- Requirements of inguinal hernia surgery must ensure:
+ Removal of hernia sac: indirect hernia dissection, cutting, and high neck ligation of the hernia sac, blocking the path of the herniated organ. Direct hernia, femoral hernia does not need to be removed, but suturing the sac inside is enough.
+ Abdominal wall restoration: increase the strength of the already weak abdominal wall, which is now weakened further by the hernia. Abdominal wall restoration can be done with autologous tissue or with artificial mesh [11], [131], [132].
1.7.2. Techniques for treating inguinal hernias with autologous tissue
1.7.2.1. Deep inguinal ring stenosis
Maybe you are interested!
-
 Corresponding Techniques for Task Type T 1 Μ , T 1 P , T 1 2
Corresponding Techniques for Task Type T 1 Μ , T 1 P , T 1 2 -
 Applying the process of caring for, preventing and treating diseases for breeding sows and piglets raised by their mothers at Thien Thuan Tuong Mineral Exploitation Joint Stock Company, Cam Pha, Quang Ninh province - 8
Applying the process of caring for, preventing and treating diseases for breeding sows and piglets raised by their mothers at Thien Thuan Tuong Mineral Exploitation Joint Stock Company, Cam Pha, Quang Ninh province - 8 -
 Obstetrics and Gynecology Traditional Medicine Part 2 - 8
Obstetrics and Gynecology Traditional Medicine Part 2 - 8 -
 Modern surgical medicine Part 2 - 1
Modern surgical medicine Part 2 - 1 -
 Traditional Medicine Systems in Countries Around the World
Traditional Medicine Systems in Countries Around the World
Marcy method, suturing the internal oblique muscle with the transverse fascia to close the deep inguinal ring, with the aim of restoring the ring mechanism at the deep inguinal ring, has a very high recurrence rate of 21 - 36% [114], [132].
1.7.2.2. Abdominal wall repair using inguinal ligament
Typical methods are Bassini, Shouldice, Ferguson, Haltesd by: suturing the internal oblique muscle, transverse abdominal muscle and superior layer of the transverse fascia to the inguinal ligament with interrupted sutures or crossed sutures [9], [11], [16], [131], [132].
1.7.2.3. Abdominal wall reconstruction using the iliopubic band
Nyhus method, suturing the combined tendon to the iliopubic band [134].
1.7.2.4. Abdominal wall reconstruction using pectinate ligament
Mc Vay method, suturing the tendon combined with the pectinate ligament with separate sutures starting from the pubic bone outward to the deep inguinal ring, with a dilated incision on the anterior surface of the rectus abdominis muscle sheath, the recurrence rate is 2%, inexperienced surgeons the recurrence rate is 15 - 20% [114], [128], [133].
1.7.2.5. XI Kimbarovski method
Deep layer: sutures the medial edge of the external oblique muscle and the tendon associated with the inguinal ligament.
Superficial layer: suture the outer edge of the external oblique fascia onto the deep layer in a cross-dressing fashion.
1.7.2.6. MA Xpaxokukoski method
Deep layer: suture the inner edge of the external oblique muscle to the inguinal ligament. Superficial layer: suture the outer edge of the external oblique muscle to the deep layer,
into the rest of this scale in a cross-over fashion [9], [11].
1.8. INGUINAL HERNIA SURGERY WITH ARTIFICIAL MESH
1.8.1. Types of artificial mesh used in hernia repair
Artificial mesh has been used in hernia repair since the late 19th century. Up to now, there have been many different materials, including two groups: non-dissolvable materials and soluble materials [12], [17], [20].
1.8.1.1. Insoluble material mesh
- Metal mesh: used quite early in hernia surgery, including silver mesh, tantalum and stainless steel. Currently not used because of disadvantages: hardness hinders the patient's activities, completely inert to the body leading to fluid accumulation, intestinal adhesion, formation of cavities, increasing the possibility of infection, the mesh can break and cause recurrent hernia at the fracture site.
- Rarely used non-metallic mesh: fortisan, polyvinyl, nylon, silastic, polytetrafluoroethylene - PTFE (Teflon), carbon fiber.
- Polymer mesh is being used in hernia:
+ Polyester ( Mercilene, Dacron): polymer of ethylene glycol and terephtalic acid, discovered in 1939, by the late 1950s it was woven into nets and marketed under the names Mercilene and Dacron.
+ Polypropylene (Marlex, Prolene, Premilene, Optilene): the most popular mesh type currently used in surgery to treat inguinal and abdominal hernias, has many advantages over other types such as being soft, can be shaped freely without fear of breaking, is well tolerated, and does not cause discomfort to the patient.
+ Expanded PolyTetraFluoroEtylene - ePTFE (Gor Tex): fluorinated polymer with the formula (-CF2-CF2-) n, discovered by chance by Plunkett in 1938. In 1963, Shisahuro Oshige discovered a process of stretching PTFE to create a fibrous, porous, continuous and uniform structure that gives it high tensile strength called ePTFE. It is inert to tissue, causes little inflammatory reaction or rejection of the artificial mesh, and has better tensile strength than polypropylene.
and polyester, flexible, soft, woven into a mesh with holes of about 20 - 25 microns allowing fibrous tissue to penetrate and organize it [12], [17], [20].
1.8.1.2. Mesh made of soluble material
The polyglycolic acid (Dexon) mesh is soft, foldable, degradable, and gradually dissolves after 90 days.
Polyglactin 910 mesh (Vicryl) is also foldable, non-elastic, and has similar physical and temporal characteristics to Dexon [12], [17], [20].
1.8.2. Open surgery to place artificial mesh
Open surgery to place the mesh can be performed classically through the anterior approach, or the preperitoneal approach. Open surgery through the anterior approach is commonly used and the common techniques depend on the size of the mesh. Flat type such as: Lichtenstein, stopper type such as: Gilbert, Rutkow and Robbins, double mesh consisting of two meshes connected together PSH ® , Kugel mesh [79], [114], [132].
- Advantage
+ Does not cause tension so less pain, quick recovery, early return to work.
+ Does not cause tension so tissue does not lack blood supply, scars heal well, low recurrence.
+ The mesh, when organized, will create a new, very strong layer of fascia, which is very necessary for people whose autologous tissue is thin and weak, and is not able to withstand the force if surgery is performed using autologous tissue techniques.
- Disadvantages
+ When using the mesh, absolute sterility is required.
+ When the infection is difficult to treat, the mesh may have to be removed.
+ The cost of the mesh is still relatively high [18], [127], [132].
1.8.2.1. Lichtenstein's artificial mesh method
Lichtenstein technique of placing artificial mesh, the posterior wall of the inguinal canal is covered with polypropylene mesh, placed along the inguinal canal from outside to inside. It is necessary for the inner end of the mesh to cover the inguinal ligament and the tail of the mesh can be tucked under the upper layer of the external oblique muscle and the mesh is sutured to fix the mesh.
mesh to the pubic tubercle, inguinal ligament, combined tendon, and rectus abdominis sheath. Suture the two tail flaps of the mesh over the spermatic cord.
1.8.2.2. Gilbert's Plug Method
This technique was described by Gilbert in 1992, applied to indirect inguinal hernias, performed under local and regional anesthesia to allow the surgeon to push the mesh into place and ask the patient to cough.
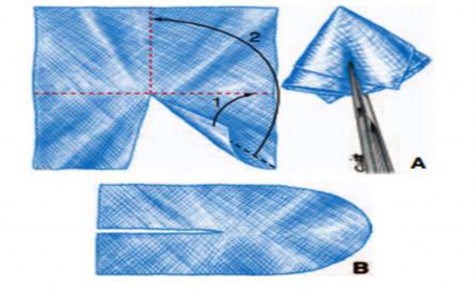
Figure 1.10. A. Creating a plug. B. Flat mesh. Source: Pélissier E., Palot JP, Ngo P., (2007) [132]
1.8.2.3. Rutkow and Robbins Plug Method
This technique was described in 1993 and is effective for indirect and direct hernias. The Perfix® plug is made of polypropylene and consists of two components: the cone-shaped part is presented as a round cone with many short wings, placed to close the deep inguinal ring. The flat part of the mesh is oval, split into two wings, hugging the spermatic cord and placed in the posterior wall of the inguinal canal without fixation.

Figure 1.11. Flat and plug grid. Source: Pélissier E., Palot JP, Ngo P., (2007) [132].
1.8.2.4. Method of placing PSH ® mesh
The PSH ® mesh consists of three interconnected parts. The lower part, a circle with a diameter of 10 cm, is placed in the Bogros space behind the transverse fascia. The upper part, a rectangle, is placed in front of the transverse fascia along the axis of the inguinal canal, behind the external oblique muscle. The cylindrical tube connecting the two parts is 1 cm high and is located in the center of the mesh. The main point of the PSH ® mesh is that it is convenient and safe when placed behind the transverse fascia, strengthening the transverse fascia to withstand abdominal wall pressure.
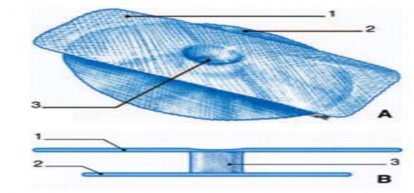
1. Shallow mesh; 2. Deep mesh; 3. Connecting tube.
Figure 1.12. PSH ® mesh . Source: Pélissier E., Palot JP, Ngo P., (2007) [132].
1.8.2.5. Kugel's mesh placement method
Along with other techniques such as Gilbert, Rutkow, Robbins, Lichtenstein. North American surgeon, Kugel developed the technique of placing artificial mesh, preperitoneal position by a short 3 - 5cm skin incision in the groin area, without approaching the inguinal canal by the anterior approach [129].
Figure 1.13. Preperitoneal space. Figure 1.14. Location of Kugel mesh.
Source: Martel P., (2007) [129].
Among the techniques using artificial mesh to treat inguinal hernia, we chose the Lichtenstein technique because according to the literature, it is easy to perform, has a low recurrence rate, a short training curve, and can be widely applied to hospitals.
1.8.3. Placement of artificial mesh by endoscopic and extraperitoneal routes
- Advantage
+ Safe, effective, bringing many benefits to patients.
+ The incision is small so the patient has less pain after surgery than with open surgery and also recovers faster.
+ Small incision so more aesthetic.
- Disadvantages
+ Complex technique, so training and surgery time is longer.
+ Not widely applied because it requires endoscopic surgical systems and instruments.
+ General anesthesia is required, which increases surgical risks.
- Commonly used endoscopic techniques, there are 3 popular techniques for placing mesh through endoscopy
+ Transabdominal extraperitoneal technique (TAPP).
+ Intraperitoneal mesh placement technique (IPOM).
+ Totally extraperitoneal mesh placement technique (TEP) [17], [18], [127].
1.8.4. Technical requirements of an artificial mesh
Since 1950, Cumberland and Scales have proposed the indispensable requirements of an ideal artificial mesh:
Not physically altered by tissue fluid, chemically inert, does not cause inflammation or mesh rejection, is non-allergenic or non-sensitizing, is non-carcinogenic, withstands mechanical stress, is soft to form desired shape, can be sterilized and does not cause infection.
Debord later added some requirements for the artificial mesh:
Must be strong, durable, neutral, the tissue must be able to grow through the mesh, stimulate fibrous tissue to cover the weak areas on the abdominal wall, create a non-adhesive surface with the abdominal wall and internal organs, and be stable in the patient's body [19], [48].
1.8.5. Tissue organization when placing artificial mesh in the groin area
The healing process of tissue after inguinal hernia surgery lasts about a year, involving the synthesis, completion of collagen and organization into a solid structure.
Wound healing and tissue repair is an automatic process consisting of three stages: acute and chronic inflammation, proliferation and tissue remodeling.
1.8.5.1. Inflammatory phase
Occurs early, just a few hours after surgery. Due to the effects of trauma during surgery, blood vessels dilate and increase blood flow to the surgical site, causing blood components and inflammatory cells: neutrophils, lymphocytes and macrophages to seep into the extracellular space of the wound area.
- In the acute inflammatory stage
Fibrin meshwork appears very early at the site of the meshwork, fibrin provides a pathway for neutrophils and macrophages to reach the foreign body. At the wound, the entire surface of the meshwork is infiltrated by polymorphonuclear leukocytes.
Inflammatory reactions occur on the surface of the tissue and mesh mainly due to monocytes and macrophages. Monocytes release cytokines including: tumor necrosis factor α (TNF-α), interleukin-6 (IL-6), interleukin-10 (IL-10), forming a local defense barrier, less likely to cause infection, wound healing and scar tissue. On the third day, most of the mesh surface is covered with a thin layer of tissue. Proliferating fibrocytes invade the gaps of the mesh.
- Chronic inflammatory phase
After the acute inflammatory phase of about 5-7 days. Mononuclear phagocytes begin to invade, becoming resident macrophages, eating germs, dead tissue fragments and foreign bodies. Because the mesh cannot be eaten, they combine with the mesh to form giant cells - foreign bodies. On the 5th day, epithelial cells have appeared and covered the entire surface of the mesh. On the 7th day, there are no more gaps and the surface of the mesh is covered by a layer of epithelial cells interwoven with many microvilli.
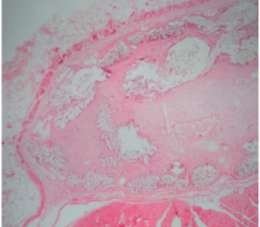
a×40 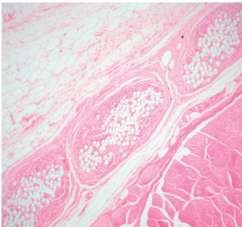 b×100
b×100
Figure 1.15. Foreign body-giant cell and fibroblast cell reactions found after mesh placement. Source: Witherspoon P., Bryson G. et al., (2004) [119].
1.8.5.2. Tissue proliferation phase
Usually about a week after surgery. Macrophages and giant cells - foreign bodies, on the surface of the mesh secrete cytokines, promoting the proliferation of fibroblasts and the formation of capillaries. Fibroblasts will synthesize collagen.
Healing continues until the entire mesh is organized into the body, the gaps in the incision are filled. As fibrous tissue infiltrates into the entire mesh gap, the mesh will contract and decrease in size by 10 - 20% in all directions.





