Stage II: The ovaries begin to develop, at this stage the ovaries increase in size and can be distinguished by the naked eye. The ovaries are slightly opaque white, with a thin membrane, making it difficult to see the egg grains with the naked eye.
Stage III: The size of the ovary increases and takes up a significant amount of space in the abdominal cavity. The ovary is pale yellow. The eggs can be seen leaking through the transparent membrane with the naked eye. They are very small and difficult to separate.
Stage IV: Large in size, occupying a large area in the abdominal cavity, bright yellow in color, darker than stage III. At this time, the eggs are large in size, relatively uniform and can be separated.
Characteristics of male fish gonads:
The seminal vesicles are two small, opaque white strips located close to the spine, covered on the outside by a thin membrane. One end is attached to the genital opening, the other end is free and lies between the internal cavity. (Figure 4.10)
Stage I: The seminal vesicles at this stage are two thin strands, and cannot be distinguished with the naked eye.
Stage II : The seminal vesicles begin to develop. During this stage, the size of the seminal vesicles increases and can be distinguished with the naked eye.
The size of the seminal vesicles at this stage increases compared to stage I. At this time, the seminal vesicles are two thin, flat, white strips. Spermatocytes appear at this stage.
Stage III: The seminal vesicles are larger than in stage II, and are opaque white. The anterior part of the seminal vesicle is larger than the posterior part.
Stage IV: During this period, the seminal vesicles are much larger in size than in the previous stage.
III. The seminal vesicles are milky white and have developed blood vessels.
4.3.2 Sexual maturity stage of Sillago sihama
Based on the research results in Figure 4.12, it shows that stage I-II of sardines in December accounts for 82.86%, January accounts for 60%, February accounts for 50%, the proportion of female fish with gonads in stage I-II accounts for the majority, but this proportion gradually decreases and reaches the lowest value in June at 18.18%. In contrast to stage I-II, the proportion of gonads in stage III from December to June increases gradually and this proportion reaches the highest in June at 51.51%. This shows that as the reproductive season approaches, the gonads develop more in stage III. (Figure 4.12)
Based on the maturity scale of author Nikolsky (1963), the results determined that the ovaries of the sablefish reached the highest stage IV in June (30.30%); in December
Stage IV ovaries were not seen (Figure 4.12). During the sampling and analysis process, stage V ovaries were not seen. This may be the stage when fish participate in reproduction, so it is very difficult to detect this stage.
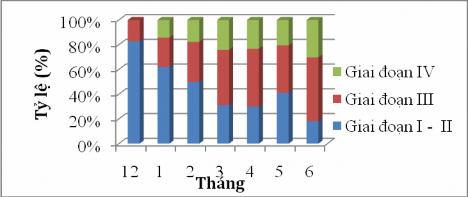
Figure 4.12 Sexual maturity stage of female Sillago sihama over the months.
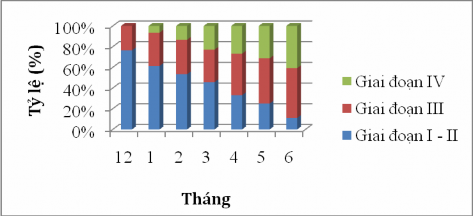
Figure 4.13 Sexual maturity stage of male sillago sihama over the months.
Similar to the ovary, the spermatogonia stage I-II in December accounted for 71.47%, January accounted for 61.30% and February accounted for 53%. The proportion of male fish with gonads in stage I-II was the majority, but this proportion gradually decreased and reached the lowest value in June at 11.11%.
The number of seminal vesicles in stage III increased gradually from December (23.53%) to June (48.15%). The number of seminal vesicles reached the highest level of stage IV in June, accounting for 30.30%; in December, the seminal vesicles did not appear in stage IV (Figure 4.13). This is the stage when the fish reach sexual maturity and participate in reproduction. It is possible that during the sampling period, only small-sized octopus species were collected or due to the reproductive habits of the fish.
Through the results of the ovarian maturity stage of female fish in Figure 4.12 and the seminal vesicle of male fish in Figure 4.13, it can be seen that before entering the reproductive period, the maturity stage mostly falls in stage I - II and accounts for a high proportion from December to March. When entering the reproductive season, the gonads develop larger and stage I - II gradually decreases, instead, stage III - IV increases for fish to participate in reproduction. The annual spawning period of sardines falls between April and June.
4.3.3 Variation in male to female ratio
The results of recording the male: female ratio of 457/551 mackerel samples collected in Tran De district - Soc Trang are shown in table 4.5. Of which, 94 samples had undetermined sex because the fish were too young to distinguish between male and female. With the results in table 4.5, the ratio ♂/♀ in the composition of mackerel samples was approximately 1: 1.04. During the analysis, it was shown that the proportion of male fish during the reproductive period was often more than that of female fish.
The sex ratio of sardines fluctuates but is almost equal, with 51% of males and 49% of females.
Table 4.5: Showing the sex ratio of the mullet Sillago sihama
Time
Male | Female | Total | Male / female ratio | |
December | 34 | 35 | 69 | 1:1.04 |
January | 42 | 31 | 73 | 1:0.70 |
February | 30 | 34 | 64 | 1:1.34 |
March | 35 | 29 | 64 | 1:0.82 |
April | 30 | 30 | 60 | 1:1 |
May | 35 | 32 | 67 | 1:0.92 |
June | 27 | 33 | 60 | 1:1.2 |
Total | 233 | 224 | 457 | 1:1.04 |
Maybe you are interested!
-
 Study on some biological characteristics of slugfish Sillago sihama Forsskal, 1775 in Tran De district - Soc Trang - 1
Study on some biological characteristics of slugfish Sillago sihama Forsskal, 1775 in Tran De district - Soc Trang - 1 -
 Characteristics of the Practice of the Right to Prosecute During the Investigation Stage of a Case of Intentionally Causing Injury or Harm to the Health of Another Person.
Characteristics of the Practice of the Right to Prosecute During the Investigation Stage of a Case of Intentionally Causing Injury or Harm to the Health of Another Person. -
 Research on some technical measures for fertilizing Coffea canephora Pierre coffee trees in the business stage on basalt soil in Dak Lak - 25
Research on some technical measures for fertilizing Coffea canephora Pierre coffee trees in the business stage on basalt soil in Dak Lak - 25 -
 Distinguishing the Crime of Sexual Intercourse or Other Sexual Acts with a Person from 13 to Under 16 Years Old (Article 145) from the Crime of Rape
Distinguishing the Crime of Sexual Intercourse or Other Sexual Acts with a Person from 13 to Under 16 Years Old (Article 145) from the Crime of Rape -
 Document Generation Diagram In Each Stage Of Contract Implementation Tvxd
Document Generation Diagram In Each Stage Of Contract Implementation Tvxd
The results of this study are different from some previous studies on the biological characteristics of silver grouse. The male-to-female ratio of silver grouse in Nha Phu lagoon - Khanh Hoa is 1:1.35 (Ho Son Lam, 2013).
Sampling in this study was random and the sample size was large enough. Therefore, the difference in the male: female ratio of silver mullet in Tran De - Soc Trang compared to other regions may be due to differences in distribution by region, or it may be due to different exploitation methods and habits of male and female fish, resulting in different net entanglement abilities.
4.3.4 Determination of fat content of Sillago sihama
According to IF Pravdin (1973), fatness represents the level of nutritional accumulation of fish, fatness changes according to each stage of body development. Especially it changes according to the development of gonads and has important significance in predicting the time of spawning of fish, the higher the fatness coefficient, the closer the time of spawning.
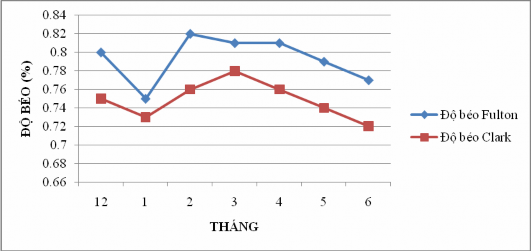
Figure 4.14: Changes in fish fat content over time
The Fulton fat and Clark fat values of fish varied; ranging from 0.75% – 0.82% (Fulton fat) and 0.72% - 0.78% (Clark fat) during the sampling period.
Based on Figure 4.14, it can be seen that from January to March, the fish are mobilizing the nutrients stored in their bodies to create reproductive products in preparation for the upcoming spawning season because during this period, the fat content of the mullet tends to increase (January, Fulton fat content: 0.75, Clark fat content: 0.73, and March, Fulton fat content: 0.81, Clark fat content: 0.78). According to Chung Lan (1969) quoted by Nguyen Van Kiem (2007), in the early stages of egg formation, the level of nutrient accumulation in the body is often higher than the metabolic process to create reproductive products. From April, the fish fat content decreases because at this time, the gonads of the mullet reach maturity. This shows that around April, May, and June may be the spawning season of the mullet.
4.3.5 Sexual maturity coefficient
The sexual maturity index (GSI) of fish in the wild reflects the seasonality of reproduction and is closely related to nutrition and living environment. Each type of fish has its own maturity index and when determining the maturity index, we can determine the fish's reproductive season. (Nguyen Van Trieu et al., 2006).
4.3.5.1 Variation in maturation coefficient of mullet Sillago sihama
Maturity coefficient is one of the indicators to determine the reproductive season and is one of the necessary conditions to recognize the maturity level of reproductive products. (Nguyen Van Trieu et al., 2006).
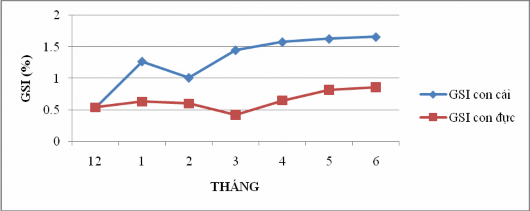
Figure 4.15: Variation in maturity coefficient of Sillago sihama over time
Based on the research results in Figure 4.15, it shows that the average maturity coefficient of sardines is high around March, April, May, June with values of 1.45; 1.58; 1.53; 1.66 respectively for females and April, May, June with values of 0.65; 0.82; 0.86 respectively for males.
According to Nguyen Van Trieu et al. (2006), the highest fish maturity coefficient during the survey was 8.4%. Thus, through the results of research on size and weight over time as well as the results of research on the sexual maturity coefficient of sardines of both sexes, we can predict that the main spawning season of fish is in April, May, June (the beginning of the rainy season every year).
This result is consistent with Nguyen Van Kiem's (1999) assessment of the maturity of most fish species in the Mekong Delta. The author believes that by the end of the dry season, most fish have gonads at stage II or III, and some may even have gonads at stage IV. By early May, most fish gonads have reached maturity, but the fish do not spawn because they do not meet the conditions for spawning. The fish will spawn when it rains.
4.3.5.2 Correlation between HSTT and gonadal development stages
The average maturity coefficients of both female and male fish increased gradually from stages I, II, III and IV with GSI indexes of 0.66%, 0.83% and 3.28% in female fish, and 0.53%, 0.63% and 0.74% for male fish, respectively (Figure 4.16).
According to Xakun and Buskaia (1968), GSI is highest in both sexes when the gonads reach stage IV maturity. This is the stage when reproduction can occur when ecological conditions are suitable. Sexually mature female fish have a GSI of 3.28%, much higher than that of male fish of 0.74%.
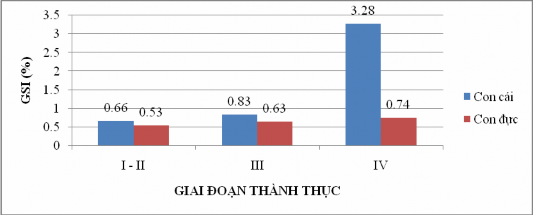
Figure 4.16: Variation of maturation coefficient of Sillago sihama according to gonad development .
Figure 4.16 shows that GSI of both sexes is highest when the gonads reach stage IV, which is consistent with the findings of Xakun and Buskaia (1968).
4.3.6 Relative fertility and absolute fertility
Reproductive capacity depends on the reproductive habits of fish. Species that do not have the habit of protecting eggs and young often have high reproductive capacity and vice versa. (Nguyen Van Kiem, 1999).
Table 4.6: Reproductive performance of Sillago sihama
Target
Average ± Standard | Oscillate | |
Weight (g) | 65.24 ± 26.78 | 22.63 114.28 |
Ovarian mass (g) | 2.2 ± 0.86 | 0.8 6.1 |
Absolute SSS (eggs/female) | 19.659 ± 7887 | 7,143 45,285 |
Relative SSS (eggs/g female fish) | 342 ± 113 | 187 730 |
Based on the recorded results shown in Table 4.6, the absolute fecundity of sardines reached an average value of 19,659 ± 7887 (eggs/female) ranging from (7,143 eggs/female to 45,285 eggs/female) and the relative fecundity reached an average of 342 ± 113 eggs/g female, ranging from (187 - 730 eggs/g female).
Compared with the research results of Ho Son Lam (2013), the reproductive capacity of sardines distributed in Nha Phu - Khanh Hoa has a higher value than the absolute SSS: ranging from
44,333 eggs/female to 120,969 eggs/female; Relative SSS was: 413 ± 152 eggs/g
female fish. This difference may be due to the effect of fish weight on fertility (in Nha Phu - Khanh Hoa, fish weight ranges from 26.62g to 229.48g; in Tran De - Soc Trang, fish weight ranges from 5.71g to 149g).
4.3.6.1 Correlation between weight and absolute fecundity of Sillago sihama
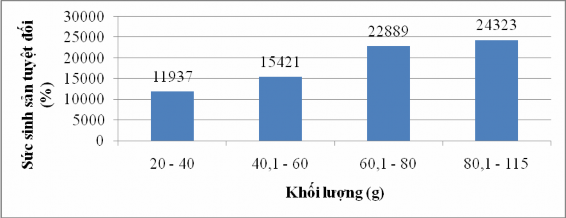 Figure 4.17 Correlation between weight and absolute fecundity of sillago sihama
Figure 4.17 Correlation between weight and absolute fecundity of sillago sihama
Figure 4.17 shows that fish mass and absolute SSS are positively correlated with each other, the larger the mass, the greater the absolute reproductive capacity and vice versa. Based on the figure
4.17 shows that the group of fish with a mass of 20g - 40g has an absolute fecundity of 11937 ± 5769 eggs/female fish; the group of fish with a mass of 40.1g - 60g has an absolute fecundity of 15421 ± 8349 eggs/female fish; the group of fish with a mass of 60.1g - 80g; 80.1g - 115g has an absolute fecundity of 22889 ± 3686 eggs/female fish; 24323 ± 6831 eggs/female fish, respectively. This shows that the larger the mass, the greater the absolute fecundity.
4.3.6.2 Correlation between length and absolute fertility
The correlation between absolute fecundity and length of 45 analyzed mullet samples is shown in Table 4.7.
Table: 4.7 Correlation between length and absolute fertility
Length group (cm)
Absolute fecundity (eggs/female) | |
10 ₋ 15 | 9.778 ± 1.933 |
15.1 – 17 | 13,560 ± 5,234 |
17.1 -21 | 21.872 ± 4.157 |
21.1 – 24 | 22.725 ± 4.029 |
Table 4.7 shows that fish length and absolute fecundity are positively correlated, the longer the length, the higher the absolute fecundity and vice versa. Based on Table 4.7, it shows that the group of fish with a length of 10-15cm has an absolute fecundity of 9,778 ± 1,933 eggs/female fish; the group of fish with a length of 15.1-17cm has an absolute fecundity of
is 13,560 ± 5,234 eggs/female fish; the group of fish with length 17.1 - 21cm has absolute fecundity of 21,872 ± 4,157 eggs/female fish; the group of fish with length 21.1 -24cm has absolute fecundity of 22,725 ± 4,029 eggs/female fish.
Based on the absolute fecundity of 45 analyzed mullet samples, it was shown that mullet are capable of reproduction when they reach a size ranging from 14cm - 23.6cm corresponding to a weight of 7.87g - 149g. The average absolute fecundity of mullet is 19,559 ± 7,887 eggs/female, ranging from 7,143 - 45,285 eggs/female.
Comparing the results of Ho Son Lam (2013), the silver mullet samples used to analyze reproductive capacity ranged in size from 152 to 322 (mm) corresponding to fish weight ranging from 26.62g to 229.48g. The average absolute fecundity of silver mullet was 29,065 ± 5,440 eggs/female, ranging from 44,333 - 120,969 eggs/female. The results showed that the absolute fecundity of silver mullet in Nha Phu lagoon - Khanh Hoa was much higher than the results of this study. The reason is that the silver mullet in this study was shorter in length than the mullet in Ho Son Lam's study (2013), thereby showing that the longer the length, the higher the fecundity.





