7.00
0.00
0.00
Magnetite
Wet selection
100.00
(800 - 1000 bones)
(8000 - 10000 bones)
Rare Earth Enrichment Research Sample
Wet selection
Proportion
0.63
100
harvest, γ, %
Content
Rare earth β , %
Actual rare earth recovery ε, %
0.15
Products from
Non-magnetic products
69.88
14.95
7.58
0.07
Rare earth rich products
Mica
Flotation
3.80
0.75 15.54
84.3
Figure 3.2 . Experimental diagram and results of rare earth enrichment from research samples. Low-intensity magnetic separation separates strongly magnetic minerals such as magnetite.
High intensity magnetic separation separates octite and weakly magnetic minerals such as mica, ferropargasite... from non-magnetic minerals mainly feldspar, quartz and other amphibole minerals.
Rare earth minerals have magnetic properties similar to some iron silicate minerals (ferropargasite), mica (kinoshitalite). To separate these minerals from each other, the buoyancy of the minerals must be used. In this flotation stage, we use the collector AНП14 to separate mica from octite and other minerals.
Summary table of results of separation of rare earth rich products from research samples
Products
Weight quantity, g | Rate of return plan γ, % | Soil content rare β, % | Land acquisition rare, ε, % | |
Magnetite | 1,870 | 7.00 | 0.00 | 0.00 |
Product no word | 18,410 | 69.88 | 0.15 | 14.95 |
Product rich in rare earths | 4,100 | 15.54 | 3.80 | 84.30 |
Mica | 2,000 | 7.58 | 0.07 | 0.75 |
Ore head | 26,380 | 100.00 | 0.63 | 100.00 |
Maybe you are interested!
-
 Rare earth recovery from copper ore processing waste and application as fertilizer for tea plants and some vegetables in Da Lat, Lam Dong province - 1
Rare earth recovery from copper ore processing waste and application as fertilizer for tea plants and some vegetables in Da Lat, Lam Dong province - 1 -
 Evaluation of Experimental Results and Verification Experiments
Evaluation of Experimental Results and Verification Experiments -
 Study on Extraction of Cerium and Rare Earth (III) from Total Rare Earth Oxides
Study on Extraction of Cerium and Rare Earth (III) from Total Rare Earth Oxides -
 Research on building database and model for assessing radiation dose in some rare earth mines and placer mines - 1
Research on building database and model for assessing radiation dose in some rare earth mines and placer mines - 1 -
 Post-Experimental Event Organization Skills Measurement Results of Experimental and Control Groups
Post-Experimental Event Organization Skills Measurement Results of Experimental and Control Groups
The summary of results in Table 3.2 shows that the rare earth content Ln 2 O 3 has been increased from 0.63% to 3.80% (enrichment factor of about 6 times) achieving a fairly high yield of 84.30%. In addition to rare earth-rich products, some by-products that meet the standards of valuable commodities such as magnetite and mica ore have also been obtained.
3.1.3. Results of analysis of ore sample composition in the rare earth rich fraction
Ca 2 Ce 3 (SiO 4 )(Si 2 O 7 )(O,OH) is a silicate mineral containing mainly rare earth elements.
Cerium group.
The ore sample after rare earth enrichment was determined for its crystal phase structure by X-ray diffraction method. The results on the X-ray diffraction diagram in Figure 3.3 show that the non-magnetic mineral components such as kaolinite and albite were separated from the ore sample. In addition to the diffraction lines characteristic of the crystalline phase of ferropargasite and mica (kinoshitalite, the line intensity decreased significantly because the flotation stage separated most of these minerals), there are also diffraction lines characteristic of the appearance of the crystalline phase of cerium allanite (octite) with the general formula
Ferropargasite phase NaCa 2 Fe 4 AlSi 6 Al 12 O 22 (OH) 2 Kinoshitalite phase BaMg 3 Al 2 Si 2 O 10 (OH) 2
Allanite phase Ca 2 Ce 3 …(SiO 4 )(Si 2 O 7 )(O,OH) Quartz phase SiO 2
Figure 3.3 . X-ray diffraction pattern of the ore sample after rare earth enrichment. Chemical composition of the rare earth enriched ore sample after enrichment was determined.
by quantitative analysis. The results of the elemental content (calculated by total oxide) showed that the content of Al 2 O 3 and SiO 2 decreased significantly, which can be explained by the fact that these two components have a large content in the mineral part.
Non-magnetic elements are separated, similar to the content of Na 2 O, MgO, BaO. On the contrary, the content of Fe 2 O 3 increases because the ferropargasite phase contains iron, which is a magnetic mineral. The results of chemical analysis of the ore composition after rare earth enrichment are fully presented in Table 3.3.
Table . Chemical composition of the rare earth rich fraction after selection
TT
Target | Ingredient, % | TT | Target | Ingredient, % | |
1 | Al2O3 | 4.33 | 7 | TiO 2 | 0.41 |
2 | High | 5.11 | 8 | CuO | 0.28 |
3 | Fe 2 O 3 | 26.10 | 9 | MnO 2 | 0.15 |
4 | Na2O | 1.42 | 10 | U 3 O 8 | 0.008 |
5 | MgO | 1.40 | 11 | ThO 2 | 0.003 |
6 | SiO 2 | 41.20 | 12 | Ln2O3 | 3.80 |
The analysis results show that the total rare earth oxides account for 3.80% by mass.
The separation and flotation of Sin Quyen copper ore waste obtained a rare earth rich fraction with rare earth content increased from 0.63% to 3.8%. In this section, we use the wet method to recover total rare earth oxides from the rare earth rich fraction.
3.2. Research on rare earth recovery by acid method
3.2.1. Research on rare earth recovery by acid leaching method
In this section, HCl, HNO 3 , H 2 SO 4 acids are used to study the decomposition of ores to recover rare earths. The rare earth-rich ore fraction is extracted with agitation in an acid solution under normal conditions with the following factors: acid nature, acid concentration, ore/acid ratio and extraction time being investigated.
3.2 1 1 Hydrochloric acid extraction method
The experiment to study the ability to extract rare earth rich ore fractions with hydrochloric acid solution was conducted for 3 days with ore/HCl ratios of 1/1, 1/2, 1/3, 1/4, 1/5, 1/6 and acid concentrations of 4 M, 8 M, 12 M to evaluate the recovery efficiency of rare earth. The results are presented in Figure 3.4 and Appendix 1.
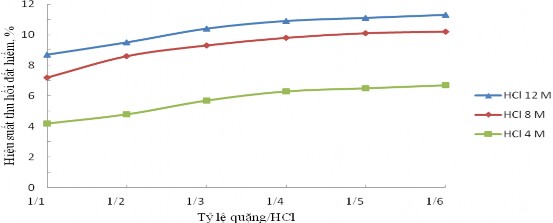
Figure 3.4. Effect of HCl acid concentration and ore/HCl ratio on rare earth recovery efficiency
From Figure 3.4, we can see that the recovery efficiency of rare earth increases with the decrease in the ore/HCl ratio and with the increase in the concentration of HCl solution. When the ore/HCl ratio decreases from 1/4 to 1/6, the recovery efficiency with HCl solution increases insignificantly. The recovery efficiency of rare earth reaches 11.0% with 12 M HCl solution, the ore/HCl ratio is 1/4.
3.2 1 Nitric acid extraction method
The experiment to study the recovery efficiency of rare earth from rare earth rich fraction using HNO 3 solution was conducted for 3 days with different ratios of ore/HNO 3 and acid concentrations. The results are presented in Figure 3.5 and Appendix 2.
HNO3 12
HNO3 8M
HNO3 4M
M
Effect of HNO 3 acid concentration and ore/HNO 3 ratio on rare earth recovery efficiency
The recovery efficiency of rare earth increases with the decreasing ratio of ore/HNO 3 and with the increasing concentration of HNO 3 acid solution , as shown clearly in Figure 3.5. When the concentration of HNO 3 increases , the reaction rate increases, the oxidation capacity of HNO 3 increases. When the ratio of ore/HNO 3 decreases from 1/4 to 1/6, the recovery efficiency of rare earth by HNO 3 solution is almost unchanged. The recovery efficiency reaches 14.9% with 12 M HNO 3 solution , the ratio of ore/HNO 3 is 1/4.
3.2 1 Sulfuric acid extraction method
The experiment to study the ability to extract rare earth-rich fractions using sulfuric acid solution was conducted for 3 days with different ratios of ore/H 2 SO 4 and acid concentrations to investigate the effect of acid concentration on rare earth recovery efficiency. The results are presented in Figure 3.6 and Appendix 3.
H 2 SO 4 18M H 2 SO 4 15M H 2 SO 4 12M H 2 SO 4 8M
H 2 SO 4 4M
Ore/H 2 SO 4 ratio
Effect of H 2 SO 4 acid concentration and ore/H 2 SO 4 ratio on rare earth recovery efficiency
The results in Figure 3.6 show that when increasing the acid concentration and decreasing the ratio of ore/H 2 SO 4, the recovery efficiency of rare earth increases. The ratio of ore/H 2 SO 4 1/4, the recovery efficiency reaches saturation at all concentrations. Under the given conditions, the recovery efficiency of rare earth reaches 24.8% when using H 2 SO 4 18 M, the ratio of ore/H 2 SO 4 1/4.
Leaching experiments of rare earth rich fraction were carried out with 12 M HCl, 12 M HNO 3 , 12 M, 15 M and 18 M H 2 SO 4 solutions. Mass ratio of ore/acid
1/4. The experimental results of the investigation of the effect of extraction time on the recovery efficiency of total rare earth oxides are shown in Figure 3.7 and Appendix 4.
H 2 SO 4 18 M
H 2 SO 4 15 M
H 2 SO 4 12 M
HNO3 12M
HCl 12M
Effect of extraction time on rare earth recovery efficiency The results in Figure 3.7 show that the higher the concentration of H 2 SO 4 and the extraction time
The longer the reaction time, the higher the recovery efficiency of rare earths because the reaction time and concentration increase.
acidity, the amount of substance in the ore that reacts to dissolve in the acid solution is more. The extraction time is from 3 days or more, the efficiency does not change significantly because the dissolution process reaches saturation. The efficiency of rare earth recovery by the extraction process decreases in the order H 2 SO 4 > HNO 3 > HCl because at room temperature H 2 SO 4 , HNO 3 are more oxidizing than HCl . The efficiency of rare earth recovery by the extraction method with acid solutions is relatively low because in octite minerals, rare earth metals are surrounded by silicate groups, so breaking this layer is difficult at room temperature.
Thus, the highest rare earth recovery efficiency was only 24.8% with 18 M sulfuric acid, 3-day extraction time, and ore/H 2 SO 4 ratio of 1/4.
3.2.2. Hydrometallurgical method with heating using sulfuric acid solution
Experimental results show that the efficiency of hydrometallurgical process of rare earth concentrate by leaching with acids is not effective because the recovery efficiency of rare earth is quite low. Therefore, it is necessary to conduct hydrometallurgical experiments under the influence of other energy sources such as temperature, microwave energy, pressure, etc. The recovery efficiency of rare earth depends on
on factors such as: acid concentration, ore/acid mass ratio, temperature and decomposition time.
3.2.2.1. Effect of sulfuric acid concentration on rare earth recovery efficiency at different temperatures
Hydrometallurgical experiments were conducted with 30 grams of 0.074 mm sized ore using H 2 SO 4 acid with concentrations from 12 to 18 M in a 250 mL flask, ore/acid ratio 1/4, hydrometallurgical temperatures 120 0 C, 150 0 C and 180 0 C,
Hydrometallurgical time of 4 hours for untreated ore and ore initially heat-treated at 500 0 C for 2 hours, stirring speed of 100 rpm. The results of the study on the influence of acid concentration and temperature on the recovery efficiency of total rare earth oxides are presented in Figure 3.8 and Appendix 5.
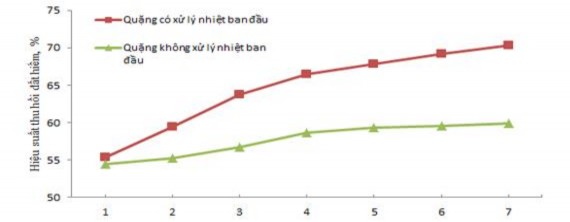
Concentration of H 2 SO 4 , M
a. Effect of H 2 SO 4 concentration on hydrometallurgical efficiency at 120 0 C
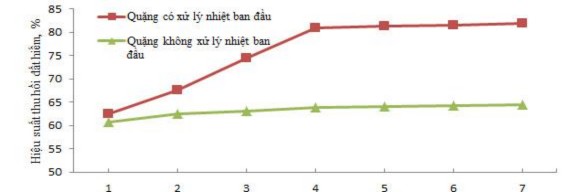
Concentration of H 2 SO 4 , M
b. Effect of H 2 SO 4 concentration on hydrometallurgical efficiency at 150 0 C
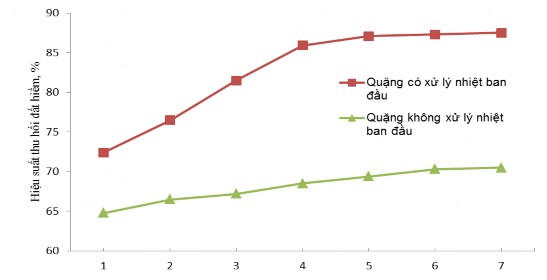
each other
Concentration of H 2 SO 4 , M
c. Effect of H 2 SO 4 concentration on hydrometallurgical efficiency at 180 0 C
Figure 3.8. Hydrometallurgical efficiency of ore atdifferent temperatures and H 2 SO 4 concentrations
From Figure 3.8, it can be seen that the recovery efficiency of rare earth for the type of ore is
The initial heat treatment efficiency is higher than that of untreated ore because the initial heat treatment releases the crystallization water in the ore, burns the impurities that come with the selection process, and partially breaks down the ore structure, converting it to a more reactive form. When the hydrometallurgical temperature increases, the recovery efficiency of rare earth increases due to the increase in the hydrometallurgical reaction rate. When the concentration of H 2 SO 4 increases , the ore decomposition efficiency increases and increases insignificantly when the concentration of H 2 SO 4 is continued to increase to greater than 15 M. Therefore, the suitable conditions for the hydrometallurgical process of ore initially treated with H 2 SO 4 acid are the concentration of H 2 SO 4 15 M and the hydrometallurgical temperature
180 0 C.
3.2.2.2. Effect of ore/H 2 SO 4 ratio on rare earth recovery efficiency
Hydrometallurgical experiments of rare earth-rich fractions initially heat-treated at 500 0 C were carried out with 15 M H 2 SO 4 , hydrometallurgical temperature 180 0 C, the ore/H 2 SO 4 ratio was varied from 1/1 to 1/6. The results of the study on the influence of the ore/H 2 SO 4 ratio on the yield of total rare earth oxides are presented in Fig.
3.9 and appendix 6.





