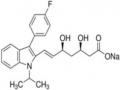
metronidazole sulfate sodium [86], potassium cholesteryl sulfate, potassium testosterone sulfate [115]...( Figure 1.13 ).
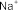





Figure 1.13. Some examples of pharmaceutical substances containing sulfate groups to increase solubility
Some methods of synthesizing sulfate groups:
Using sulfuric acid (H 2 SO 4 ) and derivatives: H 2 SO 4 acid is used to sulfate alkenes through addition reactions and obeys Markovnikov's rule. H 2 SO 4 has the ability to sulfate monohydric alcohols, however, under normal conditions , the reaction tends to eliminate more water. H 2 SO 4 is often used to sulfate polyhydric alcohols, especially cellulose. A derivative of H 2 SO 4 that has the ability to selectively sulfate alcohols is sulfamic acid (H 2 NSO 3 H). However , this acid has a weaker reaction ability and is more expensive than other sulfating agents, and sulfamic acid is also a strong organic decomposing agent [18]. Estrone sulfate is a drug synthesized by this method [45].
Using sulfur trioxide-amine complex: Another agent for synthesizing sulfate groups is the complex of SO 3 with an organic base. This complex has strong activity, sulfating alcohols, phenols, thiols and many other functional groups. The complex of SO 3 with organic bases can be pyridine, trimethylamine, triethylamine or amides such as DMF, ... These complexes are prepared by bubbling SO 3 gas into solutions of organic bases [18]. Some drugs containing sulfate groups are synthesized by this method such as: prednisolone 21-sodium sulfate [78], metronidazole sodium sulfate [86] ...
Using dicyclohexylcarbodiimide (DCC) condensing agent: Although the carbodiimide group is commonly used for the synthesis of arboxylate and pyrophosphate esters, the sulfate reaction using DCC and H 2 SO 4 was first performed by Mumma (1996). Sulfuric acid reacts with an alcohol, in the presence of DCC in a polar solvent such as DMF to produce sulfate ester. This reaction gives good yield under normal conditions, the product is easy to purify and is a simple route for the synthesis of sulfate groups [115]. Some drugs containing sulfate groups are synthesized by this method such as: potassium cholesteryl sulfate, potassium cholesteryl sulfate [115].
This method has not been applied to curcumin molecules.
1.4. RESEARCH ORIENTATION IN THE THESIS
Through the study of five directions of curcumin structural modification ( Figure 1.7 ), directions B, D, C, E only created new derivatives with biological activity, and have not yet created derivatives with improved solubility and bioavailability compared to curcumin. The direction of modification into the diaryl side chain (direction A) is the direction of modification that has received the most research attention and has created derivatives that improve the solubility and bioavailability of curcumin, while having better biological effects than curcumin (see Section 1.2.1 ). On the other hand, when studying chemical methods commonly applied to improve the solubility of pharmaceutical substances ( Section 1.3 ), it was found that the methods all focus on attaching hydrophilic functional groups to the -OH phenol or -OH alcohol positions of the original molecule. This approach has also been applied by some authors to curcumin molecules using techniques such as esterification, phosphatization, hydroxyethylation or conjugation with amino acids. Therefore, the thesis focuses on the synthesis of new molecules containing hydrophilic functional groups: -COOH, -COONa, -SO 3 Na, -OH alcohol, -OPO(OH) 2 ,
-OPO(ONa) 2 , -OSO 3 Na and amino acids by chemically modifying the -OH phenol functional group available in the original curcumin molecule (direction A) or the -OH alcohol functional group created by the curcumin hydroxyethylation process. During the research and implementation of the thesis, conducting a number of direct acylation or esterification reactions (succinate, phosphate, glutarate) on the curcumin molecule did not yield the expected products because the curcumin molecule has many active centers, reacts in many different directions, and the resulting product is less stable. Meanwhile, the direct alkylation reaction of curcumin can produce a more stable product. The hydroxyethyl curcumin derivative produced (containing the OH alcohol functional group) from this alkylation process is used to combine with the above hydrophilic functional groups. The proposed research directions of the thesis are designed as follows:
- Research direction to create new derivatives of curcumin to improve solubility
water by alkylation method to form carboxylic acid derivatives
This research direction is carried out according to the method of Ohtsu et al. (2002) [119], the -OH phenol group in the curcumin molecule is directly alkylated to create a carboxylic acid derivative, then proceed to create sodium salt from the obtained derivative. This direction is expected to obtain new derivatives of curcumin as shown in Figure 1.14 :
Figure 1.14. Modification direction to create new derivatives of curcumin to improve water solubility by alkylation method to create carboxylic acid derivatives
- Research direction to create new derivatives of curcumin to improve solubility
water by alkylation method to create sulfonate derivatives
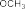
In this research direction, the -OH phenol group in the curcumin molecule is directly alkylated to form a water-soluble sodium sulfonate salt. This direction is carried out according to the method of Marvel et al. (2003) [100] and is expected to obtain new derivatives of curcumin as shown in Figure 1.15 :
Figure 1.15. Modification direction to create new derivatives of curcumin to improve water solubility by alkylation method to create sulfonate derivatives
- Research direction to create new derivatives of curcumin to improve solubility
water via the intermediate monohydroxyethylcurcumin
By converting the -OH phenol group to -OH alcohol group in curcumin molecule by alkylation reaction according to the method of author Changtam et al. (2010), two derivatives monohydroxyethylcurcumin and
dihydroxyethylcurcumin. These two derivatives do not actually have improved solubility than curcumin, however, both derivatives show potential biological activity [33]. These two derivatives contain the alcohol -OH group, which can participate in reactions to form derivatives containing hydrophilic groups more easily than the phenol -OH group in the curcumin molecule. Therefore, a main research direction in the thesis is to use hydroxyethylcurcumin derivatives as intermediates to create new curcumin derivatives with improved water solubility.
The research direction to create new derivatives of curcumin with improved water solubility through monohydroxyethylcurcumin intermediate is expected to obtain the following derivatives ( Figure 1.16 ):
Figure 1.16. Some transformations in the direction of attaching hydrophilic groups through the intermediate
monohydroxyethylcurcumin
- Research direction to create new derivatives of curcumin to improve solubility
water via the intermediate dihydroxyethylcurcumin


The research direction to create new derivatives of curcumin with improved water solubility through the intermediate dihydroxycurcumin is expected to obtain the following derivatives ( Figure 1.17 ):


Figure 1.17. Some transformations in the direction of attaching hydrophilic groups through the intermediate
dihydroxyethylcurcumin
Chapter 2. MATERIALS, EQUIPMENT, CONTENT
AND RESEARCH METHODS
2.1. MATERIALS AND RESEARCH EQUIPMENT
2.1.1. Raw materials
The main raw materials and chemicals are presented in Table 2.1 .
Table 2.1. Raw materials and chemicals used in the study
STT
Name of raw materials and chemicals | Standards, origin | |
1. | Curcumin | 98%, India |
2. | 1,4-Dioxane | AR, China |
3. | 2-Bromoethanol | > 95%, USA (AKSci) |
4. | 2-Chloroethanol | AR, China |
5. | 3-(4,5-Dimethylthiazol-2-yl)-2,5- diphenyl tetrazolium bromide (MTT) | AR, USA (Sigma-Aldrich) |
6. | 2,2-Diphenyl-1-picrylhydrazyl (DPPH) | AR, USA (Sigma-Aldrich) |
7. | Acetone | AR, USA (Sigma-Aldrich) |
8. | Acetone | AR, China |
9. | Acetonitrile | AR, China |
10. | 4-(2-hydroxyethyl)-1- piperazinethansulfonic acid (HEPES) | AR, USA (Sigma-Aldrich) |
11. | Acetic acid | AR, China |
12. | Acetylsalicylic acid (ASA) | AR, USA (Sigma-Aldrich) |
13. | Ascorbic acid | AR, USA (Sigma-Aldrich) |
14. | Boric acid | AR, China |
15. | Hydrochloric acid | AR, China |
16. | Nitric acid 65–68 % | AR, China |
17. | Phosphoric acid | AR, China |
18. | Sulfuric acid | AR, China |
19. | Trichloroacetic acid (TCA) | AR, USA (Sigma-Aldrich) |
20. | Ammonium acetate | AR, China |
21. | Acetic anhydride | > 99.0 %, Spain |
22. | Glutaric anhydride | AR, China |
23. | Succinic anhydride | AR, China |
24. | Carrageenan | AR, USA (Sigma-Aldrich) |
Maybe you are interested!
-
 Semi-synthetic study of some curcumin derivatives to improve water solubility for pharmaceutical applications - 38
Semi-synthetic study of some curcumin derivatives to improve water solubility for pharmaceutical applications - 38 -
 Semi-synthetic study of some curcumin derivatives to improve water solubility for pharmaceutical applications - 48
Semi-synthetic study of some curcumin derivatives to improve water solubility for pharmaceutical applications - 48 -
 Semi-synthetic study of some curcumin derivatives to improve water solubility for pharmaceutical applications - 46
Semi-synthetic study of some curcumin derivatives to improve water solubility for pharmaceutical applications - 46 -
 Semi-synthetic study of some curcumin derivatives to improve water solubility for pharmaceutical applications - 2
Semi-synthetic study of some curcumin derivatives to improve water solubility for pharmaceutical applications - 2 -
 Draw the Frame or General Formula of Each Drug Group (If Any); Structural Formula of Substances in the Chapter.
Draw the Frame or General Formula of Each Drug Group (If Any); Structural Formula of Substances in the Chapter.

STT
Name of raw materials and chemicals | Standards, origin | |
25. | Chloroform | AR, China |
26. | Dichloromethane | AR, China |
27. | Diethyl ether | AR, China |
28. | Dimethyl sulfoxide (DMSO) | AR, China |
29. | Dimethylformamide | AR, Germany (Merck) |
30. | RAW 264.7 cell line | Italy (provided by Prof. Dr. Domenico Delfino, University of Perugia) |
31. | Cancer cell lines: Hela, HepG2, HL-60, K562, MCF7 | Italy (by Prof. Dr. JM PezzTuto, Long-Island University, US and Prof. Jeanette Maier, University of Milan provide) |
32. | Ellipticin | AR, USA (Sigma-Aldrich) |
33. | Ethanol | AR, China |
34. | Ethanol 96 % | Industry, Vietnam |
35. | Ethyl acetate | AR, China |
36. | Ethyl chloroacetate | AR, China |
37. | Ethyl phenylpropiolate (EPP) | AR, USA (Sigma-Aldrich) |
38. | Fetal bovine serum (FBS) | America (Life Technologies) |
39. | Isopropanol | AR, Korea |
40. | Potassium chloride | AR, China |
41. | Potassium bromide | AR, Germany (Merck) |
42. | Potassium carbonate | AR, China |
43. | Potassium dihydrogen phosphate | AR, Germany (Merck) |
44. | Potassium hydroxide | AR, China |
45. | Potassium iodide | AR, China |
46. | N2 gas | 99%, Vietnam |
47. | Lipopolysaccharides (LPS) from Escherichia coli | USA (Sigma-Aldrich) |
48. | Methanol | AR, China |
49. | Methyl chloroacetate | AR, China |
50. | DMEM culture medium | America (Life Technologies) |
51. | N -1-Napthylethylendiamine dihydrochloride | AR, USA (Sigma-Aldrich) |
52. | Sodium acetate | AR, China |
53. | Sodium carbonate | AR, China |
STT
Name of raw materials and chemicals | Standards, origin | |
54. | Sodium chloride | AR, China |
55. | Sodium nitrite | AR, USA (Sigma-Aldrich) |
56. | Sodium bicarbonate | AR, China |
57. | Sodium hydroxide | AR, China |
58. | Sodium phosphate | AR, China |
59. | Sodium sulfate anhydrous | AR, China |
60. | Sodium sulfite | AR, China |
61. | n -octanol | AR, Germany (Merck) |
62. | N G -Methyl- L -arginine acetate ( L -NMMA) | AR, USA (Sigma-Aldrich) |
63. | n -Hexane | AR, China |
64. | Ninhydrin | AR, China |
65. | N,NƟ -Dicyclohexylcarbodiimide (DCC) | AR, Germany (Merck) |
66. | Distilled water | Vietnam |
67. | Phosphoryl chloride | AR, China |
68. | Phosphoryl pentachloride | AR, China |
69. | Pyridine | AR, China |
70. | Silica-gel particle size 0.063-0.2 mm | AR, Merck, Germany |
71. | Sulfanilamide | AR, USA (Sigma-Aldrich) |
72. | Sulforhodamine B (SRB) | AR, USA (Sigma-Aldrich) |
73. | tert -Butoxycarbonyl (Boc) | AR, China |
74. | Triethylamine | AR, China |
75. | p -toluenesulfonic acid monohydrate (TsOH.H 2 O) | AR, Germany (Merck) |
76. | L -Valine | > 99 %, Germany (Merck) |
2.1.2. Equipment and tools
Table 2.2. List of tools and equipment used
STT
Name of tool, equipment, model | Origin | |
1. | Silica-gel F 254 70- 230 mesh | Virtue |
2. | Flask holder 50 mL, 100 mL, 250 mL, 500 mL | Korea |
3. | 1 neck flask 50 mL, 100 mL, 250 mL, 500 mL, 1000 mL | Virtue |
4. | 2-neck flask 100 mL, 250 mL, 500 mL, 2000 mL | Virtue |
5. | 100 mL 3-neck flask | Virtue |