2.4.2.2. Effect of cultured embryo mass on embryo biomass growth
Culture material: using embryos through shaking liquid culture after 60 days of culture from 60-day-old embryos of experiments on direct cloning of embryos from leaf tissue.
To investigate the effect of cultured embryo mass on embryo biomass growth, the following initial embryo mass was used: 0.3; 0.6; 1.2; 1.8 g corresponding to 0.5%, 1%, 2% and 3% (w/v) in a V250 mL triangular flask containing 60 mL of experimental culture medium for the effect of DHST, 30 g/L of sugar. Collect data on fresh embryo mass (g), embryo multiplication coefficient (times) and morphological characteristics of embryos/embryo clusters at 30 NSCs.
The experiment was arranged in a single-factor design, with 3 flasks/replication, and each treatment was repeated 3 times. Observe, monitor, and collect data on the number of embryos.
2.4.2.3. Effect of cultured embryo size on secondary embryogenesis
Using materials such as asexual embryos with different sizes: small (7 - 8 mm), medium (~ 10 mm), large (~ 15 mm). Proceed to implant 30 small/medium/large embryos into a V250 mL triangular flask containing 60 mL of the same culture medium as the experiment affecting the mass of cultured embryos, with 30 g/L of sugar.
The experiment was arranged in a single-factor design, with 3 flasks/replication, and each treatment was repeated 3 times. Observe, monitor, and collect data on the number of secondary embryos formed, morphological characteristics, and embryo/embryo cluster color formed at 30 NSCs.
2.4.2.4. Effect of sugar concentration on embryo biomass growth
Inoculate 0.3 g embryo mass (0.5% - w/v) into a V250 mL triangular flask containing 60 mL of medium as in the above experiment, sucrose was investigated at different concentrations (20, 30, 40, 50 g/L); each experiment was repeated 3 times. Collect data on fresh embryo mass (g), embryo multiplication coefficient (times) and morphological characteristics, embryo/embryo cluster color at 45 NSC. The experiment was arranged completely randomized in a single-factorial manner.
2.4.2.5. Effect of light intensity on embryo biomass growth
Inoculate 0.30 g embryo mass (0.5% - w/v) into V250 mL triangular flask containing 60 mL of the above medium, 30 g/L sucrose, and culture under different lighting conditions: dark (0 lux), lighting with intensity of 2,000 lux (~ 27 μmol m -2 s -1 ), 4,000 lux (~ 54 μmol m -2 s -1 ), each experiment was repeated 3 times. Observe, monitor, and collect data on
Fresh embryo weight (g), embryo multiplication coefficient (times), morphological characteristics, embryo/embryo cluster color at 30 NSC. The experiment was arranged in a completely randomized single-factorial design.
2.4.2.6. Effect of coconut water ratio on secondary embryogenesis
Single embryos of average size (~ 10 mm) were cultured in V250 mL triangular flasks containing 60 mL of experimental medium to influence the mass of cultured embryos, 30 g/L sucrose, coconut water was surveyed at different ratios (0%, 5%, 10%). Cultured 30 single embryos/flask, each treatment was repeated 3 times. Collected data on the number of embryos formed, morphological characteristics, embryo/embryo cluster color at 21 NSCs.
The experiment was arranged in a completely randomized design with a single factor.
2.4.2.7. Creating seedlings from shaken liquid culture embryos
Using medium-sized single embryos (~ 10 mm), transplant into a V250 mL triangular flask containing 60 mL of MS or ½MS medium (1/2 macronutrient) [105] without adding growth regulators, 20 g/L sucrose, transplant 30 single embryos/flask, each experiment was repeated 3 times; observe and record the embryo development to the mature stage at 14 NSC. Then, transfer the embryos to a suitable solid medium (inheriting the experimental results) for the embryos to develop into seedlings in 60 days.
2.4.3. Observation of primary and secondary embryonic anatomical structures
Cut and create LMTBs along the embryo/root body, across the cotyledon (of the primary embryo) carrying the secondary embryo with a razor blade, the cut sample is stained using the double staining method of the Department of Botany - Faculty of Pharmacy, University of Medicine and Pharmacy, Ho Chi Minh City [43]. Observe the specimen with a Leica stereo microscope (20X objective) and take pictures.
2.4.4. Monitoring indicators and determination methods
Number of embryos/clusters = Total number of embryos/number of clusters
Embryo multiplication factor = Number of embryos/embryo cluster
Number of employees/staff
Fresh weight of collected embryos (g): Collect and weigh the embryo weight in each treatment after 30 days of rearing.
Embryo biomass multiplication factor (times) = 𝐾ℎ𝑙𝑛𝑔 𝑡ươ𝑖 𝑝ℎ𝑖 𝑡ℎ𝑢 𝑛ℎ𝑛(𝑔)
The number of people who pass the exam (𝑔)
2.5. Content 3. Creating adventitious roots
2.5.1. Direct formation of adventitious roots from leaf tissue
2.5.1.1. Effect of auxin (NAA/IBA) and mineral medium on adventitious root formation directly from leaf tissue
47
The individual effects of auxin and mineral medium on adventitious root induction of leaf tissue were investigated to determine the appropriate type and concentration of auxin on a suitable mineral medium.
Using materials such as leaf pieces I (10 x 10 mm), leaf pieces II (3 x 10 mm), inoculated upside down on the contacting leaf into MS, ½MS, B5 and SH media supplemented with individual growth regulators NAA/IBA at the same concentrations (0; 0.5; 1; 2; 3; 4; 5) mg/L, 30 g/L sucrose, 4,000 lux lighting, 12 hours of lighting/day. Inoculate 5 leaf pieces I/plate, 6 plates/replication; 10 leaf pieces II/plate, 3 plates/replication; each treatment is repeated 3 times.
The experiment was completely randomized with two factors: the factor of NAA/IBA concentration levels and the factor of mineral environment with 4 different environments (MS, ½MS, B5, SH). The experiment was monitored, data were collected on the rate of rooting samples (%), number of roots/sample, and root length (mm) at 30 days post-experimentation.
2.5.1.2. Effect of sugar concentration on adventitious root formation directly from leaf tissue
To investigate the effect of sugar concentration on root formation from leaf fragment explants, mineral medium with the most active root-forming substance (NAA/IBA) (inherited from the above experiment) was used as the culture medium, with sugar at different concentrations (20; 30; 40; 50 g/L).
Culture materials: leaf fragment I (10 x 10 mm), leaf fragment II (3 x 10 mm). The samples were inoculated into the above culture medium with 5 leaf fragments I/plate, 6 plates/replication; 10 leaf fragments II/plate, 3 plates/replication; each treatment was repeated 3 times. The experiment was arranged completely randomly, in a single-factorial manner. During the experiment, data on the percentage of root-forming samples (%), number of roots/sample, and root length (mm) were collected at 30 days after planting.
2.5.1.3. Effect of lighting conditions on adventitious root formation directly from tissue
leaf
To investigate the effect of lighting conditions on adventitious root formation from
Leaf fragments were cultured on mineral medium with appropriate auxin type and concentration. Sucrose concentration had the most effective effect on adventitious root formation from leaf fragments (inheriting the results from the two experiments above). The culture plates were placed under different lighting conditions of 4,000 lux, 2,000 lux, and complete darkness.
The culture materials used were leaf fragments I (10 x 10 mm) and leaf fragments II (3 x 10 mm). With leaf fragment I, plant 5 samples/plate, 6 plates/replication; with leaf fragment II, plant 10 samples/plate, 3 plates/replication; each treatment was repeated 3 times. The experiment was arranged in a completely randomized design.
48
naturally, in a single factorial manner. Observe and monitor the experiment, record data on the percentage of rooting samples (%), number of roots/sample (rooting), root length (mm) collected at 30 days after planting.
2.5.1.4. Survey of the anatomical morphology of roots regenerated directly from leaf tissue
10-day-old root primordia, ~4 mm long (formed directly on the upper surface of leaf fragments cultured on ½MS medium with 3 mg/L NAA) were cut longitudinally using a razor blade, microdissected and stained using the double-staining method [43]. The slides were observed using a Leica stereomicroscope (20X magnification) and photographed.
2.5.2. Creating adventitious roots from shoots
2.5.2.1. Creating adventitious roots from shoots originating from nursery stem nodes
Sterilize the sample.
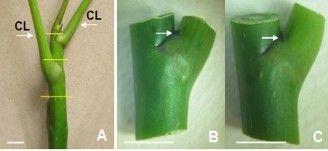
Figure 2.3. Stem burning material used for shoot culture.
A. Tree branches (horizontal lines are the positions of cutting to create stem nodes; CL: leaf petioles); B, C. Stem nodes used for bud culture (arrows indicate the position of dormant buds) (10 mm horizontal bar).
Use young branches (8 - 10 cm) of NGBCC plants (Figure 2.3 A). First, remove the leaves, wash the branches under running water for 10 minutes, then wash again with soapy water. Cut the branches into segments with dormant buds - branch nodes (hereinafter referred to as stem nodes) ~ 1.5 cm long, do not use the apical buds, then sterilize with 70% alcohol for 1 minute, with 30% Javel water (v/v) for 30 minutes, wash again 3 times with sterile distilled water. The stem nodes, after sterilization, cut off the dead tissue at both ends (Figure 2.3 B, C) are used as culture material in the shoot induction experiment.
Creating shoots from nursery stem nodes
Sterilized stem nodes were inoculated into MS medium supplemented with 0.5 mg/L BA and 2 mg/L kinetin, 30 g/L sucrose [3]. Shoot formation was recorded at 60 NSCs.
Creating adventitious roots from shoots
Shoots (~1 cm high) were separated and cultured in ½MS adventitious rooting medium supplemented with 0.2 mg/L NAA [3], 20 g/L sucrose, 10 g/L agar, pH 5.8; IBA was also used.
with a concentration of 0.2 mg/L. Transplant 2 axillary buds/pot, transplant 15 pots/repetition, each treatment is repeated 3 times. Monitor and record data on the following indicators: rooting shoot ratio (%), number of roots/shoot, root length (mm) at 60 NSC.
The experiment was arranged in a completely randomized, single-factorial design.
2.5.2.2. In vitro adventitious root formation from stem shoots
In vitro shoot induction from stem nodes
In vitro plants ~ 5 months old (originating from asexual embryos grown on ½MS medium without added DHST, containing 30 g/L sucrose) had all leaves removed, then the stems were cut into ~ 8 mm long sections carrying axillary buds and transplanted into the shoot-forming medium as above. Shoot formation was monitored at 60 NSC.
Creating adventitious roots from shoots
Separate 60-day-old axillary buds (0.7 - 1 cm high), transplant into adventitious rooting medium as in the above case. Transplant 2 axillary buds/pot, transplant 15 pots/repetition, each treatment is repeated 3 times. Monitor and record data on the following indicators: rooting shoot ratio (%), number of roots/shoot, root length (mm) at 60 NSC.
2.5.2.3. Creation of adventitious roots from shoots of asexual embryonic origin
Using plantlets derived from asexual embryos with a stem height of ~ 15 mm, cutting off the taproot, keeping the stem (cotyledon) and the upper leaves, used as adventitious root material. The samples were cultured in ½MS medium supplemented with NAA or IBA at the same concentration of 0.1 mg/L, 20 g/L sucrose. Culture 2 shoots/pot, culture 15 pots/replication, each treatment was repeated 3 times. Monitoring indicators: Ratio of root-forming samples (%); number of roots/sample; root length (mm) and morphological characteristics, root color at 30 NSC
2.5.3. Indicators and monitoring methods
Rooting rate (%) = (Number of rooting samples/number of explants) x 100% Number of roots/sample = Total number of roots/number of explants
Root length (mm) = Total length of roots/number of roots measured
The parameters of the in vitro rooting experiment from stem shoots were collected at 60 NSCs:
Rooting rate of shoots (%)= | (Number of rooted shoots/number of transplanted shoots) x 100% | |
Number of roots/shoots | = | Number of roots formed/number of shoots forming roots |
Root length (mm) | = | Total root length/number of roots measured |
Maybe you are interested!
-
 Comparison of Somatic Embryo, Protocorm and Cyst Formation Ability Between Culture Media in Dendrobium Orchid Species
Comparison of Somatic Embryo, Protocorm and Cyst Formation Ability Between Culture Media in Dendrobium Orchid Species -
 Effect of Initial Glyphosate Concentration on Processability of Electrochemical Fenton Process
Effect of Initial Glyphosate Concentration on Processability of Electrochemical Fenton Process -
 Managing cultural activities at the Center for Culture, Sports and Tourism of Phu Ninh district, Phu Tho province - 1
Managing cultural activities at the Center for Culture, Sports and Tourism of Phu Ninh district, Phu Tho province - 1 -
 Some Pictures of the Transformation of Traditional Culture of Thai People in Mai Chau
Some Pictures of the Transformation of Traditional Culture of Thai People in Mai Chau -
 Comparison of Geographical Conditions, Structure of Culture and Tourism Activities
Comparison of Geographical Conditions, Structure of Culture and Tourism Activities
2.6. Content 4. Adventitious root propagation in liquid medium
2.6.1. Effect of growth regulators on root branching
Using single roots (primary roots, ~ 1 cm long) directly regenerated from leaf fragments on ½MS solid medium with 3 mg/L NAA at 20 NSC time point as culture material.
Inoculate ~ 120 single roots into a V250 mL triangular flask containing 60 mL (~ 0.5% - w/v) of ½MS liquid medium supplemented with NAA or IBA at different concentrations of 0, 1, 2, 3 mg/L. After 20 days of culture, randomly take 30 roots/time, measure 3 times, collect data on primary root length (mm), number of primary secondary roots, length of primary secondary roots (mm), number of secondary roots (hereinafter referred to as lateral roots) formed and root morphology characteristics.
2.6.2. Effect of sugar concentration on root biomass growth
Inoculate 0.3 g root mass (0.5% - w/v) into a V250 mL triangular flask containing 60 mL of ½MS medium supplemented with NAA/IBA at concentrations inheriting the experimental results (2.6.1); sucrose at concentrations (20, 30 and 40 g/L). Collect data on fresh root mass (g), root multiplication coefficient (times) and morphological characteristics, root color, root cluster at 30 NSC, each treatment is repeated 3 times. The experiment is designed in a two-factor model: growth hormone factor (NAA, IBA) and sucrose concentration factor with three concentration levels (20, 30, 40 g/L).
2.6.3. Effect of cultured root mass on root biomass growth
To investigate the effect of cultured root mass on root biomass increase, root mass of 0.35 g; 0.70 g; 1.4 g; 2.1 g corresponding to 0.5%, 1%, 2% and 3% (w/v) was used in a V250 mL triangular flask containing 70 mL of ½MS medium supplemented with NAA or IBA at experimental concentrations (2.6.1), sucrose concentration was used from the experiment (2.6.2). Data were collected on fresh root mass (g), root multiplication coefficient (times) and morphological characteristics, root color, and root cluster at 30 NSCs, each treatment was repeated 3 times. The experiment was designed in a two-factor model: growth factor (NAA, IBA) and cultured root mass factor with four concentration levels (0.35; 0.7; 1.4; 2.1%).
2.6.4. Survey of root biomass growth over time
Transplant the most suitable root mass (from the results of experiment 2.6.3) into a V250 mL triangular flask containing 70 mL of the same culture medium as in experiment (2.6.3). After 28 days of culture, replace 70 mL of the culture medium with the same composition as the initial medium. Collect data on fresh root mass (g), root multiplication coefficient (times) at 7, 14, 21, 28, 35, 42 and 49 NSC, each treatment repeated 3 times. Experiment
arranged in two-factor style: quality factor (NAA, IBA) and time factor with time levels (7, 14, 21, 28, 35, 42, 49 days).
2.6.5. Indicators and monitoring methods
Primary root length (mm) = Root length/number of roots measured
Number of primary lateral roots = Number of roots formed from primary roots/number of primary roots
Length of primary lateral roots (mm) = Length of lateral roots/number of lateral roots Number of secondary lateral roots = Number of lateral roots per 1st lateral root
1st Anniversary Celebration
Fresh root weight (g): fresh root weight obtained by 30 NSC Multiplier (times) = 𝐾ℎ𝑖𝑙𝑛𝑔 𝑟𝑡ℎ𝑢 𝑛ℎ𝑛 (𝑔)
The ceremony of the 2nd lunar month (3)
2. 7. In vitro culture conditions
The experiments were carried out at room temperature of 25 - 28 o C, average humidity of 50 - 60%, all experiments were placed outdoors under fluorescent light, 12 hours of light/day, light intensity ~ 4,000 lux (except for the experiment of direct embryogenesis in dark conditions placed in complete darkness; experiment of the effect of light intensity on embryo biomass growth; experiment of the effect of lighting conditions on adventitious root formation in light conditions of 2,000 lux, in dark conditions the sample was placed in complete darkness). Using solid medium (agar 10 g/L, pH 5.8; autoclaved at 1 atm/121 o C for 20 minutes); experiment in liquid medium shaking at a shaking speed of 80 rpm.
2.8. Statistical methods and data processing
The experiment was arranged completely randomly, each treatment was repeated 3 times. The average results of 3 repetitions were processed and analyzed by one-factor ANOVA, testing the difference between treatments by LSD or Duncan. The 2-factor experiments were processed by one-way multivariate analysis of variance (One-way MANOVA) of Duncan's test with p ≤ 0.05 using SPSS 25.0 software. (Data in percentage of variation from 0 - 100% were converted to arcsin √𝑥 when processed statistically, or from 70 - 100% were converted to (x + 0.5) 1/2 .
CHAPTER 3. RESULTS AND DISCUSSION
3.1. Creating asexual embryos
In this study, directly formed asexual embryos were defined as embryos that regenerated immediately after the first culture, different from embryos that regenerated indirectly through two culture stages: callus formation and embryogenesis from callus [108][109].
3.1.1. Direct asexual embryogenesis from leaf tissue
3.1.1.1. Effect of auxin (NAA/IBA) and mineral medium on direct asexual embryogenesis from leaf tissue

Figure 3.1. Developmental forms of asexual embryos formed directly from leaf tissue.
A,B . Single globular embryo, cluster of globular embryos; C. Heart-shaped embryo (1) and globular embryo (2); D. Torpedo-shaped embryo; E,F,G,H . Embryo with developed cotyledons and radicle; I. Complete seedling with cotyledons, true leaves (single, compound), typical taproot. Arrows indicate cotyledons (white), radicle (black). Horizontal bar 2 mm.
After 5 weeks of culture, asexual embryos appeared in many treatments. In SH medium supplemented with 5 mg/L NAA, embryos appeared earliest and most, in ½MS medium, embryos appeared slowly and less. Structural differentiation during embryogenesis from NGBCC leaf fragments began from the spherical embryo form, appearing about 32 - 35 NSCs, pale yellow in color (Figure 3.1A), the heart-shaped embryo stage was green, appearing about 10 days later, this is a sign of pre-two cotyledons, a typical form of embryogenesis in dicotyledons (Figure 3.1C). The torpedo embryo form (Figure 3.1D) continued to develop to the stage with two cotyledons about 15 - 20 days later (Figure 3.1E,F,G,H). Embryos developed directly





