Landfill
Parameter
Unit | Nyanza (Rwanda) | Ouled Fayet (Algeria) | Jebel Chakir (Tunisia) | |
BOD 5 | mg/l | 674 | 388 | 500 |
BOD5 / COD | mg/l | 0.28 | 0.11 | 0.1 |
SS | mg/l | 1,150 | - | - |
TSS | mg/l | - | 10.4 | - |
+ NH4 | mg/l | 36.5 | 3.159 | - |
- NO 3 | mg/l | 20 | 0.81 | - |
- NO 2 | mg/l | - | 0.75 | - |
N-total | mg/l | 1,207 | 5,236 | - |
3- PO 4 | mg/l | 42.6 | - | - |
P-total | mg/l | - | 37.3 | 8.79 |
2- SO 4 | mg/l | 2,599 | 995 | 76 |
- HCO 3 | mg/l | - | - | 21,750 |
Cl - | mg/l | - | 2.8 | 4,870 |
Cu | mg/l | - | - | 0.14 |
CD | mg/l | - | 1.7 | - |
Zn | mg/l | - | 1.5 | 0.94 |
Cr | mg/l | - | - | 2.24 |
Pb | mg/l | - | - | - |
Mn | mg/l | - | - | - |
Fe | mg/l | - | 21.5 | - |
Source of quotation | [28] | [33] | [90] | |
Maybe you are interested!
-
 Study on botanical characteristics, chemical composition and acetylcholinesterase enzyme inhibitory effects of two species Piper thomsonii (C. DC.) Hook. f. var. thomsonii and Piper hymenophyllum Miq., Piperaceae family - 24
Study on botanical characteristics, chemical composition and acetylcholinesterase enzyme inhibitory effects of two species Piper thomsonii (C. DC.) Hook. f. var. thomsonii and Piper hymenophyllum Miq., Piperaceae family - 24 -
 Study on botanical characteristics, chemical composition and acetylcholinesterase enzyme inhibitory effects of two species Piper thomsonii (C. DC.) Hook. f. var. thomsonii and Piper hymenophyllum Miq., Piperaceae family - 23
Study on botanical characteristics, chemical composition and acetylcholinesterase enzyme inhibitory effects of two species Piper thomsonii (C. DC.) Hook. f. var. thomsonii and Piper hymenophyllum Miq., Piperaceae family - 23 -
 Study on botanical characteristics, chemical composition and acetylcholinesterase enzyme inhibitory effects of two species Piper thomsonii (C. DC.) Hook. f. var. thomsonii and Piper hymenophyllum Miq., Piperaceae family - 21
Study on botanical characteristics, chemical composition and acetylcholinesterase enzyme inhibitory effects of two species Piper thomsonii (C. DC.) Hook. f. var. thomsonii and Piper hymenophyllum Miq., Piperaceae family - 21 -
 Study on botanical characteristics, chemical composition and acetylcholinesterase enzyme inhibitory effects of two species Piper thomsonii (C. DC.) Hook. f. var. thomsonii and Piper hymenophyllum Miq., Piperaceae family - 27
Study on botanical characteristics, chemical composition and acetylcholinesterase enzyme inhibitory effects of two species Piper thomsonii (C. DC.) Hook. f. var. thomsonii and Piper hymenophyllum Miq., Piperaceae family - 27 -
 Characteristics of Business Activities and Business Management Organization at Small and Medium Enterprises in Vietnam
Characteristics of Business Activities and Business Management Organization at Small and Medium Enterprises in Vietnam
1.2.2 . Composition of Vietnamese wastewater
Vietnam still
not yet applied
The method of classification at the source should be component
of leachate is very complex . The leachate not only contains organic matter but also
dissolved inorganic substances, metals
energy, toxic organic substances. Therefore, the problem of
porch
now most of the songs
Landfill in Vietnam encountered
must but no way
The optimal solution is the problem of wastewater treatment .
The composition of leachate can vary greatly, depending on the age of the landfill, the time of sampling – rainy or dry season and following different trends. Therefore, surveying the characteristics of leachate at landfills over a long period of time, right from the beginning of operation, can provide important information as a basis for choosing appropriate treatment technology.
In addition, the design and operating practices of landfills also have important influences on the characteristics of the effluent . For example, although landfills
Go Cat and Phuoc Hiep (Tam Tan), Ho Chi Minh City both treat a source of waste at the same time, but due to the greater depth of the landfill and the recycling of waste water , the organic decomposition process at Phuoc Hiep landfill occurs relatively quickly, within only 5 - 6 months from the time the landfill goes into operation [5].
The results of wastewater analysis are summarized from sources [5, 10, 13, 14] in table 1.5, pH in the range of 6.5 - 8.5 . COD value at the landfill is high: 327 - 22,783 mg/l. Nitrogen concentration fluctuates greatly from 62 - 2,427 mg/l. It can be seen that the most typical characteristics of wastewater are high TDS , BOD5 , COD, total nitrogen content and large fluctuations over time.
Thus, the physicochemical characteristics of leachate are divided into two types: new leachate (2-3 years after the landfill goes into operation) and old leachate ( from the 4th-5th year onwards). It can be seen that new leachate is also divided into two different types: in the first 3-6 months, new leachate is acidic, with high to very high concentrations of COD, BOD, heavy metals, and relatively low pH and NH4 + . In the next stage , the concentration of free ions decreases significantly , pH is neutral, NH4 + begins to increase , but
COD and BOD are still very high.
Comment:
In general, leachate in some landfills in our country also has a large range of organic matter, COD from several hundred to over ten thousand mg/l, even higher than some landfills in Taiwan and Indonesia. The BOD5/COD ratio in some landfills in our country is higher than some landfills in Europe, America and Africa. The suspended solids content presented in Table 1.5 is higher than some landfills in the world.
In many countries around the world, many landfills have applied waste classification at source and applied solid waste recovery and recycling technologies, so the composition and properties of leachate are less complex than landfills in Vietnam. Most solid waste in our country is not classified. Therefore, the composition of leachate in Vietnam not only changes over time but is also more complex than in some other countries. The high and complex composition of leachate in our country is also due to the impact of landfill operations that do not ensure a sanitary landfill and humid, rainy climate conditions. Therefore, choosing appropriate leachate treatment technology in our country also faces many difficulties.
14
Table 1.5. Characteristics of leachate composition in some cities in Vietnam
Parameter
Unit | Nam Son BCL (Hanoi) | BCL Go Cat (Ho Chi Minh) | BCL Thuy Phuong (Hue) | Trang Cat BCL (Hai Phong) | BCL Xuan Son (Hanoi) | |
pH | - | 6.81 – 7.98 | 7.4 – 7.6 | 7.7 – 8.5 | 6.5 – 8.22 | 7.7 |
TDS | mg/l | 6,913 – 19,875 | - | - | 4.47 – 9.24 | - |
TSS | mg/l | 120 – 2,240 | 700 – 2,020 | 42 – 84 | 21 – 78 | 986 |
COD | mg/l | 1,020 – 22,783 | 13,655 – 16,814 | 623 – 2,442 | 327 – 1,001 | 3,540 |
BOD 5 | mg/l | 495 – 12,302 | 6,272 – 9,200 | 148 – 398 | 120 – 465 | 2,150 |
BOD5 / COD | - | 0.485 – 0.540 | 0.459 – 0.547 | 0.234 – 0.163 | 0.370 – 0.465 | 0.607 |
Total N | mg/l | 423 – 2,253 | 1.821 – 2.427 | - | 179 – 507 | 62 |
+ N – NH 4 | mg/l | - | 1,680 – 2,887 | 184 – 543 | - | 17.2 |
- N – NO 3 | mg/l | - | 0 – 6.2 | - | - | 12.5 |
Total P | mg/l | 6.51 – 24.80 | 10.3 – 19.8 | - | 3.92 – 8.562 | 4.31 |
CaCO 3 hardness | mg/l | - | - | 1,419 – 4,874 | - | - |
Cl - | mg/l | - | - | 518 – 1,199 | - | - |
As | mg/l | 0.001 – 0.003 | - | - | 0.047 – 0.086 | 0.2 |
Pb | mg/l | 0.050 – 0.086 | - | - | < 0.05 | 0.34 |
CD | mg/l | 0.010 – 0.025 | - | - | < 0.01 | 0.14 |
Hg | mg/l | 0.0001 – 0.0009 | - | - | 0.0001 | - |
BCL Age | year | 7 | 7 | 9 | 2 | 10 |
Source of quotation | [5] | [13] | [14] | [5] | [10] | |
1.3. METHODS OF LEAKAGE TREATMENT
Wastewater treatment technology is very diverse, with dozens of types of technology being used in practice based on basic technological processes at each level.
The two basic treatment methods applied in leachate treatment are physicochemical and biological methods [1, 5]. Chemical and physicochemical methods include: Coagulation, adsorption, ion exchange, oxidation, precipitation, membrane filtration and sedimentation. Biological methods: Anaerobic, aerobic, anoxic microbial treatment and their combinations.
Each method aims at a processing goal for an object, but a method can also be applied to simultaneously process many objects and to process an object, many different methods can be used. However, in practice, a method or a combination often only aims at one main object.
Some leachate treatment technologies are commonly applied today [7]:
- Anaerobic microbiological treatment → aerobic microbiological treatment → stable pond → discharge to receiving source.
- Anaerobic microbiological treatment → aerobic microbiological treatment → chemical oxidation → stable pond → discharge to receiving source.
- Biological treatment → brought to the treatment plant with domestic wastewater.
- Biological treatment → reverse osmosis (RO) filtration.
- Coagulation → sedimentation (or flotation) → anaerobic treatment → aerobic treatment → stabilization pond → discharge to receiving source.
- Chemical oxidation → sedimentation → anaerobic biological treatment → aerobic biological treatment → stabilization pond → discharge to receiving source.
- Coagulation → sedimentation (or flotation) → anaerobic biological treatment → aerobic biological treatment → adsorption and color treatment → stabilization pond → discharge to receiving source.
Thus, the common leachate treatment technologies are multi-stage treatment, in which biological technology is applied in most of the chains. Physico-chemical technologies are also commonly applied such as coagulation, oxidation, etc.
The effectiveness of some leachate treatment methods was evaluated by Abbas et al. (2009) [17], specifically as follows:
- The method of combining leachate treatment with domestic wastewater is highly effective in treating new and medium leachate, but this method has the disadvantage of residual biomass and nutrients.
- Aerobic process is suitable for treating fresh and medium leachate, the disadvantage is that it is inhibited by difficult to biodegrade substances and biomass residues.
- Anaerobic process is suitable for treating new and medium leachate, the disadvantage is that it is inhibited by substances that are difficult to biodegrade, very slow and produces biogas.
- Coagulation is suitable for old and medium leachate to remove heavy metals and suspended solids. However, this method produces a lot of landfill sludge.
- Chemical oxidation works well with old and medium leachate to remove organic substances. This method has the disadvantage of generating excess ozone, creating sludge with a lot of iron (Fenton process).
- Stripping is suitable for old and medium leachate to treat ammonium, this method requires additional equipment to control air pollution.
- Ion exchange is suitable for all types of leachate to treat dissolved compounds, cations/anions but is expensive.
- Ultrafiltration method is suitable for removing high molecular weight substances but the cost is very high and limited in application due to frequent replacement of filter membranes.
- Nano filtration is well applied to all types of leachate treatment that requires sulfate.
This method is also very expensive.
- Reverse osmosis membrane filtration can be effective in treating leachate to remove inorganic and organic compounds but is very expensive and requires pretreatment.
For many years, biological and physicochemical methods have been the common technologies for leachate treatment. Some membrane filtration methods are effective but expensive. Oxidation methods are suitable for leachate treatment of medium and old age.
1.4. OXIDATION PROCESSES USING OZONE AGENT
1.4.1. Chemical properties of ozone
Ozone (O 3 ) is a strong oxidizing agent with an oxidation potential of 2.07V [44], ozone can react with many organic and inorganic substances in water,
can clean wastewater from phenol, petroleum products, H2S , arsenic compounds, surfactants, cyanides, dyes, aromatic hydrocarbons, disinfectants. Ozone has the molecular formula O3 , at high concentrations it is blue .
Table 1.6 . Physicochemical properties of O 3 molecules [44]
Quantitative analysis
48 | |
Boiling point at atmospheric pressure | -119 ± 0.3 0 C |
Low temperature | -12.1 ± 0.1 0 C |
H (enthalpy) at 298 0 K, 1atm | 34220 ± 240 calories |
Critical pressure | 55.3x105Pa |
Relative density during the atmosphere | 1,657 kg.N/m 3 |
Absolute density under standard conditions | 2,143 kg.N/m 3 |
1.4.2. Oxidation mechanism of ozone
Under standard temperature and pressure conditions, ozone is a pale blue gas, heavier than air. Ozone is unstable, easily decomposed into atomic oxygen and molecular oxygen. Ozone can dissolve in many different solvents, under normal conditions, the solubility of ozone in water is 14 times that of oxygen, however, stability depends on many environmental factors such as cations, metals, heavy metal oxides, temperature, humidity and pressure.
Ozone can oxidize organic compounds in water in two ways :
Direct oxidation by ozone molecules dissolved in water.
Indirect oxidation via hydroxyl radical ( OH )) when O 3 decomposes in water.
Direct oxidation by O 3 molecules occurs relatively slowly compared to indirect oxidation via hydroxyl radicals ( OH ) produced by the decomposition of ozone [11, 41, 59, 98].
Two oxidation pathways of O 3with organic matter
ok
Gottschalk et al . (2010) [46] described in Figure 1.2.
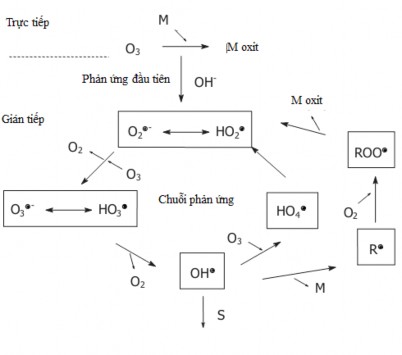
(S: interfering substance , M : other low concentration pollutant , R : reaction product)
Figure 1.2. Indirect and direct ozonation models
In acidic environments , the direct oxidation pathway by molecular O 3 is dominant. O 3 reacts with organic compounds containing C=C double bonds or aromatic ring bonds and decomposes them into carboxylic acids and aldehydes [63, 102]. Molecular O 3 reacts easily with organic substances such as amines, phenols and aromatic ring compounds but reacts slowly with carboxylic acids, aldehydes and alcohols [53]. In alkaline environments, in the presence of OH - ions ions , O 3 decomposes rapidly and forms radicals
flexible hydroxyl ( OH ) and this radical oxidizes organic substances in water and wastewater[53]. The reaction of molecular O 3 ( E 0 = 2.07V) is slow and limited while the radical OH radical ( E 0 = 2.80V) reacts with most organic substances in water and wastewater [34, 47, 104].
Reaction of O3
and OH - ions form radicals
O and root
HO . Reaction
2
2
between O 3 and O 2 radicals
re-forms ozonide radicals
O3 , this radical is decomposed immediately
immediately and form the mobile hydroxyl radical OH In summary, the reaction of the three fractions
2
O 3 forms two mobile hydroxyl radicals OH [70]
3 O 3
OH H 2OH 4 O
(1.1)
Therefore, instead of using Ozone alone, many research projects have developed in the direction of finding agents that coordinate with ozone or catalysts to create
origin
OH
to enhance the oxidation efficiency of ozone when it is necessary to treat stable compounds.
stable, difficult to decompose in water and wastewater. The most studied added agents are H 2 O 2 , catalysts are salts Ni(II), Co(II), metal oxides TiO 2 , MnO 2 ...
The process of ozonation of wastewater simultaneously oxidizes the impurities carrying
color, disinfect, detoxify, saturate water with oxygen. Oxidation of organic compounds by ozone can form intermediate products: alcohols, aldehydes, ketones, acids and due to its strong oxidizing ability, it can oxidize further to produce CO 2 and H 2 O.
1.4.3. The process of combining Ozone with other agents
a. Perozon process
The basic difference between the single ozone and perozone processes is that the single ozone process oxidizes pollutants mainly directly by the strong oxidizing properties of the O 3 molecule itself in water. Meanwhile, the perozone process oxidizes pollutants mainly indirectly through the hydroxyl radical ( OH ) is made up of
combine
ozone and hydrogen peroxide (O 3 /H 2 O 2 ).
When H 2 O 2 combines with O 3, it begins the O 3 decomposition cycle to form radicals.
flexible hydroxyl OH [70]:
H
2 O 2
HO H
(1.2)
2
HO O HO O
(1.3)
2 3 2 3
2
The reaction continues via the indirect pathway and forms the mobile hydroxyl radical OH .
3
2 O H
2 O 2
2 HO 3 O
(1.4)
In the Perozon process, due to the presence of hydrogen peroxide, the ozone consumption and decomposition process is significantly increased, enhancing the transfer of ozone from the gas phase to the liquid phase.
Hydroxyl radical formation reaction ( OH ) in reaction 1.4 shows the radical
OH
born
In the Perozone system reaction, there are more (2 mol O 3 will produce 2 OH radicals )) compared to the reaction
in single ozone system (3 mol O 3 only produce 2 OH radicals )). Because the oxidation process through





