significantly or even slightly increased. With silica gel, when the concentration increased from 0-1.5%, the tensile strength also decreased from 17.1 mN/m to 16.4 mN/m. However, when it increased to 2%, the SCBM of the solution tended to increase again. At a concentration of 2% by mass of silica gel, the SCBM value was 16.5 mN/m. With siloxane, due to its nature as a surfactant, increasing its concentration in the solution will cause the SCBM of the system to decrease. The results showed that when the concentration of siloxane increased from 0-2% by mass, the SCBM of the system decreased from 17.1 mN/m to 16 mM/m. Therefore, the thesis chose the concentration of sodium silicate at 1.0%, silica gel at 1.5% and siloxane at 1.5% for further studies.
On the other hand, the results showed that when increasing the content of sodium silicate, silica gel and siloxane from 0 - 2.0% by mass, the SCBM of the solution gradually decreased, proving that when adding sodium silicate, silica gel or siloxane compounds to the fire-fighting foam, interactions occurred with the surfactants, changing the SCBM of the solution. Some scientists in the world have proposed hypotheses to explain this phenomenon such as Vatanparast et al. (2017) [109] that hydrophilic silica particles in silica-containing compounds after being dispersed into the surfactant solution often do not adsorb at the air/liquid interface. Meanwhile, hydrocarbon surfactant molecules, especially non-ionic surfactants, preferentially adsorb on the surface of silica nanoparticles [110-113]. Therefore, the change in SCBM of AFFF fire-fighting foam is not due to the adsorption of silica nanoparticles at the air/liquid interface.
When there is no nano silica in the solution, fluorinated surfactant molecules and hydrocarbons coexist at the air/liquid interface. When nano silica is added, the number of charged surfactant molecules at the air/liquid interface decreases with increasing nano silica concentration. The uncharged fluorinated surfactants are adsorbed onto the air/liquid interface thereby reducing the SCBM to a value close to that of the solution containing only fluorinated surfactants. Therefore, the SCBM of foams containing nano silica particles shows the presence of more fluorinated surfactants than hydrocarbon surfactants [114]. On the other hand, the addition of nano silica particles and the adsorption of hydrocarbon surfactants onto the surface of nano silica increases the viscosity. The molecular interaction mechanism with and without nano silica particles is illustrated in Figure 3.13.
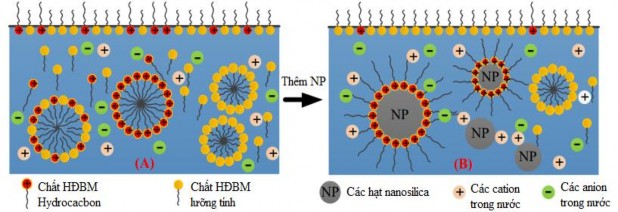
Figure 3.13 . Molecular interactions at the air/liquid interface [105]
(A) without silica nanoparticles and (B) with silica nanoparticles
Continuing to investigate the effect of silicon-containing compounds on AFFF foam properties, the results are presented in Table 3.48.
Table 3.48. Effect of silicon-containing compounds on AFFF solution
Sample
Surface tension (mN/m) | Interfacial surface tension (mN/m) | Spread coefficient | Viscosity (mPa.s) | |
AFFF solution | 17.08 | 3.73 | 3.42 | 1.21 |
AFFF solution containing sodium silicate | 16.18 | 2.00 | 6.05 | 1.45 |
AFFF solution containing silica gel | 16.30 | 3.53 | 4.4 | 1.86 |
AFFF solution containing siloxane | 16.58 | 3.18 | 4.47 | 1.35 |
Maybe you are interested!
-
![Water Film Forming Fire Extinguishing Foam for Fuel Tanks and Gas Stations [9]](data:image/svg+xml,%3Csvg%20xmlns=%22http://www.w3.org/2000/svg%22%20viewBox=%220%200%2075%2075%22%3E%3C/svg%3E) Water Film Forming Fire Extinguishing Foam for Fuel Tanks and Gas Stations [9]
Water Film Forming Fire Extinguishing Foam for Fuel Tanks and Gas Stations [9] -
 Competing Competitive Advantages of Convenient Air and Sea Access
Competing Competitive Advantages of Convenient Air and Sea Access -
 Basic Refrigeration Refrigeration and Air Conditioning Engineering - Intermediate - Ha Nam Vocational College 2020 - 1
Basic Refrigeration Refrigeration and Air Conditioning Engineering - Intermediate - Ha Nam Vocational College 2020 - 1 -
 Linking low-cost air travel in international economic integration in Vietnam - 22
Linking low-cost air travel in international economic integration in Vietnam - 22 -
 Proportion of Highly Liquid Assets Before and After Deducting Loans from State Bank and Commercial Banks in the Market of Some Commercial Banks
Proportion of Highly Liquid Assets Before and After Deducting Loans from State Bank and Commercial Banks in the Market of Some Commercial Banks
The results showed that the spreading coefficient of the foam solutions all had positive values, proving that these AFFF solutions were capable of forming a water film on the surface of cyclohexane. In addition, when silicon-containing compounds were added to the composition, the spreading coefficient increased, which means that the formation and spreading speed of the water film on the surface of cyclohexane increased.
The viscosity of the typical AFFF solution is 1.21 mPa.s and increases with the addition of sodium silicate, silica gel and siloxane (1.45; 1.86 and 1.35 mPa.s). This is explained by the fact that these compounds have very high viscosity, so when added to the foam solution, the viscosity of the fire-fighting foam solution will increase.
3.3.2. Effect of some silica-containing compounds on foam stability
Figure 3.14 shows the solution samples after foaming using a double syringe connected together, the foam produced is released into a glass vial. Sample number 1 is the AFFF foam solution containing 1% sodium silicate; sample number 2 is the AFFF foam solution containing 1.5% silica gel; sample number 3 is the AFFF foam solution containing 1.5% siloxane. All concentrations are in mass and sample number 4 is the control AFFF solution sample. At the initial time, the 4 foam samples have the same volume. The half-life of the foam under the influence of gravity occurs over time. After 25 minutes, the liquid released from bubbles 1, 2, 3 and 4 reaches the initial liquid volume of 20 ml.

Initial time
After 5 minutes | |
After 10 minutes |
After 15 minutes |
After 20 minutes |
After 25 minutes |
Figure 3.14 . Foam samples formed by the 2-syringe technique.
The foam volume of the four foam samples decreased over time due to the increasing amount of liquid released. After the first 5 minutes, the amount of water released in samples 1 and 2 was greater than in samples 3 and 4, and the foam height of the samples did not change. After 10 minutes, sample 4 had the least amount of water released, but the foam height decreased lower than samples 2, 3, and 4. At the 15th minute, sample 3 also had a decrease in foam height, while samples 1 and 2 had no change in foam compared to the initial time. After 25 minutes, there was almost no foam left in samples 3 and 4, and the amount of foam in sample 2 began to decrease, and the bubbles inside were larger than those in sample 1. This proves that silica nanoparticles have the ability to stabilize foam more than conventional AFFF. Although the half-life is faster than AFFF, the foams formed are more durable. In which, the AFFF foam solution sample containing sodium silicate at a concentration of 1% has the most stable foam.
Previous studies have also shown that silica nanoparticles have the ability to stabilize foam by dispersing into the foam membrane and interface [67, 114, 115]. The results obtained in the thesis also confirm that silica nanoparticles disperse into the foam membrane, making the foam more stable. However, silica nanoparticles accelerate the drainage process of foam. This phenomenon is due to the adsorption of a large amount of hydrocarbon surfactant molecules onto the surface of silica nanoparticles, reducing the amount of hydrocarbon surfactants at the air/liquid interface and electrostatic interactions in the foam sample. This makes the structure of nanoparticles heterogeneous on the foam membrane and at the interface, which are factors that accelerate the drainage process of foam. From the results obtained, the thesis selected the sodium silicate content of 1% and silica gel of 1.5%.
Continue to study the effects of sodium silicate and silica gel on foam stability when used at concentrations of 1% and 1.5%, respectively. Observing the foam morphology of these two foam samples under a microscope gives the results as shown in Figure 3.15.
silicate (sample 1)
AFFF contains 1% sodiumAFFF contains 1.5% gel silica (sample 2) | AFFF (Form 3) | ||
1 minute |
|
|
|
5 minute |
|
|
|
10 minute |
|
|
|
20 minute |
|
|
|
Figure 3.15 . Change in foam bubble morphology over time
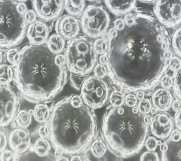
The time-dependent foam morphology of the foam samples shows that the bubble sizes of the three samples are almost the same in the initial stage (1 min). The bubbles gradually grow and coalesce over time due to the pressure difference in the bubbles of different sizes [115]. However, the morphology of the foam bubbles is also different for different foam samples. The bubbles in sample 1 do not change much from 1 min to 20 min. The large bubbles contain many small bubbles inside.
This phenomenon is due to the stability of the bubble membrane and the high viscosity of the foam. When the foam bubbles gradually grow larger and burst, the small foam bubbles are released, which helps increase the durability of the foam layer. Therefore, after 20 minutes, the foam height does not change much, but only the size of the foam bubbles changes. With sample 2, in the first 10 minutes, the foam bubbles also gradually grow in size and the large bubbles also contain small bubbles, but the number of small bubbles inside is less than in sample 1. By the 20th minute, many large foam bubbles exist alone without containing small bubbles inside, so the foam height begins to decrease rapidly. With sample 3, the amount of bubbles in the sample decreases significantly over time. After 20 minutes, most of the foam bubbles only exist alone, so the bubbles are less durable than in samples 1 and 2, leading to a rapid increase in water secretion during this period. The results showed that silica nanoparticles have a significant effect on foaming and foam stability. At certain concentrations, silica nanoparticles can inhibit the solidification process and enhance foam stability.
The influence of nano silica on foam stability is explained as analyzed by the group of authors Y. Sheng [105] shown in Figures 3.16 and 3.17.

Figure 3.16 . Remaining bubble structure after 2 hours [105]
The red arrows indicate the inter-bubble space and the green arrows indicate the bubble membrane. The blue arrows indicate the liquid droplets evaporated from the bubble membrane. The yellow arrows indicate the traces left after the bubble membrane collapse. The bubble membranes in the sample are stabilized by nano silica so that when the liquid in the foam has completely drained, the bubble structure still exists. The bubbles are small in size and have a complete structure. The mechanism to explain this phenomenon is shown in Figure 3.17 [105].
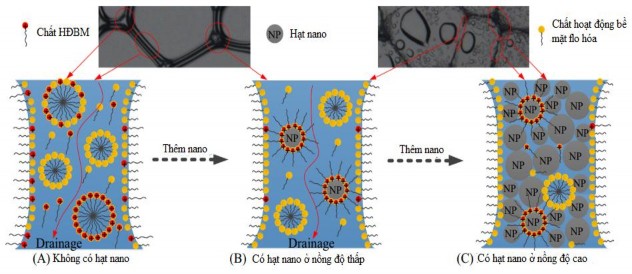
Figure 3.17 . Mechanism of influence of nano concentration on foam stability [105]
In the absence of nano silica particles, the foam is stabilized by a mixture of hydrocarbon and fluorinated surfactants. When adding nano silica particles at low concentrations, it will destroy the adsorption equilibrium and reduce the amount of surfactants at the interface. Therefore, it cannot slow down the half-life of the foam film, resulting in poor foam stability. As the concentration of nano silica particles increases, they gradually aggregate in the foam film and the inter-bubble space, slowing down the foam drainage. When the concentration of nano silica increases, the nano silica particles will fill the film and the inter-bubble space, resulting in slow drainage and half-life, and the foam has high stability.
3.4. Testing
3.4.1. Small-scale testing
3.4.1.1. Small-scale testing of water-based foam concentrate
To evaluate the thermal stability of the studied fire extinguishing foam system, fire extinguishing efficiency tests were conducted using strong and light spray methods. The test results on a scale of 0.6 m 2 using strong and light spray methods are presented in Table 3.49.
Table 3.49 : Test results to determine the fire extinguishing time of water-based foam on a small scale.
STT
Test time | Fire Extinguishing Time (seconds) | Re-ignition time (≥ 300 seconds) | ||
Strong spray (≤ 180 seconds) | Light spray (≤ 300 seconds) | |||
1 | First time | 79 | 90 | 409 |
2 | 2nd time | 75 | 101 | 419 |
3 | 3rd time | 78 | 94 | 413 |
Medium | 77 | 95 | 414 | |
The test results show that the fire extinguishing ability of the foam sample on a small scale, the fire extinguishing time is relatively fast 77 seconds for the strong spray method, the light spray method is 95 seconds. This proves the compatibility and heat resistance of the mixture of surfactants and additives. The re-ignition time is 414 seconds compared to TCVN.
3.4.1.2. Small-scale testing of alcohol-resistant water-based foam concentrate
The results of the fire extinguishing efficiency test on a scale of 0.25 m 2 of the alcohol-resistant water-film forming fire-extinguishing foam concentrate are presented in Table 3.50.
Table 3.50 : Test results of fire extinguishing efficiency of alcohol-resistant water-based foaming agent
STT
Test times | Test results | ||
Ignition time (≤ 180 seconds) | Re-burn time (≥ 600 seconds) | ||
1 | First time | 75 | 767 |
2 | 2nd time | 71 | 754 |
3 | 3rd time | 68 | 759 |
Medium | 71 | 760 | |
The test results show that the fire extinguishing efficiency of the alcohol-resistant water-based foaming agent with a fire extinguishing time of 71 seconds meets the TCVN 7278-3:2003 standard, and the re-ignition time is 760 seconds, higher than the standard of 160 seconds in the standard. Therefore, it proves that the constructed foaming agent system is compatible and has good thermal stability.

![Water Film Forming Fire Extinguishing Foam for Fuel Tanks and Gas Stations [9]](https://tailieuthamkhao.com/uploads/2022/07/13/nghien-cuu-xay-dung-he-chat-hoat-dong-be-mat-ben-nhiet-ung-dung-trong-3-1-120x90.jpg)