwdriver to install the front inner bumper with 3 screws.
3. Prepare the vehicle to adjust the fog light convergence. Prepare the vehicle:
- Make sure there is no damage or deformation to the vehicle body around the fog lights.
- Add fuel to the fuel tank
- Add oil to standard level.
- Add engine coolant to standard level.
- Inflate the tire to standard pressure.
- Place spare tire, tools and jack in original design position
- Do not leave any load in the luggage compartment.
- Let a person weighing about 75 kg sit in the driver's seat.
4. Prepare to check the fog light convergence
a/ Prepare the vehicle status as follows:
- Place the car in a dark enough place to see the lines. The lines are the dividing line, below which the light from the fog lights can be seen but above which it cannot.
- Place the car perpendicular to the wall.
- Keep a distance of 7.62 m between the center of the fog lamp and the wall.
- Park the car on level ground.
- Press the car down a few times to stabilize the suspension.
Note: A distance of approximately 7.62 m is required between the vehicle (fog lamp center) and the wall to adjust the convergence correctly. If the distance of 7.62 m cannot be achieved, set the correct distance of 3 m to check and adjust the fog lamp convergence. (Since the target area varies with the distance, please follow the instructions as shown in the figure.)
b/ Prepare a piece of thick white paper about 2 m high and 4 m wide to use as a screen.
c/ Draw a vertical line through the center of the screen (line V).
d/ Set the screen as shown in the picture. Note:
- Keep the screen perpendicular to the ground.
- Align the V line on the screen with the center of the vehicle.
e/Draw the reference lines (H, V LH and V RH lines) on the screen as shown in the figure.HINT:
Mark the center of the fog lamp on the screen. If the center mark cannot be seen on the fog lamp, use the center of the fog lamp or the manufacturer's name mark on the fog lamp as the center mark.
H line (fog light height):
Draw a line across the screen so that it passes through the center mark. Line H should be at the same height as the center mark of the fog light bulb.
Line V LH, V RH (center mark position of left fog lamp LH and right fog lamp RH):
Draw two lines so that they intersect line H at the center marks.
5. Check the fog light convergence
a/ Cover the fog lamp or remove the connector of the other side fog lamp to prevent light from the unchecked fog lamp from affecting the fog lamp convergence test.
b/ Start the engine.
c/ Turn on the fog lights and make sure that the dividing line is outside the standard area as shown in the drawing.
6. Adjust the fog light convergence
Use a screwdriver to adjust the fog light to the standard area by turning the toe adjustment screw.
Note: If the screw is adjusted too far, loosen it and then tighten it again, so that the last rotation of the light adjustment screw is clockwise.
3. Self-study questions
1. Describe the operating principle of the lighting system with automatic headlight function
2. Describe the operating principle of the lighting system with the function of rotating headlights when turning
3. Draw diagram and connect lighting system on Hyundai Porter car
4. Draw diagram and connect lighting system on Honda Accord 1992
5. Draw the lighting circuit on a 1993 Toyota Lexus
LESSON 3 MAINTENANCE AND REPAIR OF SIGNAL SYSTEM
I. IMPLEMENTATION GOAL
After completing this lesson, students will be able to:
- Distinguish between types of signals on cars
- Correctly describe common symptoms and suspected areas causing damage.
- Connecting signal circuits ensures technical requirements
- Disassemble, install, check, maintain and repair the signal system to ensure technical requirements.
- Ensure safety in work and industrial hygiene
II. LESSON CONTENT
1. General description
The signal system equipped on cars aims to create signals to notify other vehicles participating in traffic about the vehicle's operating status such as: stopping, parking, braking, reversing, turning...
Signals are used either by light such as headlamps, brake lights, turn signals….. or by sound such as horns, reverse music….
Just like the lighting system. A signal system circuit usually consists of: battery, fuse, wire, relay, electrical load and control switch. Only some switches of the signal system are on the combination switch. The switches of other signals are usually located in different locations such as in the gearbox or brake pedal……
2. Maintenance and repair
2.1. Turn signals and hazard lights
The installation location of the turn signal is shown in Figure 3.1. The turn signal control switch is located in the combination switch under the steering wheel. Turning this switch to the right or left will make the turn signal turn right or left.
The hazard light switch is used when the vehicle has a problem while participating in traffic. When the hazard light switch is turned on, all the turn signals on the vehicle will light up at a certain frequency. The hazard light switch is usually placed separately from the turn signal switch (some old cars integrate the hazard and turn signal switches on the same combination switch cluster).
Figure 3.1 Turn signal switch Figure 3.2 Hazard switch
The part that generates the flashing frequency for the lights is called a turn signal relay. The turn signal relay usually has 3 terminals: B (positive power supply); E (negative power supply); L (providing the turn signal switch to distribute to the
lamp)
2.1.1. Circuit diagram
To generate the frequency for the turn signal, a turn signal relay is used in the turn signal circuit. The current from the turn signal relay will be sent to the turn signal switch assembly to distribute the current to the turn signal lights for the driver's purpose.
Figure 3.3. Schematic diagram of a turn signal circuit without a hazard switch
1. Battery; 2. Electric lock; 3. Turn signal relay; 4. Turn signal switch; 5. Turn signal lamp; 6. Turn signal lamp; 7. Hazard switch
Figure 3.4 Schematic diagram of turn signal circuit with hazard switch
1. Battery; 2. Combination switch cluster; 3. Turn signal;
4. Turn signal light; 5. Turn signal relay
Today's cars no longer use three-pin turn signal relays (B, L, E) but use eight-pin turn signal relays (figure 3.5) (pin number 8 is used for hazard lights).
For this type, the current supplying the turn signal lights is supplied directly from the turn signal relay to the lights.
div.maincontent .p { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 14pt; margin:0pt; } div.maincontent p { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 14pt; margin:0pt; } div.maincontent .s1 { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 13pt; } div.maincontent .s2 { color: black; font-family:"Times New Roman", serif; font-style: italic; font-weight: normal; text-decoration: none; font-size: 14pt; } div.maincontent .s3 { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 14pt; } div.maincontent .s4 { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 13pt; } div.maincontent .s5 { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 13pt; vertical-align: 1pt; } div.maincontent .s6 { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 11pt; } div.maincontent .s7 { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 14pt; vertical-align: -9pt; } div.maincontent .s8 { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 11pt; } div.maincontent .s9 { color: #008000; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 14pt; } div.maincontent .s10 { color: black; font-family:"Times New Roman", serif; font-style: italic; font-weight: normal; te
Car body electrical practice - 8
zt2i3t4l5ee
zt2a3gs
zt2a3ge
zc2o3n4t5e6n7ts
If the voltage is out of specification, replace the wire or connector.
If the voltage is within specification, install the front fog light relay and follow step 5.
Step 5 Check the front fog light switch
- Remove the D4 connector of the fog light switch
- Use a multimeter to measure the resistance of the front fog light switch.
Measurement location
Condition
Standard
D4-3 (BFG) -D4-4 (LFG)
Light switchFront Fog OFF
>10kΩ
D4-3 (BFG) -D4-4 (LFG)
Front fog light switchON
<1 Ω
- Standard resistor
D4 connector is located on the combination switch assembly.
If the resistance is out of specification, replace the combination switch (the fog light switch is located in the combination switch).
If the resistance is within specification, follow step 6.
Step 6 Check wiring and connectors (front fog light relay-light selector switch)
- Disconnect connector D4 of the combination switch assembly
- Use a voltmeter to measure the voltage value of jack D4 on the wire side.
Measurement location
Control modecontrol
Standard
D4-3 (BFG) - (-) AQ
TAIL
11 to 14 V
D4 connector for the wiring of the combination switch assembly
If the voltage does not meet the standard, replace the wire or connector.
If the voltage is within standard, there may have been an error in the previous measurements.
Step 7 Check the front fog lights
- Remove the front fog light electrical connector.
- Supply battery voltage to the fog lamp terminals
Jack 8, B9 of front fog lamp on the electrical side
blind first.
Power supply location
Terms and Conditions
Battery positive terminal - Terminal 2Battery negative terminal - Terminal 1
Fog lightsbefore morning
- If the light does not come on, replace the bulb.
If the light is on, re-plug the jack and continue to step 8.
Step 8 Check wiring and connectors (relay and front fog lights)
- Disconnect the B8 and B9 connectors of the front fog lights.
- Use a voltmeter to measure voltage at the following locations:
Measurement location
Switch location
Terms and Conditions
B8-2 - (-) AQ
Electric lock ON TAIL size switchFog switch ON
11 to 14 V
B9-2 - (-) AQ
Electric lock ONTAIL size switch Fog switch ON
11 to 14 V
B8 and B9 connectors on the front fog lamp wiring side
Voltage is not up to standard, repair or replace the jack. If up to standard, there may have been an error in the measurement process.
2.2.4. Procedure for removing, installing and adjusting fog lights 1. Procedure for removing
- Remove the front inner ear pads
Use a screwdriver to remove the 3 screws and remove the front part of the front inner ear liner
-Remove the fog light assembly
+ Disconnect the connector.
+ Use a screwdriver to remove 3 screws to remove the fog light cover
2. Installation sequence
-Rotate the fog lamp bulb in the direction indicated by the arrow as shown in the figure and remove the fog lamp from the fog lamp assembly.
-Rotate the fog light bulb in the direction indicated by the arrow as shown in the figure and install the light into the fog light assembly.
- Use a screwdriver to install the fog light cover
-Install the electrical connector
Attention: Be careful not to damage the plastic thread on the lamp assembly.
- Install the front inner ear pads
Use a screwdriver to install the front inner bumper with 3 screws.
3. Prepare the vehicle to adjust the fog light convergence. Prepare the vehicle:
- Make sure there is no damage or deformation to the vehicle body around the fog lights.
- Add fuel to the fuel tank
- Add oil to standard level.
- Add engine coolant to standard level.
- Inflate the tire to standard pressure.
- Place spare tire, tools and jack in original design position
- Do not leave any load in the luggage compartment.
- Let a person weighing about 75 kg sit in the driver's seat.
4. Prepare to check the fog light convergence
a/ Prepare the vehicle status as follows:
- Place the car in a dark enough place to see the lines. The lines are the dividing line, below which the light from the fog lights can be seen but above which it cannot.
- Place the car perpendicular to the wall.
- Keep a distance of 7.62 m between the center of the fog lamp and the wall.
- Park the car on level ground.
- Press the car down a few times to stabilize the suspension.
Note: A distance of approximately 7.62 m is required between the vehicle (fog lamp center) and the wall to adjust the convergence correctly. If the distance of 7.62 m cannot be achieved, set the correct distance of 3 m to check and adjust the fog lamp convergence. (Since the target area varies with the distance, please follow the instructions as shown in the figure.)
b/ Prepare a piece of thick white paper about 2 m high and 4 m wide to use as a screen.
c/ Draw a vertical line through the center of the screen (line V).
d/ Set the screen as shown in the picture. Note:
- Keep the screen perpendicular to the ground.
- Align the V line on the screen with the center of the vehicle.
e/Draw the reference lines (H, V LH and V RH lines) on the screen as shown in the figure.HINT:
Mark the center of the fog lamp on the screen. If the center mark cannot be seen on the fog lamp, use the center of the fog lamp or the manufacturer's name mark on the fog lamp as the center mark.
H line (fog light height):
Draw a line across the screen so that it passes through the center mark. Line H should be at the same height as the center mark of the fog light bulb.
Line V LH, V RH (center mark position of left fog lamp LH and right fog lamp RH):
Draw two lines so that they intersect line H at the center marks.
5. Check the fog light convergence
a/ Cover the fog lamp or remove the connector of the other side fog lamp to prevent light from the unchecked fog lamp from affecting the fog lamp convergence test.
b/ Start the engine.
c/ Turn on the fog lights and make sure that the dividing line is outside the standard area as shown in the drawing.
6. Adjust the fog light convergence
Use a screwdriver to adjust the fog light to the standard area by turning the toe adjustment screw.
Note: If the screw is adjusted too far, loosen it and then tighten it again, so that the last rotation of the light adjustment screw is clockwise.
3. Self-study questions
1. Describe the operating principle of the lighting system with automatic headlight function
2. Describe the operating principle of the lighting system with the function of rotating headlights when turning
3. Draw diagram and connect lighting system on Hyundai Porter car
4. Draw diagram and connect lighting system on Honda Accord 1992
5. Draw the lighting circuit on a 1993 Toyota Lexus
LESSON 3 MAINTENANCE AND REPAIR OF SIGNAL SYSTEM
I. IMPLEMENTATION GOAL
After completing this lesson, students will be able to:
- Distinguish between types of signals on cars
- Correctly describe common symptoms and suspected areas causing damage.
- Connecting signal circuits ensures technical requirements
- Disassemble, install, check, maintain and repair the signal system to ensure technical requirements.
- Ensure safety in work and industrial hygiene
II. LESSON CONTENT
1. General description
The signal system equipped on cars aims to create signals to notify other vehicles participating in traffic about the vehicle's operating status such as: stopping, parking, braking, reversing, turning...
Signals are used either by light such as headlamps, brake lights, turn signals….. or by sound such as horns, reverse music….
Just like the lighting system. A signal system circuit usually consists of: battery, fuse, wire, relay, electrical load and control switch. Only some switches of the signal system are on the combination switch. The switches of other signals are usually located in different locations such as in the gearbox or brake pedal……
2. Maintenance and repair
2.1. Turn signals and hazard lights
The installation location of the turn signal is shown in Figure 3.1. The turn signal control switch is located in the combination switch under the steering wheel. Turning this switch to the right or left will make the turn signal turn right or left.
The hazard light switch is used when the vehicle has a problem while participating in traffic. When the hazard light switch is turned on, all the turn signals on the vehicle will light up at a certain frequency. The hazard light switch is usually placed separately from the turn signal switch (some old cars integrate the hazard and turn signal switches on the same combination switch cluster).
Figure 3.1 Turn signal switch Figure 3.2 Hazard switch
The part that generates the flashing frequency for the lights is called a turn signal relay. The turn signal relay usually has 3 terminals: B (positive power supply); E (negative power supply); L (providing the turn signal switch to distribute to the
lamp)
2.1.1. Circuit diagram
To generate the frequency for the turn signal, a turn signal relay is used in the turn signal circuit. The current from the turn signal relay will be sent to the turn signal switch assembly to distribute the current to the turn signal lights for the driver's purpose.
Figure 3.3. Schematic diagram of a turn signal circuit without a hazard switch
1. Battery; 2. Electric lock; 3. Turn signal relay; 4. Turn signal switch; 5. Turn signal lamp; 6. Turn signal lamp; 7. Hazard switch
Figure 3.4 Schematic diagram of turn signal circuit with hazard switch
1. Battery; 2. Combination switch cluster; 3. Turn signal;
4. Turn signal light; 5. Turn signal relay
Today's cars no longer use three-pin turn signal relays (B, L, E) but use eight-pin turn signal relays (figure 3.5) (pin number 8 is used for hazard lights).
For this type, the current supplying the turn signal lights is supplied directly from the turn signal relay to the lights.
div.maincontent .p { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 14pt; margin:0pt; } div.maincontent p { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 14pt; margin:0pt; } div.maincontent .s1 { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 13pt; } div.maincontent .s2 { color: black; font-family:"Times New Roman", serif; font-style: italic; font-weight: normal; text-decoration: none; font-size: 14pt; } div.maincontent .s3 { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 14pt; } div.maincontent .s4 { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 13pt; } div.maincontent .s5 { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 13pt; vertical-align: 1pt; } div.maincontent .s6 { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 11pt; } div.maincontent .s7 { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 14pt; vertical-align: -9pt; } div.maincontent .s8 { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 11pt; } div.maincontent .s9 { color: #008000; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 14pt; } div.maincontent .s10 { color: black; font-family:"Times New Roman", serif; font-style: italic; font-weight: normal; te -
 General Theoretical Basis of Corporate Finance and Corporate Financial Analysis.
General Theoretical Basis of Corporate Finance and Corporate Financial Analysis. -
 Organizing Capacity Training to Develop School Education Programs According to the New General Education Program for Management and Teaching Staff
Organizing Capacity Training to Develop School Education Programs According to the New General Education Program for Management and Teaching Staff -
 General Accounting for Increase and Decrease of Fixed Assets
General Accounting for Increase and Decrease of Fixed Assets -
 General Overview of Agribank Branch in Trieu Son District - Thanh Hoa
General Overview of Agribank Branch in Trieu Son District - Thanh Hoa
White to yellow crystalline powder, hygroscopic; soluble in water, ethanol; soluble in methanol.
Uses:
Inhibits HMG-CoA reductase; exhibits direct action, without hydrolysis like lovastatin; completely absorbed in the gastrointestinal tract.
Indications: Similar to lovastatin.
Dosage: Adults, take 20-40mg/time/24 hours in the evening. Undesirable effects and precautions: Similar to lovastatin. Storage: Avoid moisture and light.
206
LESSON 25. HORMONES AND ANALOGUES
LESSON OBJECTIVE:
1. Draw the framework or general formula of each drug group (if any); structural formula of the substances in the chapter.
2. Present the physical and chemical properties and qualitative and quantitative tests; effects and uses of each specific drug.
3. Describe the relationship between the structure and effects of steroid hormones; the mechanism of action and indications for each group of drugs used to treat diabetes.
This chapter will present some female sex hormones, male sex hormones, adrenal cortex hormones, pituitary hormones, thyroid hormones, and pancreatic hormones.
Female sex hormones (estrogen, progestin), male sex hormones (androgen), and adrenal cortex hormones (corticosteroids) all have steroid structures, according to the following three basic frameworks:
1. FEMALE SEX HORMONES
1.1. Estrogens
1.1.1. Natural estrogens
In the body, the main hormone is estradiol, produced by the ovaries, this substance is converted into estrone, estriol and some other substances such as equilin equilenin. The metabolites still have the effects of estradiol but are significantly weaker than estrone. The effects of estradiol are collectively called estrogenic effects.
Estron (Foliculin) is used as an injectable solution in oil, now commonly used as the piperazine sulfate ester salt under the name estropipate (Ogen, Ortho-est) for oral use in tablet form.
Estradiol, about eight times more potent than estrone, is most commonly used as the alcohol (Estrace, Climara) or as an ester, usually estradiol monobenzoate. Estrogens have an aromatic ring on the A ring.
ESTRADIOL MONOBENZOATE
Brand name : Benzo-gynoestryl, Progynon-depot
207
Recipe :
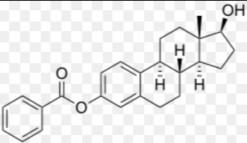
C 25 H 28 O 5 ptl: 376.5
Scientific name : 3,17β-dihydroxy-1,3,5(10)- estratrien-3-benzoate.
Properties :
White crystalline powder, odorless. Practically insoluble in water, slightly soluble in alcohol, in oil, easily soluble in acetone.
Qualitative :
Melting point measurement: 191-198 o C Using IR spectrum.
Reaction with sulisbmolybdic reagent (1 m in 5 ml of reagent) gives a yellowish blue color, examined under 365 UV lamp has green fluorescence, adding dilute H 2 SO 4 (1 ml H 2 SO 4 and 9 ml of water) gives a pink color and yellow fluorescence.
Perform TLC to identify and test for related impurities.
Quantitative
Measure the absorbance at λmax=231 nm in alcohol medium, calculate the result according to A(1%, 1 cm)=500.
Uses:
Stable (3)-benzoate ester form (-OH phenol lock), has suitable pharmacokinetics, gradually releases estradiol in the body.
Estradiol monobenzoate is more therapeutically potent than estrone or estropipate (which are salts of esters).
(estrone, estriol, equilin . . . ) still have an effect, so estradiol monobenzoate has a long-lasting effect (several days).
Assign:
208
Used as replacement therapy after menopause, ovarian failure, after oophorectomy. Replaces estrone or etropipate in most cases.
For intramuscular (IM) injection in ethyloleate or a suitable ester or in oil. Usually 0.1% concentration, 1 mg or 5 mg ampoules.
Dosage: Depends on the specific case, for example:
Postmenopausal disorders, anti-postmenopausal osteoporosis... 5 mg/time/3-4 weeks. Also used in tablet form (usually 0.1 mg tablets) to take orally at a dose of 0.3-0.6 mg/24 hours, depending on the case.
1.1.2. Semi-synthetic estrogens
General characteristics: More persistent in metabolic effects, stronger and often longer lasting, but more frequent and stronger adverse effects.
Ethinylestradiol and derivatives.
ETHINYLESTRADIOL
Brand name : Estinyl; Progynon; Estigyn
Recipe :
C 20 H 24 O 2 equation: 296.41
Scientific name : Dihydroxy-3β, 17β-ethiny-17α-estratrien 1,3,5(10). Is 17α-ethinylestradiol.
Preparation : Semi-synthetic according to the reaction:
209
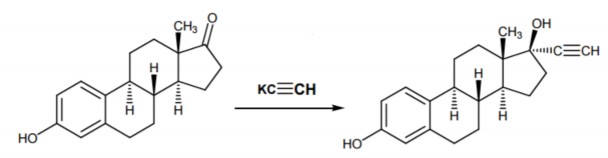
Estrone Ethinyl estradiol
Properties :
The preparation is in the form of a white to cream colored refined powder. Insoluble in water, soluble in alcohol, ether, chlorobrom, in alkali metal hydroxide solution.
Melting point: 181 – 185 o C (if the preparation is in polymorphic form, the melting point is 142 – 146 o C).
Qualitative:
Test by TLC, together with relevant impurity test.
Color reaction with H 2 SO 4 (1 mg of preparation/1 ml of concentrated H 2 SO 4 ): gives orange-red color, examines under 365 UV lamp with green fluorescence; dilute with water (add 10 ml), gives purple color and purple precipitate.
20
UV spectrum has Amax=281 nm with A(1%,1 cm)=69-73(C 0.01 , MeOH). It is a choleretic agent with [α] D =-274 – 30 (C 5 , pyridine).
Related impurities test : By TLC, the main impurity was estrone.
Quantitative :
Based on the reaction to create silver salt AgNO 3 , releasing HNO 3 ; quantify HNO 3 with 0.1N NaOH; determine the end point with a potentiometer.
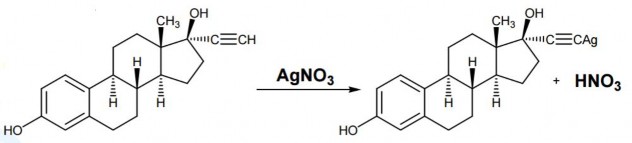
Uses :
210
The presence of 17α-ethinyl inhibits the action of liver metabolizing enzymes on the molecule, thus maintaining its strong oral effect. When injected, the effect of ethinylestradiol is equivalent to estradiol, but if both are taken orally, the effect is 15-20 times stronger than estradiol. Ethinylestradiol is one of the most potent oral estrogens.
Ethinylestradiol has antiovulatory effects at relatively low doses, and is most widely used in combination with progestins for oral contraception.
Assign:
Has similar indications to estradiol and estradiol ester derivatives. Some common indications are:
- Postmenopausal replacement therapy: 0.01-0.02 mg/24h (with progestin). However, for this indication, natural estrogen is preferred.
- Female hypogonadism, prostate cancer, breast cancer (in postmenopausal women), 0.1-1mg/time x 3 times/24 hours.
- In combined oral contraceptives: ethinylestradiol – levonorgestrel.
Dosage form:
Tablets 0.02mg; 0.05mg; 0.5mg (used to treat cancer); round tablets containing ethinylestradiol.
Etherification of (3)-OH to (3)-OCH 3 gives mestranol, and (3) gives quinestrol (trade name Estrovis), both of which are prodrugs of ethinylestradiol, administered orally with prolonged action.
Synthetic non-steroidal substances have estrogenic and anti-estrogenic effects.
Due to the size, configuration and position of the functional groups of these substances relative to estradiol, they have estrogenic or antiestrogenic effects. The structural difference in the antiestrogenic substances is the addition of side chains. Chlorothrianixen, diethylstilbestrol, are classic substances, have strong effects but are highly toxic. Antiestrogenic substances have the effect of stimulating ovulation and are used to treat infertility, while raloxifene also has the effect of maintaining the balance of bone metabolism and lipid metabolism (reducing LDL).
CHLOMIPHEN CITRATE
Brand name : Serophen, clomid, milophen.
Recipe :
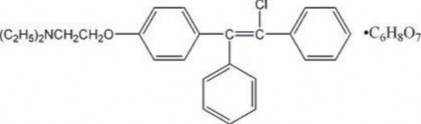
211
An anti-estrogen most widely used for the treatment of infertility; taken in 5-day courses. First course: Starting on the 5th day of the menstrual cycle: 50mg/24 hours (5 consecutive days).
Clomiphene is also used to treat postmenopausal breast cancer.
In the above formula: replace –Cl with –C 2 H 5 , replace –N(C 2 H 5 ) 2 with –N(CH 3 ) 2 and change the position of the two aromatic rings attached to C, so that –C 2 H 5 and –C 6 H 4 -O-CH 2 -CH 2- N(CH 3 ) 2 have the front configuration, we have tamoxifen citrate (trade name Novaldex). This substance has the same effect and use as clomiphene citrate, mainly for treating infertility.
1.2. Progestins
The natural hormone is progesterone, secreted by the corpus luteum and is abundant during pregnancy due to the placenta. Semi-synthetic substances are more stable, have a strong, long-lasting effect and can be taken orally. All of the above substances are collectively called progestins (progesterone), in structure they have a pregnan framework.
1.2.1. Progesterone PROGESTERON
Trade name : Lutein
Recipe :
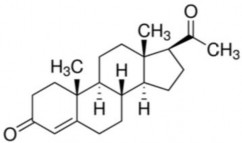
C 21 H 30 O 2 equation: 314.47
Scientific name : Pregn-4-en-3,20-dion or diceto-3,20-pregnen-4
For the first time, in 1934, A. Butenandt extracted gold from pig's body.
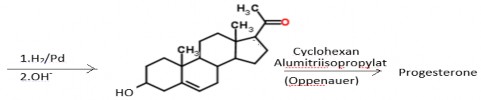
Preparation :
In industry, semi-synthetic from diosgenin (60% yield) or stigmasterol:
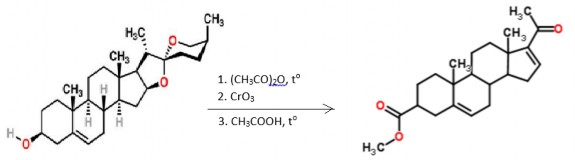
Diosgenin DPA
212
(dehydroprednenolone)
Prednenolone
Nature:
20
The preparation is in the form of a white crystalline powder, insoluble in water, soluble in oil, alcohol, ether, chloroform. Melts at 126 – 131 o C, the polymorph melts at 121 o C. Hydrophilic, [α] D = +
175 ÷+ 183 o (C 2 , dioxin).
Qualitative :
- Zimmermann reaction (of ketone function): gives purple-red color.
- Hydrazone formation reaction:
+ Preparations
+ 2,4 dinitrophenyl hydrazine 1/ EtOH, in the presence of HCl
+ Heat
- Reaction to form dioxime progesterone, precipitate recrystallized in methanol, measured melting point 134-137 o C:
+ Preparations
+ HO – NH 2 .HCl/EtOH and OH -
+ Heat
- Measure UV spectrum from methanol solution, measured in parallel with standard.
- Measure IR spectrum, compared with standard spectrum
Use TLC (for identification and testing of related impurities)
Quantitative :
- Precipitation weighing or photometric measurement
- HPLC (USP 2000) is the common method.
Uses :
- Is the main hormone of the body. Used as a replacement treatment for the following cases: corpus luteum deficiency, menstrual disorders, amenorrhea, painful menstruation, uterine bleeding, underdeveloped and inactive reproductive organs, infertility; with an average dose of 5-10mg/24h, combined with estrogen.
- Especially to prevent miscarriage: intramuscular injection 25 g/24 hours, continuously or 25-100 mg (can be increased if necessary)/time x 2 times/week.
- Endometrial cancer: use high doses.
Dosage form :
213