bromoethanol into the reaction mass. Stir and heat the reaction mixture (55 °C) for 6 h. Monitor the reaction by spectrophotometry with the solvent system dichloromethane : methanol (9.0 : 1.0; v/v).
After the reaction is complete, cool the reaction mass, filter out the potassium salts, and evaporate under vacuum to remove acetone. Dissolve the remaining portion in the flask with 500 mL of dichloromethane and transfer to a 1000 mL volumetric flask. Wash the organic phase several times with distilled water to neutral pH to completely remove the potassium salts and some of the excess reagent. Add 150 mL of distilled water to the flask, alkalize with sodium hydroxide to aqueous phase pH ~ 12-13, extract the alkaline aqueous phase 3 times, and combine the aqueous phase. Wash the aqueous phase again with dichloromethane 3 times, each time about 75 mL. Acidify the aqueous phase with 1M HCl to pH 10, extract twice with dichloromethane (500 mL each time). Wash the dichloromethane phase with water to neutral pH. Dry the organic phase with sodium sulfate overnight, and filter to remove the drying agent. Distill and collect all the solvent until it is dry, crystallize the dry residue in methanol to obtain orange-yellow needle-shaped crystals. Filter, dry the product at 60 °C for 3 hours, weigh, and obtain 3.26 g PH6 , yield 29.1%.
Comment: When upgrading to 5 g/batch scale, the reaction efficiency decreased significantly. On SKLM, a large amount of di- and tri-alkylated impurities was observed. Therefore, we continued to investigate the concentration of the reagent (amount of acetone solvent) and the feeding method.
a, Survey of the influence of agent concentration
In this experiment, we investigated the effect of the concentration of the reagent in the reaction mixture (amount of acetone solvent used) on the reaction efficiency at a 5g scale with fixed factors: molar ratio 2-bromoethanol: curcumin = 2.0
: 1, amount of DMF solvent, using K 2 CO 3 as base and reacting at 55 °C for 6 hours, the results are presented in Table 3.20 below:
Table 3.20. Results of the survey on the effect of agent concentration on reaction efficiency at a scale of 5 g/batch
STT
Acetone volume (mL) | Agent concentration (%, v/v) | Product Weight (g) | H % | |
1 | 400 | 0.90 | 3.26 | 29.12 |
2 | 500 | 0.72 | 3.78 | 33.76 |
3 | 600 | 0.60 | 3.95 | 35.28 |
4 | 700 | 0.51 | 3.98 | 35.55 |
Maybe you are interested!
-
 Test Results of Optimal Mode Effect on Mechanical Properties
Test Results of Optimal Mode Effect on Mechanical Properties -
 Expert Survey Results on Research Scales
Expert Survey Results on Research Scales -
 Survey Results on Corporate Communication
Survey Results on Corporate Communication -
 Customer Satisfaction Survey Results on International Payment Activities at Anz Bank Vietnam
Customer Satisfaction Survey Results on International Payment Activities at Anz Bank Vietnam -
 C: Survey Results for the Criterion “Challenging Work”
C: Survey Results for the Criterion “Challenging Work”
Comment: At the scale of 5 g/batch, the concentration of the reagent affects the reaction efficiency.
When the amount of solvent used is increased, from 400 to 600 mL, the concentration of the reagent is more diluted, the mono-alkylation reaction to create PH6 occurs more selectively, the reaction efficiency increases. However, when the amount of solvent used is further increased to 700 mL, the efficiency does not change significantly. Therefore, we use the appropriate amount of solvent, 600 mL of acetone.
b, Survey the influence of the feeding method
In this experiment, we investigated the effect of the loading method on the reaction efficiency at a scale of 5g with the following fixed factors: molar ratio of 2- bromoethanol: curcumin = 2.0: 1, amount of acetone solvent and amount of DMF solvent, using K 2 CO 3 as base and reacting at 55 °C for 6 hours. A total of 600 mL of acetone was divided to dilute the reagent added to the reaction. The results are presented in Table 3.21 below:
Table 3.21. Results of the survey on the influence of the method of loading the agent on the reaction efficiency at the scale of 5 g/batch
STT
How to load agent | Product Weight (g) | H % | |
1 | Do not dilute the agent. | 3.98 | 35.55 |
2 | Dilute the agent with 100 mL of acetone. | 4.16 | 37.16 |
3 | Dilute the agent with 200 mL of acetone. | 4.48 | 40.01 |
4 | Dilute the agent with 300 mL of acetone. | 4.2 | 37.51 |
Comment: Thus, the way of loading the reactant affects the reaction efficiency at the scale of 5g/batch. When the agent is pre-diluted with 200 mL of solvent, the reaction achieves the best efficiency, equivalent to the efficiency obtained at the laboratory scale.
From the above surveys, the PH6 synthesis scale was upgraded to 5.0 g/batch with the following synthesis process ( Improved process ):
Stir a mixture of 10.00 g (27.15 mmol; 1 eq) curcumin; 18.80 g (135.64 mmol; 5.0 eq) anhydrous K 2 CO 3 ; 400 mL anhydrous acetone; 10.0 mL DMF in a 2000 mL flask for 30 min to activate. Slowly add a solution of 3.60 mL (54.40 mmol; 2.0 eq) 2-bromoethanol reagent dissolved in 200 mL anhydrous acetone to the reaction mass. Stir and heat the reaction mixture (55 °C) for 6 h. Monitor the reaction by spectrophotometry with the solvent system dichloromethane : methanol (9.0 : 1.0).
After the reaction is complete, cool the reaction mass, filter to remove potassium salts, and distill under vacuum with acetone. Dissolve the remainder in the flask with 500 mL
dichloromethane and transfer to a 1000 mL volumetric flask. Wash the organic phase several times with distilled water to neutral pH to completely remove potassium salts and some residual reagent. Add 150 mL of distilled water to the flask, alkalize with sodium hydroxide to aqueous phase pH ~ 12-13, extract the alkaline aqueous phase 3 times, combine the aqueous phase. Wash the aqueous phase again with dichloromethane 3 times, each time about 75 mL. Acidify the aqueous phase with 1M hydrochloric acid to pH 10, extract twice with dichloromethane (500 mL each time). Wash the dichloromethane phase with water to neutral pH. Dry the organic phase with sodium sulfate overnight, filter to remove the drying agent. Distill to recover all the solvent to the residue, crystallize the residue in methanol to obtain orange-yellow needle-shaped crystals. Filter, dry the product at 60 °C for 3 hours.
Conducting 3 batches to investigate the repeatability of the synthesis process, the results were obtained as shown in Table
3.22 after:
Table 3.22. Results of repeatability survey of PH6 synthesis process at 5 g/batch scale
STT
Sense | KL SP PH6 (g) | H % | t° water (°C) | |
1 | Orange-yellow needle-shaped crystals | 4.54 | 40.5 | 159.0–161.5 |
2 | Orange-yellow needle-shaped crystals | 4.57 | 40.8 | 159.8–161.7 |
3 | Orange-yellow needle-shaped crystals | 4.52 | 40.4 | 158.8–160.5 |
Medium | 4.54 | 40.6 | 159.2–161.2 | |
RSD% | 0.55 | 0.55 | ||
Comment: The improved PH6 synthesis processshowed stable performance when scaled up to 5 g/batch.
3.2.2.3. Scale up PH6 synthesis to 25 g/batch
Using the improvements at the 5 g/batch scale, carry out the PH1 synthesis reaction at the 25 g/batch scale, using a 5000 mL Chemglass reactor with a heat jacket. The procedure is as follows:
Stir a mixture of 50.00 g (135.75 mmol; 1 eq) curcumin; 94.0 g (678.2 mmol; 5.0 eq) anhydrous K 2 CO 3 ; 2000 mL anhydrous acetone; 50 mL DMF in a 5000 mL reaction vessel for 30 min to activate. Slowly add a solution of 18.0 mL (272 mmol; 2.0 eq) 2-bromoethanol reagent dissolved in 1000 mL anhydrous acetone to the reaction mass. Stir and heat the reaction mixture (55 °C) for 6 h. Monitor the reaction by spectrophotometry with the solvent system dichloromethane : methanol (9.0 : 1.0).
After the reaction is complete, cool the reaction mass, filter out the potassium salts, and vacuum distill on a Buchi R-220 to recover the acetone. Dissolve the remainder in
flask with 2.5 L of dichloromethane and then divided into two batches, each batch of 1.25 L of solution, transferred to a 2000 mL volumetric flask.
For each portion, wash the organic phase several times with distilled water to neutral pH to completely remove potassium salts and some residual reagent. Add 375 mL of distilled water to the extraction flask, alkalize with sodium hydroxide to pH 12-13 of the aqueous phase, extract the alkaline aqueous phase 3 times, and combine the aqueous phase. Wash the aqueous phase again with dichloromethane 3 times, each time about 180 mL. Acidify the aqueous phase with 1M hydrochloric acid to pH 10 and add 1.25 L of dichloromethane, shake and collect the organic phase. Wash the dichloromethane phase with water to neutral pH. Combine the 2 batches, dry the organic phase with sodium sulfate overnight, filter to remove the drying agent. Distill to residue, collect all solvent. Crystallize the residue in methanol to obtain orange-yellow needle-shaped crystals. Filter and dry the product at 60 °C for 3 hours, obtaining 24.05 g PH6 , reaction efficiency 42.9%, melting point: 158.8-160.5 °C.
Comment: Stable response performance when scaling up.
The results of reaction surveys, process development and scale-up of PH6 derivative synthesis are summarized in Table 3.23 below:
Table 3.23. Summary table of reaction survey results, process development and scale-up of PH6 synthesis
Product size
1 g/batch | 5 g/batch | 25 g/batch | |
KL of curcumin raw material (g) | 2.00 | 10.00 | 50.00 |
Agent volume (mL) | 0.72 | 3.6 | 18.0 |
Molar ratio of agent: curcumin | 2 : 1 | 2 : 1 | 2 : 1 |
Reaction temperature (°C) | 55 | 55 | 55 |
Response time (hours) | 6 | 6 | 6 |
Amount of reacting solvent (mL) | 80 | 600 | 3000 |
Concentration of the participating substance (ratio) agent: solvent, etc.) | 0.9 % | 0.6 % | 0.6% |
How to load | Add the reagent directly to the reaction mass. | Dilute the agent before adding to the mass. react | Dilute the agent before adding to the mass. react |
Refining method | Column chromatography or extract | Extract and replace pH change | Extract and replace pH change |
Product weight (g) | 0.92 | 4.54 | 24.05 |
Efficiency (%) | 41.1 | 40.6 | 42.9 |
The flow chart of PH6 synthesis is as follows ( Figure 3.1 ):
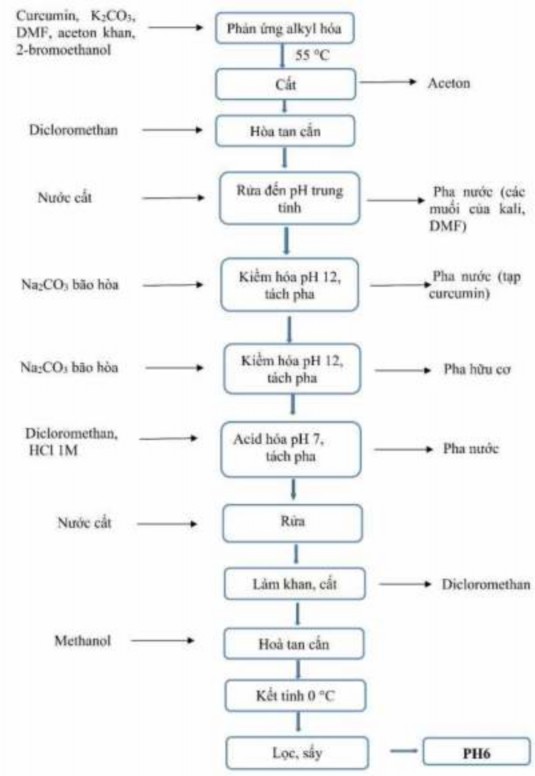
Figure 3.1. Flow chart of PH6 synthesis process
Some images of PH6 synthesis at 25 g/batch scale ( Figure 3.2 ):
Curcumin loading Reaction process


Figure 3.2. Some images of PH6 synthesis at 25 gram/batch scale
3.2.3. Development of a synthesis process for mono- O -(2-(succinyloxy)ethyl)- curcumin derivative (PH9)
3.2.3.1. Survey of some factors affecting the efficiency of PH9 synthesis reaction at laboratory scale
Performing the reaction according to Section 3.1.4.2 with intermediate PH6 , we investigated some factors affecting the reaction efficiency to be able to upgrade the synthesis process of derivative PH9 . The investigated factors: molar ratio of succinic anhydride: PH6 , reaction temperature, reaction time.
a, Investigation of the molar ratio of succinic anhydride : PH6
To investigate the effect of the molar ratio of succinic anhydride : PH6 on the reaction efficiency: Carry out the reaction according to Section 3.1.4.2 , keeping the reaction parameters unchanged: anhydrous pyridine solvent, temperature 100 °C, time 2.5 hours, the results are presented in Table 3.24 below:
Table 3.24. Results of the survey on the effect of mole ratio on the reaction efficiency to create PH6
STT
Succinic anhydride mole ratio : PH6 | Traces of components on SKLM | Weight of PH9 product (g) | H % | |
1 | 1.0 : 1 | Intensity of PH6 stain is dark, stain PH9 is slightly strong | 0.09 | 36.21 |
2 | 1.5 : 1 | PH6 stain intensity is less intense, PH9 stain intensity is darker | 0.13 | 52.31 |
3 | 2.0 : 1 | PH6 stain intensity, stain PH9 is darker | 0.17 | 68.41 |
4 | 2.5 : 1 | PH6 stain intensity is very faint, PH9 stain is darker, appears impurities | 0.15 | 60.36 |
Comment: When the molar ratio of succinic anhydride : PH6 increases, the amount of PH9 productproduced increases, and the reaction efficiency increases. The PH9 trace is the darkest and the reaction efficiency is the highest at the molar ratio of succinic anhydride : PH6 = 2.0 : 1, but when the amount of succinic anhydride continues to increase, impurities appear, reducing the reaction efficiency. Therefore, the best molar ratio for the reaction is: succinic anhydride : PH6 = 2.0 : 1.
b, Survey the influence of temperature
To investigate the effect of temperature on reaction efficiency: Conduct the reaction according to Section 3.1.4.2, keeping the reaction parameters unchanged: anhydrous pyridine solvent, molar ratio of succinic anhydride : PH6 = 2.0 : 1, time 2.5 hours, the results are presented in Table 3.25 below:
Table 3.25. Results of the reaction temperature survey to create PH9
STT
Temperature (°C) | Traces of components on SKLM | Product Weight (g) | H % | |
1 | 70 | PH6 stain is dark, PH9 stain is very faint, not clear | 0.04 | 16.09 |
2 | 80 | PH6 stain is dark, PH9 stain is light | 0.08 | 32.19 |
3 | 90 | PH6 stain is still dark, PH9 stain pretty clear | 0.11 | 44.26 |
3 | 100 | PH6 stain is faint, PH9 stain is faint bold, clear | 0.17 | 68.41 |
4 | 110 | PH6 stain is faint, PH9 stain is dark, impurities appear below product trace | 0.14 | 56.33 |
Comment: The survey results show that at 100 °C, the PH9 product is produced in the largest quantity, with the highest yield. When the temperature is increased, the reaction creates additional acylated impurities.
c, Investigate the effect of reaction time
Using the molar ratio of succinic anhydride : PH6 = 2.0 : 1, pyridine catalyst and reaction at 100 °C, we investigated the effect of time on the reaction, the results are presented in Table 3.26 :
Table 3.26. Results of reaction time survey to create PH9
STT
Response time (hours) | Traces of components on SKLM | KL SP (g) | H % | |
1 | 1.0 | PH6 stain is dark, PH9 stain is light | 0.07 | 28.17 |
2 | 1.5 | PH6 mark is still dark, PH9 mark is quite clear | 0.12 | 48.28 |
3 | 2.0 | PH6 stain is faint, PH9 stain is bold and clear | 0.18 | 72.43 |
4 | 2.5 | PH6 stain is faint, PH9 stain is dark, impurities appear below the product stain | 0.17 | 68.41 |
Comment: When the reaction time is less than 1.0 hour, the amount of product produced is very small. Increasing the reaction time, the amount of product increases and at 2.0 hour, the amount of product produced is the highest. Extending the reaction time to 2.5 hours, impurities appear on the SKLM and the reaction efficiency decreases. Thus, the most reasonable time for the reaction is 2 hours.
Through surveying the factors affecting reaction efficiency, the table of best parameters for the reaction process is obtained as follows:
Table 3.27. Summary table of the best parameters for the PH9 synthesis process
Succinic anhydride : PH6
Solvent | Temperature react | Response time (hours) | KL (g) | H % | |
2.0 : 1 | pyridine | 100 °C | 2 | 0.18 | 72.43 |
3.2.3.2. Scale up PH9 synthesis to 2 g/batch
Based on the above surveys, the synthesis process of PH9 at a scale of 2 g/batch is built as follows:
Add a mixture of 2.00 g (4.85 mmol; 1 eq) PH6 ; 1.00 g (9.70 mmol; 2.0 eq) succinic anhydride; 2.0 mL anhydrous pyridine into a 100 mL round-bottom flask. Stir and reflux the mixture.