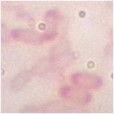
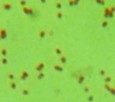
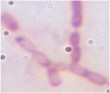
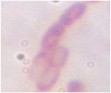
Table 3.5. Morphological and physiological characteristics of 11 selected bacterial strains
STT
Symbol strain | Gram m | Spore | Catalyst | Colony characteristics | Cell morphology | Cell image (x1000) | |
1 | CM2-4 | + | + | + | Atypical spreading, wavy edges, flat surface white inside | Large, individual bacilli |
|
2 | CG2 | - | - | + | Weak growth, flat edges, clear white | Bacillus small, short, round head, standing alone |
|
3 | CG4 | - | - | + | Colonies are round, white, flat-edged, with flat, convex surfaces. cone-shaped | Bacillus small, short, round head, standing alone |
|
4 | CG4-1-2 | + | - | + | Weak growth, ivory white, fringed edges, surface plane | Large, long bacilli |
|
5 | D1-8 | + | + | - | Large, opaque white, convex colonies with smooth surface smooth | Bacillus, chain forming |
|
6 | D1-12 | + | + | - | Large colonies grow widely, opaque white with slightly yellow color, rough surface, fringed edges wash | Bacillus, chain forming |
|
Maybe you are interested!
-
 Morphological Characteristics of K Lines (Winter-Spring Crop 2017-2018 in Gia Lam - Hanoi)
Morphological Characteristics of K Lines (Winter-Spring Crop 2017-2018 in Gia Lam - Hanoi) -
 Characteristics of Business Activities and Business Management Organization at Small and Medium Enterprises in Vietnam
Characteristics of Business Activities and Business Management Organization at Small and Medium Enterprises in Vietnam -
 Characteristics of Activities During the 1000th Anniversary of Thang Long
Characteristics of Activities During the 1000th Anniversary of Thang Long -
 Summary Table of Characteristics of Similar Studies Around the World
Summary Table of Characteristics of Similar Studies Around the World -
 Some characteristics and risk factors of hearing loss in children from 2 to 5 years old in inner-city kindergartens in Hanoi - 1
Some characteristics and risk factors of hearing loss in children from 2 to 5 years old in inner-city kindergartens in Hanoi - 1
T2-11 | + | - | - | Colony atypical form, opaque white surface in the middle, wrinkled and clear edges | Small bacilli |
| |
8 | TM1- 7-1 | + | - | + | Weak growth, round colonies, opaque white, shiny, with defects in the middle | Short, round bacilli |
|
9 | G4 | + | + | _ | Opaque white colonies, convex, smooth surface, large | Bacillus |
|
10 | MP1 | + | - | + | Colonies are round, smooth, convex and pale yellowish white. | Microscopic observation of short rod cells or bridge |
|
11 | MP3 | + | - | + | Colonies are round, smooth, convex and pale yellowish white. | Microscopic observation of short rod cells |
|
7
The results of Gram staining, spore-forming ability and observation of cell morphology and colony morphology showed that 7/11 (63.6%) selected strains, namely CM2-4, CG4-1-2, G4, D1-8, D1-12, T2-11, G4, had many characteristics similar to the genus Bacillus such as gram-positive bacilli, spore-forming and catalase-producing. This result is similar to the published results that Bacillus accounts for a high proportion in the termite gut microbiota [25][6][9]. Bacillus strains often have a high ability to synthesize cellulase and therefore it is understandable that they account for a large proportion of selected strains.
Using 2 types of kits, Kit 20E and Kit 50 CHB, to determine the biochemical characteristics of the selected strains. The results are shown in Appendix 1, Table 6 and Table 7.
After having the characteristics table of the bacterial strains, we continued to analyze the 16S rDNA sequence to identify the selected bacteria.
3.2.2. Naming of termite gut bacteria
Combining the 16S RNA naming method and the biochemical and physiological characteristics of bacteria, based on the reference to the naming document according to Bergey [97]. We have the results of naming the bacterial strains as shown in Table 3.8.
Table 3.6 Results of identification of selected bacterial strains from termite gut
STT
Strain symbol | Naming | ID (API)/Processor Similarity on one's own (%) | Naming method | |
1 | CM2-4 | Bacillus lentus | 99.9 | API 50CHB |
2 | CG2 | Trabulsiella guamensis | 99.63 | 16S rDNA |
3 | CG4 | Klebsiella variicola | 100 | 16S rDNA |
4 | CG4-1-2 | Bacillus firmus | 97.2 | API 50CHB |
5 | T2-11 | Bacillus sonorensis | 100% | 16S rDNA |
No compatibility found copper | API 50CHB | |||
6 | TM1-7-1 | Pseudomonas aeruginosa | 99.9 | 16S rDNA |
7 | G4 | Bacillus subtilis/amyloliquefaciens | 99.7 | API 50CHB |
Bacillus subtilis | 97.14 | 16S rDNA | ||
8 | MP1 | Cellulosimicrobium sp. | 99.93 | 16S rDNA |
9 | MP3 | Cellulosimicrobium funkei | 100 | 16S rDNA |
It can be seen that the isolated bacterial strains are relatively diverse, most of the isolated species in the study have been previously published, such as the publication of Sreeremya et al. who also isolated Pseudomonas fluorescens , Bacillus subtilis , E.coli and Serratia marscens from the gut of higher termites in India [34]. Another publication by Wenzel et al. reported that aerobic culture conditions were applied and 119 cellulolytic bacterial strains were isolated from the gut of the termite Zootermopsis angusticollis , these strains belong to the groups Cellulomonas , Microbacterium , Bacillus , Rhizobium , Pseudomonas [9]. The results of bacterial isolation from termite guts in Nepal also showed that the isolated strains were identified as Bacillus , Cellulomonas and Enterobacter [96].
In Vietnam, according to the publication of Nguyen Thi Thao and colleagues, the number of bacterial species capable of producing cellulase in termite intestines mainly belongs to the Firmicutes phylum (15/24 species), most of which belong to the Clostridia class , Clostridiales order . The most dominant species capable of producing cellulase is Pseudomonas fluorescens of the Pseudomonadaceae order. Of the 18 species producing hemicellulase, the most dominant species is Clostridium thermocellum of the Firmicutes phylum, followed by 3 species of the Bacteroidetes phylum. The bacteria capable of producing cellulase and hemicellulase are estimated to be C. thermocellum , Ruminococus flavefacien and Bacilus subtilis [39].
Bacillus subtilis is a species that has been studied extensively in the world and in Vietnam, and is widely used in practice . B. subtilis has been isolated from termite guts and other sources, such as isolation from silkworm guts published by A.Anand et al. (2021) [98], isolated from termite guts according to the publication of Tarayre C et al. (2014) when studying the termite species Reticulitermes santonesis [99]. With similar results, other authors have also isolated bacteria of the genus Bacillus when isolating on different types of termites such as Zootermopis angusticollis , Reticultermes Hesperus , Coptotermes curvignathus , Reticulitermes speratus [75]. Most of the isolated strains of the genus Bacillus produce Endoglucanase, a few have additional β-glucosidase activity and total FP-cellulase activity.
Studies on the isolation of Klebsiella are still very limited. In 2010, when Cho MJ and colleagues (2010) conducted research on the termite species Reticulitermes speratus, they isolated a cellulase-synthesizing bacterial strain belonging to the species Klebsiella sp. He also demonstrated that they have the ability to synthesize three types of enzymes: cellobiohydrolase, β-glucosidase and Endoglucanase [3]. In another study by Mattéotti C and colleagues (2011), the isolated bacterial strain belonging to the species Klebsiella sp . had β-glucosidase activity [100].
Trabulsiella guamensis , is a member of the genus Gammaproteobacteria in the family Enterobacteriaceae. In 2007, Chou et al. isolated a strain of Trabulsiella odontotermitis [10]. However, there has been no published study on the cellulose-degrading ability of this strain.
In the study of Mark Silby et al. (2011), a bacterial strain belonging to the species Pseudomonas sp was isolated from the gut of the termite Micocerotermes diversus [101]. Pseudomonas is also a genus capable of producing endoglucanase and FP-cellulase in a recent study by Bholay AD, et al. (2014) when studying the isolation of bacteria from termite guts [15]. When isolating bacteria from the gut of termites, Pourramezan selected the strain Pseudomonas aeruginosa for
cellulase activity was highest at pH 7.0 and 30ºC [43]. This is also the species present with the highest proportion in Coptotermes gestroi termites in Vietnam by Do Thi Huyen and colleagues [17].
Cellulosimicrobium has been isolated from termite guts as published by Agapia et al. (2002) and in other publications such as Yon 2007, Liu 2015 and Huang 2012 [102][103][12]. However, studies on cellulose hydrolyzing enzyme systems as well as studies on the collection and application of cellulase from this bacterial species are still very limited.
From the results of the above studies combined with published documents, we found that Bacillus and Cellulosimicrobium are groups present with high probability in termite gut bacteria. These two groups have also been proven to have high cellulase activity and in addition to CMCase activity, they also have other activities that can effectively decompose cellulose. Next, these two groups belong to the Gram (+) bacteria group, so they carry less pathogenic factors than Gram (-) strains. For these reasons, we selected two strains, Bacillus subtilis G4 and Cellulosimicrobium sp MP1, to study cellulase extraction and evaluate the cellulase decomposition potential of the strains.
3.3. Study on cellulase extraction from Bacillus subtilis G4 bacteria
3.3.1. Research on selecting suitable environment and conditions for cellulase biosynthesis process
3.3.1.1. Selection of cellulase biosynthesis medium
2
1.5
1
0.5
0
LB
M1
M2
M6
Biosynthetic environment
CMCase activity (U/ml)
Bacillus subtilis G4 was cultured on LB, M1 M2 and M6 media for 48 hours at 37 C, shaking speed 100 rpm, then CMCase activity was determined on the corresponding media, the results were obtained as shown in Figure 3.3
Figure 3.3 CMCase activity of B. subtlis G4 strain on culture media
The results of Figure 3.3 show that, with different culture environments, the ability of G4 to produce CMCase is also different, in which M6 is the suitable environment.
M6 is a medium containing rice bran, soybean flour and casein, this is a medium in which the ingredients are of natural origin, it can be seen that the G4 strain grows better than the medium in which the ingredients are pure chemicals. M6 medium is also the medium used by Li and his colleagues to cultivate endo-cellulase from Bacillus subtilis YJ1 bacteria[75]. We chose M6 as the medium for cellulase biosynthesis for further studies. On M6 medium, the bacterial strain isolated from termite gut was published by Sreena and his colleagues. with activity from 0.4 U/mg - 1.2 U/mg on medium
unoptimized field [5].
3.3.1.2 Effect of fermentation parameters on CMCase activity
To obtain high activity enzymes on M6 medium, factors such as temperature, shaking speed, fermentation time and pH of the medium were studied. The results are shown in Figure 3.4.
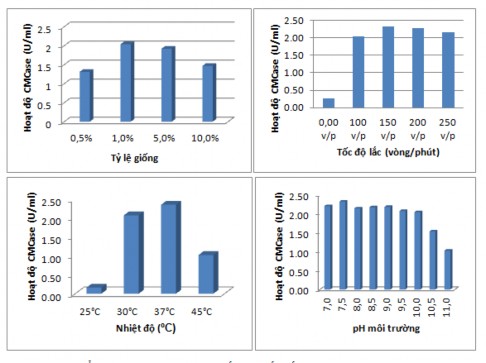
Figure 3.4. Effect of culture parameters on CMCase activity of B. subtilis G4. Seed ratio (A), shaking speed (B), temperature (C) and pH of medium (D).
The above results show that the factors of seed feeding ratio, shaking speed, environmental temperature and environmental pH have a great influence on enzyme activity. By single-factor research method, the results show that the highest enzyme activity is obtained when the seed feeding ratio is 1%, shaking speed is 150 rpm, culture environment temperature is 37ºC, environmental pH is 7.
pH = 7.0 is also the most suitable pH for the enzyme production process of Bacillus sp S3B8 isolated from termite gut [104]. However, the publication of Sreeremya et al. also showed that Bacillus strains isolated from termite gut had the greatest cellulase activity at 40ºC, pH from 9-10 [34]
The enzyme biosynthesis process exists during a period of time with the highest enzyme activity. After this time, enzyme activity will decrease due to physiological factors. Therefore, enzyme collection time is an important factor in the enzyme biosynthesis process.
3.00
2.50
2.00
1.50
1.00
0.50
0.00
24h 48h 72h 96h
Incubation time (hours)
CMCase activity (U/ml)
The G4 bacterial strain was grown under the above selection conditions, varying the time to determine the time with the highest CMCase activity. The results are shown in Figure 3.5.
Figure 3.5 Effect of culture time on CMCase biosynthesis from G4 bacteria
The results of determining the activity at different times showed that the most suitable time for enzyme production from G4 bacteria was 72 hours of culture. For bacteria of the Bacillus species from termite guts and some other isolated sources that have been studied, the most suitable time for enzyme production is from 60 hours to 72 hours [34][105][106].
The enzyme activity obtained from G4 bacteria was also equivalent to the activity of bacteria isolated from termite guts on culture medium after studying the influencing parameters according to the publication of Screena et al., the activity of bacteria ranged from 2.52-6.01 U/mg [25]. However, the cellulase activity from G4 bacteria was much higher than that of Bacillus subtillis bacteria isolated from other sources such as the publication of Reka et al. who isolated Bacillus subtilis strains from saline soil with the highest reaching 0.62 UI/ml [107], another publication of Ariffin et al. also isolated Bacillus pumilus EB3 strain from palm bark with the highest CMCase activity reaching 0.079U/ml [108]. Bacillus subtilis strain isolated from primary forest in India also had the highest CMCase activity of 0.48 U/ml (published by Ramalingam et al., 2014) [109].
3.3.1.3. Effects of nutrient sources on cellulase biosynthesis
In addition to physical factors, changing the nutrient sources in the culture medium also greatly affects the enzyme activity. In this study, the effects of nitrogen and carbon sources and soybean meal, a basic component in the M6 medium, on CMCase activity will be studied. The results are presented in Figure 3.6.
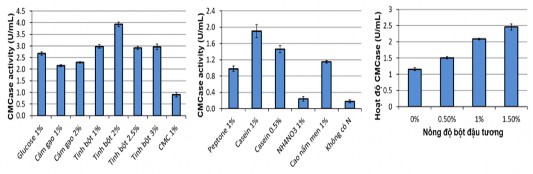
Figure 3.6 . Effects of carbon (A), nitrogen (B) and soybean powder sources on CMCase activity of B. subtilis G4
Results Figure 3.6 (A) show that starch is the best carbon source for cellulase biosynthesis of B. subtilis G4 strain. CMCase activity increased 1.4 times compared to rice bran at the same concentration of 1%. If the starch concentration is further increased from 1% to 2%, CMCase activity continues to increase 1.3 times, however, if the starch concentration is further increased to 2.5% or 3%, CMCase activity decreases. Notably, CMC gives lower CMCase activity than other carbon sources. In the study of Y.Lugani, R.Singla and colleagues or Behera, Mishra (2016), CMC gives higher CMCase activity than other carbon sources for B. licheniformis and Bacillus sp Y3 and B. licheniformis CDB12 [110][107].
This result shows that starch as carbon source has a great influence on the CMCase production process of B. subtilis G4. The positive effect of starch on cellulase biosynthesis of B. subtilis G4 can be explained by the fact that the genes encoding starch and cellulose hydrolyzing enzymes share the same control gene in the G4 bacterial genome.
The results of Figure 3.6 (B) show that Casein is the best nitrogen source and has a great impact on the cellulase production process of strain G4. This result is similar to the publication of Sing et al. 2014, casein is a better nitrogen source than yeast extract and other nitrogen sources for the cellulase production process of Bacillus VITRKHB [111]. However, the study of Abou-Taleb et al. 2009 showed that yeast extract