The MTHFR enzyme maintains the stability of the folate metabolism cycle and prevents the risk of increased homocysteine levels in the blood. The reduction in MTHFR enzyme activity due to the MTHFR C677T gene affects folate metabolism, causing increased homocysteine levels in the blood and leading to many pathological conditions including osteoporosis.

Figure 1.4. Folate metabolism cycle
Source: Goyette P et al (1995) 61
Maybe you are interested!
-
 Model of Some Related Studies.
Model of Some Related Studies. -
 Foreign Studies on Improving Competitiveness for Tourism Development
Foreign Studies on Improving Competitiveness for Tourism Development -
 Comparison of Distribution by Number of Cesarean Sections Between Studies
Comparison of Distribution by Number of Cesarean Sections Between Studies -
 Summary Table of Characteristics of Similar Studies Around the World
Summary Table of Characteristics of Similar Studies Around the World -
 Correlation Chart Between Inflation Fluctuation and Loan-to-Assets Dispersion
Correlation Chart Between Inflation Fluctuation and Loan-to-Assets Dispersion
1.2.1.3. MTHFR gene polymorphism
- Currently, about 50 polymorphisms on the MTHFR gene have been found, of which two common MTHFR polymorphisms are MTHFR rs1801133 (C677T) and MTHFR A1298C, both of which reduce the activity of the MTHFR enzyme and lead to increased blood homocysteine levels.
- Many studies have shown that the MTHFR rs1801133 gene polymorphism has a greater influence on MTHFR enzyme activity than other polymorphisms of the MTHFR gene 62 .
- In this study we considered the MTHFR rs1801133 genotype polymorphism (MTHFR C677T), the nucleotide C is changed to T at position 677, which changes valine at position 222 to alanine 63 .
1.2.1.4. SNP rs1801133 - C677T - Ala222Val
- MTHFR rs1801133 gene polymorphism ( MTHFR C677T) will reduce the activity of the MTHFR enzyme. People carrying the MTHFR C677T gene in the homozygous recessive form ( MTHFR 677TT) have 70% less enzyme activity than normal ( MTHFR 677CC) and in the heterozygous form ( MTHFR 677CT) have 30 - 40% less enzyme activity than normal ( MTHFR 677CC) 10.64 . Many studies have shown that people carrying the homozygous TT genotype of the MTHFR rs1801133 gene polymorphism have risk factors for many diseases such as osteoporosis, cardiovascular disease, stroke, hypertension, preeclampsia, glaucoma, mental disorders, and some types of cancer.
1.2.1.5. Mechanism of osteoporosis caused by MTHFR gene
The MTHFR C677T gene alters or reduces the activity of the enzyme methylene tetrahydrofolate reductase, leading to increased blood homocysteine levels. The mechanism by which increased blood homocysteine levels may affect bone density and osteoporosis is likely multifactorial and interacting 65 . Increased homocysteine increases osteoclast activity, decreases osteoblast activity, and directly affects bone matrix 66 (Figure 1.5).
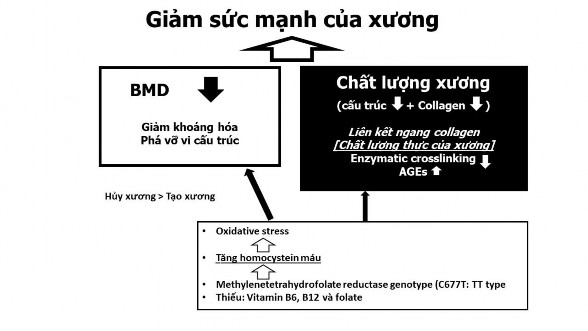
Figure 1.5. Effect of MTHFR gene on bone
Source: Saito M, Marumo K (2018) 67
- In vitro studies have shown that high homocysteine levels may modulate bone remodeling by promoting osteoclast activity, inducing apoptosis in stromal cells, osteocytes, and osteoblasts, and inhibiting trophoblast differentiation. Homocysteine induces apoptosis through the action of reactive oxygen species and nuclear factor kappa B (NF-kappa B). Intracellular reactive oxygen species stimulate osteoclast activity, playing an important role in increased bone resorption. Loss of
This imbalance between bone formation and bone resorption may result in low BMD in hyperhomocysteinemic individuals 67 .
- Roman Thaler et al. (2011) demonstrated that homocysteine stimulates the synthesis of interleukin 6 (IL-6), which can regulate the growth and differentiation of osteoclasts leading to increased bone resorption, which explains the high bone turnover in some patients with elevated plasma homocysteine levels.
Furthermore, homocysteine inhibits the activity of the collagen cross-linking enzyme lysyl oxidase (Lox) (due to inhibition of mRNA corresponding to the enzyme), it reduces the expression of the Lox enzyme through IL-6, JAK2, Fli1 (Friend leukemia integration transcription factor 1) leading to a decrease in bone matrix quality 68 .
- The thiol groups in homocysteine undergo auto-oxidation, thereby causing oxidative stress and generating reactive oxygen species (ROS). The oxidative stress then induces the synthesis of matrix metalloproteinases. The matrix metalloproteinase family consists of three major subgroups, interstitial collagenases, gelatinases, and stromelysins, which are involved in extracellular matrix degradation and bone remodeling. Both collagenases and gelatinases affect bone collagen by osteoblasts69 .
- One of the proposed mechanisms by which homocysteine affects bone is that elevated blood homocysteine levels reduce bone blood flow. A study by Thomas Vacek et al. in 2012 found that blood flow to the tibia was reduced in mice with elevated blood homocysteine levels. Blood flow to bone is important because bone is a living tissue composed of cells that require nutrients to grow and maintain. Reduced blood flow to bone may be a cause of osteoporosis 66 .
1.2.1.6. Studies on MTHFR rs1801133 correlation with bone density
- Bo Abrahamsen et al. (2003) studied 1748 postmenopausal Danish women and showed that people with the TT genotype of the MTHFR rs1801133 gene polymorphism had reduced bone density at the CSTL, CXĐ, and ĐTXĐ compared to people with the CC and TT genotypes in the early postmenopausal stage. After 5 years of hormone replacement therapy, people with the TT genotype still had reduced bone density at the proximal femur 7 .
- Robert R. McLean et al. (2004) studied 1632 subjects including both men and women (Framingham Offspring Study conducted on residents of Framingham town, Massachusetts, USA). Participants were quantified for plasma folate concentration and determined the MTHFR rs1801133 polymorphism. The results determined that there was an association between the TT genotype polymorphism of MTHFR rs1801133 and bone density depending on serum folate concentration. People with the TT genotype had lower bone density than people without the TT genotype 70 .
- Morten M. Villadsen et al. (2004) studied 724 subjects including 388 osteoporosis patients and 336 controls and showed that the TT genotype of MTHFR rs1801133 increased the risk of fracture and was a predictor of reduced BMD at the lumbar spine in Danish women 71 .
- Xiumei Hong et al. (2007) studied 1899 Chinese postmenopausal women and found that people carrying the T allele of the MTHFR rs1801133 polymorphism had a higher tendency to osteoporosis than people who did not carry the T allele, but the difference was not statistically significant 8 .
- Zhu et al. (2008) conducted a cohort study on 1213 Australian women aged 70 to 85 years and found an association between high blood homocysteine levels due to the MTHFR rs1801133 gene polymorphism and reduced femoral bone density but did not increase the risk of fracture 72 .
- Masataka Shiraki and colleagues (2008) studied 502 Japanese postmenopausal women and found that people with the TT genotype had a higher rate of osteoporosis and fractures than people without the TT genotype 9 .
- Agueda et al. (2010) studied 944 Spanish postmenopausal women and found that the MTHFR rs1801133 gene polymorphism was not statistically significantly associated with bone density of the femoral neck and lumbar spine, however, the authors found that the heterozygous genotype 677TT
MTHFR increases the risk of vertebral fractures 73 .
- Wang et al. (2011) analyzed 20 studies with 3525 patients and 17909 control subjects and showed a mild correlation between MTHFR rs1801133 and bone mineral density CXĐ, CSTL, ĐTXĐ and total body in East Asians 74 .
- In 2012, Aniel Jessica Leticia Brambila – Jabia and colleagues studied 71 Mexican rheumatoid arthritis patients and found that people with homozygous genotype TT had lower BMD than people with heterozygous genotype CT and both groups had lower BMD than people with homozygous genotype CC 75 .
- In 2013, Chutaporn Tongboonchoo and colleagues studied 346 postmenopausal Thai women and found that those with the CT heterozygous genotype had a higher risk of decreased bone density than the CC genotype 76 .
- In 2016, Hong-Zhuo Li and colleagues meta-analyzed 21 studies on 33,045 subjects and showed that the MTHFR rs1801133 gene polymorphism was associated with femoral neck BMD in postmenopausal women, in Caucasians and in men. When meta-analyzing 22 studies on 32,271 subjects, the authors also showed an association between this gene polymorphism and lumbar spine BMD in postmenopausal women 77 .
- In 2020, Xiao-Chen et al. synthesized 7 case-control studies in Chinese, Mexican and Thai women, investigating the association between MTHFR rs1801133 gene polymorphism and osteoporosis risk. The results showed that people with the T allele had an increased risk of osteoporosis in the co-dominant model of the TT genotype compared to the CC genotype (OR = 2.36, 95%CI: 1.81 – 3.08, p<0.05), the dominant model of the TT and CT genotypes compared to the CC genotype (OR = 1.47, 95%CI: 1.21 – 1.77, p<0.05), the recessive model of the TT genotype
compared with CC and CT genotypes (OR = 2.16, 95%CI: 1.71 – 2.74, p<0.05) 78 .
- In 2020, Massimo De martinis and colleagues studied 252 Italian postmenopausal women and found a significant association between homocysteine, BMD and interleukin 6 levels in osteoporosis in postmenopausal women 65 .
- In 2021, Nakamo et al. observed significant associations of LRP5 rs3736228 and MTHFR rs1801133 with the prevalence of knee OA, hip OA, and osteoporosis in elderly Japanese women from a randomly selected study group 79 .
- In 2014, Guan et al. analyzed 7 case-control studies with 4258 patients and 3454 healthy people. The study results showed that there was no association between the MTHFR rs1801133 polymorphism and osteoporotic fractures in postmenopausal women 80 .
- In 2019, Soewarlan WDHP and colleagues studied postmenopausal Indonesian women and found no association between the MTHFR rs1801133 gene polymorphism and BMD 81 .
- In summary, we found that there are many studies showing that people with the TT genotype of the MTHFR rs1801133 gene polymorphism are at risk of reduced bone density such as the studies of Bo Abrahamsen on Danish postmenopausal women, Massimo De Martinis on Italian postmenopausal women, Xiumei Hong on Chinese postmenopausal women, Masataka Shiraki on Japanese postmenopausal women. However, the study of Soewarlan WDHP and colleagues on Indonesian postmenopausal women, a country in the same Southeast Asian region as Vietnam, has not found a relationship between this gene polymorphism and bone density. In Vietnam, there is currently no study on the MTHFR rs1801133 gene polymorphism with bone density and risk of fracture in postmenopausal women.
1.2.2. Overview of the LRP5 gene and SNP rs41494349
1.2.2.1. Location and structure of the LRP5 gene
- Cytogenetic region: 11q13.4 means the gene is located on the long arm (q) of chromosome 11 at position 13.4, starting from base pair 68,298,865 to base pair 68,449,274. By gene sequence analysis, Gong identified the LRP5 gene consisting of 23 exon coding regions and spanning 100kb with a large extracellular region, a single transmembrane region, and a cytoplasmic tail 82 . In bone tissues, LRP5 is found in osteoblasts and osteocytes, but not in osteoclasts 83 .

Figure 1.6. Location of LRP5 gene
- SNP Q89R (rs41494349) is located at exon 2, the first coding region of the LRP5 protein. At SNP Q89R, nucleotide A is replaced by nucleotide G.
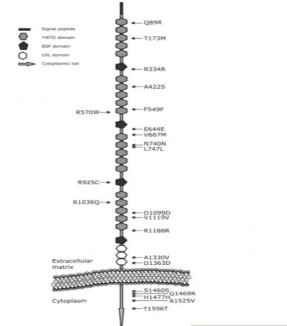
Figure 1.7: Schematic diagram of LRP5 protein and exon locations.
Source: Saarinen A (2011) 84





