Steam oxidation method, carried out at 580 0 C in a closed furnace chamber containing steam, will create a thin layer of Fe 3 O 4 5-10 µm that is tightly packed and adheres well to the surface of the part, increasing hardness and wear resistance, for example used to manufacture pistons of hydraulic shock absorbers, sprockets, ratchets, etc. This is also a cheap method to seal open pores on the surface of pressed and sintered powder parts.
3.4.3.2.Materials based on Cu powder and Cu alloy
Among non-ferrous metals, Cu and its alloys occupy an important position in the powder metallurgy industry, first of all for the manufacture of self-lubricating sponge silver and filter plates (to be discussed in detail later), about 25% of the output is used in the paint (colorant) manufacturing industry. In the field of machine manufacturing, it only plays a very modest role, mainly for the manufacture of some small machine parts in fields such as telephones, cameras, camcorders, watches, weapons, electrical and electronic equipment and tools, etc.
Pure Cu powder and parts made from pure Cu powder are used in the field of electrical engineering, requiring high electrical conductivity equal to 85-90% of the electrical conductivity of pure Cu made by melting method (with standard resistivity ρ = 1.7241µΩ.cm at 20 0 C). To do this, the powder must have high cleanliness and high pressing force, ensuring the pressed powder has a tightness of 90-95%.
Tin bronze powder is usually used with 10% Sn. If pressed and sintered normally , it is difficult to achieve high density due to blistering phenomenon. To have high density (γ = 8.2 g/cm 3 ) and high plasticity (δ> 15%), it is necessary to use pressing and sintering twice.
Maybe you are interested!
-
 Practice of Determining the Crime of Resisting a Law Enforcement Officer According to the CU Increases the Aggravated Penalty
Practice of Determining the Crime of Resisting a Law Enforcement Officer According to the CU Increases the Aggravated Penalty -
 State management of youth work in Čư M'gar district, Dak Lak province - 15
State management of youth work in Čư M'gar district, Dak Lak province - 15 -
 Current Status of Ecotourism Activities in Cu Chi
Current Status of Ecotourism Activities in Cu Chi -
 State management of youth work in Čư M'gar district, Dak Lak province - 18
State management of youth work in Čư M'gar district, Dak Lak province - 18 -
 Ministry of Education and Training (2014), "Training Materials for Teaching and Testing, Evaluating Learning Outcomes Based on the Orientation of Developing Student Capacity in the Subject"
Ministry of Education and Training (2014), "Training Materials for Teaching and Testing, Evaluating Learning Outcomes Based on the Orientation of Developing Student Capacity in the Subject"
Tin-nickel bronze powder with 5% Ni added to tin bronze can be heat treated after sintering (quenching and aging) to obtain phase hardening, thanks to heat treatment can create mechanical properties in a wide range. Generally, to have a part with high tightness (high density), it is necessary to apply the method of pressing and sintering twice.
Laminate powder, sintering of laminate is difficult because Zn has a strong tendency to evaporate, easily
lost during sintering in hydrogen atmosphere. This must be prevented by placing the part in a sealed container, reducing the H2 flow or, preferably, sintering in an atmosphere rich in Zn vapour.
3.4.3.3. Materials based on aluminum powder and aluminum alloys
Aluminum powder and its alloys are mainly used as colorants in the paint industry. Their use in the form of pressed and sintered powder is very late, because Al powder is too plastic and adheres to the mold wall, sintering is also difficult because Al is very susceptible to oxidation,...
Al alloys have the advantage of being light and resistant to corrosion in air. They can replace Cu alloys and sintered steel when subjected to medium loads (<300 MPa), or replace some plastic parts when working with steel parts.
3.4.3.4. Materials based on Titanium powder and Titanium alloy
Sometimes derived from pure Ti powder, but often from a familiar titanium alloy, for example, Ti-6Al-4V type formed by 700MPa pressing pressure, achieving a not small tightness
more than 95%, sintered at 1200-1300 0 C in a vacuum of 10 -2 Pa, forged at 200MPa pressure at 950 0 C, annealed at 700 0 C for 2h. After doing so, it is possible to achieve a tightness of nearly 100% and high mechanical properties approximately equal to the molten alloy: σ b = 1000-1050 MPa, δ = 10-15%. The first applications were in aircraft manufacturing (screws, rivets, ...), in the chemical and food industries to replace stainless steel, some details in submarines, etc.
3.4.4. Porous and permeable alloys
By powder metallurgy method, it is possible to manufacture parts with high porosity, evenly distributed and these pores are connected to each other and to the surface, they are applied in two fields : self-lubricating porous bearings and filter membranes.
3.4.4.1. Self-lubricating porous bearings
Tin bronze type, porous bush made of tin bronze (10% Sn) with 25% porosity, after being oiled in a vacuum at 70 0 C, can work throughout the machine's life without adding grease. When the shaft rotates, there is friction that heats the bushing, oil will be secreted from the pores, creating good lubrication, the shaft rotates smoothly in a hydrodynamic balance. When the shaft stops rotating, the temperature drops and the oil is sucked back into the pores.
The range of applications is increasingly expanding, from small household appliances (vacuum cleaners, washing machines, refrigerators, blenders, etc.) to the manufacturing industry of motorbikes, cars, etc. because they are cheap, easy to install, work stably, firmly and quietly.
Iron and steel based type to increase load capacity, can use sponge bearing based on Fe, Fe-Cu and add graphite powder (to reduce friction), lubrication can be equivalent to tin bronze type but can withstand higher load, used in car engines, when starting the engine does not get stuck even in freezing weather.
In addition, the bimetal method can be used, on copper-plated low-carbon steel strips (bearing base), a continuous layer of Cu-Pb powder can be coated, then rolled, coated with a layer of coarse Cu powder (grain size 150µm), then sintered at 800 0 C, this porous layer will create good adhesion conditions for the organic resin layer mixed with lead.
The self-lubricating porous silver bearing made of aluminum alloy has the advantage of being light and cheap (compared to tin bronze), but the production cost is higher (because the manufacturing technology is more complicated), the load-bearing capacity is lower, so it is also limited in use. They can replace the bearings based on organic resin.
The type of foam silver that works at high temperatures is foam silver made of Ni-Cr alloy powder, or other heat-resistant metal powders (Mo, W, Co-Mo, ...), used in jet turbine engines, in liquid NaOH pumps of nuclear reactors, ... If working in air, it can withstand temperatures up to 700 0 C, if in a vacuum up to 900 0 C.
3.4.4.2. Filter membranes
The filter membranes are produced from spherical or isoaxial powder, with uniform particle size ( d max / d min ≈ 1.5), low compression pressure, with porosity of 35-40%, evenly distributed pores,
The average pore diameter is about 1/6 d min.
a. Raw materials for making filter membranes
Raw materials can be bronze powder, stainless steel 18.9 series, Ni, precious and rare metals (Ti, Zr, Ta, Ag, Pt) or alloys with high temperature oxidation resistance (25% Cr- 20% Ni steel, inconel, hastelloys) with special manufacturing methods (such as spraying powder solution onto a suitable substrate, extrusion, rolling powder,...) can produce thin-walled tubular and vessel parts with permeability similar to filter paper combined with the heat resistance and rigidity of ceramic and the durability against sudden temperature changes of metal.
Metal felts are manufactured from short cut fibers (diameter 10-150
µm), through light pressing and sintering to obtain a porosity of 40-95% with a pore diameter of 5-200 µm. In a similar way, woven fabrics can also be made from sintered metal fibers. The use of felt or woven fabrics mentioned above has expanded the application range of sintered powder materials. Compared with powder materials, filter membrane materials have advantages, can create very thin filter membranes (up to 0.1 mm), large sheet capacity, high mechanical properties, high bending strength limit, tensile strength limit can reach 200 Mpa, easy to create large working surfaces.
The main disadvantage is cutting (due to expensive materials and complex manufacturing).
b. Uses of foam materials
The main use of sintered porous materials is to make filter membranes to separate solid particles mixed in liquids and gases.
In the field of porous electrodes as the poles of Ni, Ag batteries, porous Ni poles of batteries
ignition. Tantalum chemical capacitors are made from very fine powder (particle size about 2
µm) by reduction of K2TaF2 , or by grinding very clean metal pieces (after hydrogen embrittlement), pressing to 65 % porosity, sintering in vacuum at 1800 0 C for 5 min.
In the medical field, porous sintered materials (stainless steel, Ti and Ti alloys, carbon fiber materials, etc.) are used to implant onto the porous surface of bones. When the bones grow, they adhere well to the porous pores of the material, creating a unified, solid mass (dental implants, bone grafts, etc.).
In the field of turbine engines, people use Co alloys and Ni alloys in
Sintered fiber form for manufacturing gaskets to seal the gap between the rotor and stator.
The silencer in the exhaust pipe of a turbojet engine and the noise barrier in the receiver of a telephone are made of porous material from stainless steel.
3.4.4.3. Friction materials
a. Overview of friction materials
Friction material is a type of material used in the manufacture of friction type joints.
(cone shoes) to transmit motion or to make brake shoes that prevent motion,
In other words, it is used to regulate the power and speed of vehicles and machines. In the past, for this purpose, people often used natural materials such as wood, leather, animal horns, etc., followed by composite materials based on asbestos with binders such as rubber, phenolic resin or cresol resin, etc. Recently, people have realized that asbestos powder is harmful to human health, so they have sought to replace it with other materials. The development of the powder metallurgy industry has opened up a good solution for this type of material.
The requirements for friction materials are to have a stable coefficient of friction during operation, good wear resistance, ensure long life, not wear out the transmission object, good heat resistance, guaranteed heat conduction, and certain mechanical strength. In addition, in each specific case, it must satisfy requirements such as receiving or transmitting smooth motion, ensuring stable transmission with little vibration, good corrosion resistance, and stability when the weather changes (temperature, humidity of the working environment, etc.). To satisfy the above requirements, friction materials are often heterogeneous materials (multi-component), including components with mutually supportive properties (similar to composite materials): binders and additives (fillers). The binder is often a metal powder with good sintering ability such as Cu, Fe powder or their alloys. Additives to adjust friction properties are divided into two families with opposite properties: those that reduce friction and are prone to slipping, such as graphite, metals with low melting points such as Pb, Bi, Cd or metal sulfides and sulfates, and those that increase friction are usually hard particles such as oxides, carbides, silicates or phosphites, etc. A very important component, almost indispensable for friction-resistant materials, is graphite powder. In addition to the effect of reducing friction and causing slipping, graphite also contains components of metal powder, oxides, carbides, silicates, metal phosphites, etc. This cushioning layer has the effect of transmitting firmly and stably, reducing vibration and causing less wear on working surfaces, especially important when transmitting
high power and speed
b. Friction materials based on copper powder
This is a friction material widely used in the manufacture of clutch pads, brake pads for shock absorbers, medium and large capacity vehicles. The binder is Cu powder and tin bronze powder.
The friction (transmission) objects of this alloy are gray cast iron, carbon steel, low alloy steel in a well-chemical state or without heat treatment, used in the fields of tractors, excavators, road construction machines, trains, cranes, compressors, crawler vehicles, etc. Note that in the case of wet friction (with oil), the transmission torque also depends on the groove configuration on the working surface of the cone plate.
c. Friction materials based on iron powder
This is a friction material used in dry (oil-free) transmissions and is
used in most industrial fields, because it is cheaper, has a higher coefficient of friction and guaranteed wear resistance .
The friction object of this alloy with medium load is pearlite gray cast iron, carbon steel with minimum tensile strength of 500MPa. With larger load, low alloy cast steel or well-tempered steel must be used. With high load, hard chrome-plated steel must be used.
d. Friction materials with high content of non-metallic components
This is a type of friction material used for high-power transmissions (heavy loads). The binder is still Cu powder, Fe powder, or Ni, Mo powder. Commonly used non-metallic components are metal oxides (Al 2 O 3 , SiO 2 , MgO, TiO 2 ), mineral powders (mullite, sillimanite, silicate). To adjust the friction properties, graphite, low-melting metals (Pb, Bi, Cd, ...), sulfite, carbide, phosphite or intermetallic phases are often added (such as Laves phase in Co-Mo-Si, Co-Mo-Cr-Si alloy systems, they have high hardness, good wear resistance). The proportion of non-metallic particles can account for more than 50% of the volume of the material.
In addition, carbon-carbon fiber composite materials mixed with ceramic hard particles are also used as brake pads for high-capacity vehicles and machines.
This type of friction material is used in high-power (heavy-load) transmissions, for example as brake pads for aircraft landing gear, clutch pads, brake pads for excavators, heavy trucks and military vehicles (such as brake pads for tanks, tracked vehicles, etc.), as well as other heavy machinery . [2]
Review questions
Question 1. Describe the characteristics of pure aluminum.
Question 2. Describe how to classify aluminum alloys and how to symbolize aluminum alloys. Question 3. Describe the characteristics of pure copper.
Question 4. Describe how to classify copper alloys and how to symbolize copper alloys. Question 5. What are powder materials and the characteristics of powder materials.
Question 6. Describe the types of hard alloys and their symbols, and give some illustrative examples.
CHAPTER 4
INORGANIC MATERIALS (CERAMIC)
Inorganic materials are made up of chemical compounds of metallic elements combined with non-metallic elements, or of non-metallic elements combined with each other.
About 75% of the elements in the periodic table of chemical elements participate in the formation of inorganic materials. The main elements and the possibilities of their combinations to form inorganic materials are shown in Figure 4.1.
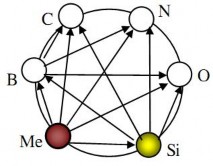
Figure 4.1. Ability to bond to form inorganic materials
According to the diagram, a metal can combine with boron to form boride, with carbon to form carbide, with nitrogen to form nitride, with oxygen to form oxide, with silicon to form silicide. The same is true when starting from silicon or boron.
The above combination makes inorganic materials very rich and diverse in chemical composition and properties. Common chemical compounds in inorganic materials are: single metal oxides (for example, Al2O3 in corundum ceramics), single semi-metallic oxides (for example, SiO2 in quartz glass), mixtures of many metal oxides (for example, porcelain, silicate glass), non-metallic elements (for example, boron, carbon), carbides, nitrides of metals and semi-metals (for example, TiC, SiC, BN, ZrN), ...
Depending on the purpose, inorganic materials can be classified in many different ways: by chemical composition, structure, technological method, field of use, etc. In this book, inorganic materials are divided according to their combination characteristics and presented in 3 main groups:
- Ceramics and refractories.
- Glass and glass ceramics.
- Cement and concrete.
To distinguish from other large groups of materials (metallic materials, organic materials). Currently, people often use the concept of ceramic materials to refer to all types of non-metallic inorganic materials including all three groups of inorganic materials mentioned above.
Inorganic materials are a large group of materials, within the scope of this chapter only mentioned
to the basic issues and its main representatives.
4.1. Structure of inorganic materials
Inorganic materials include many different types, but share common characteristics in structure and properties. These characteristics are clearly different from the structure and properties of metals and organic materials.
4.1.1. Atomic bonds in inorganic materials
Inorganic materials are made up of metallic and non-metallic elements, that is, of atoms with different chemical natures, for example, different atomic sizes, electron shell structures, coordination numbers, polarizability, binding forces, etc. Therefore, compared to metals, inorganic materials are often determined by the radius ratio between cations and anions. Based on this geometric correlation, atoms are arranged in a specific coordination shape with a constant coordination number. In inorganic material systems made up of many different types of atoms (cations), due to differences in the sizes of cations, rearrangements of anions can occur and lead to the formation of other coordination shapes and coordination numbers. In other words, in inorganic materials, a cation can, together with anion, form many different types of structural units with different coordination numbers.
The most important structural characteristic of inorganic materials is the type of bonding between the atoms that make them up. Inorganic materials there is no metallic bonding, but a combination of ionic and covalent bonding.
Due to the characteristics of ionic-covalent complex bonds, the bonding energy in inorganic materials is relatively large, ranging from 100 - 500 Kj.mol -1 (for metals, it is 60 - 250 Kj.mol -1) .
The characteristics of the covalent ion-covalent complex bond decisively influence a number of characteristic properties of inorganic materials such as high melting point, high density, hardness, brittleness, transparency and high electrical insulation.
Inorganic materials are composed of atoms with different chemical natures, so the polarization and deformation factors in the material's bonds are significant. In particular, cations with large charges and anions with large sizes will skew the electron distribution and increase the proportion of covalent bonds. Polarization and ion deformation have a profound effect on the properties of the material, especially the elasticity, mechanical strength, hardness and color of the material.
4.1.2. Crystalline and amorphous states
Inorganic materials can exist in different structural states: crystalline (e.g., SiC ceramics, single-oxide ceramics), amorphous (e.g., glassy materials), or both crystalline and amorphous (e.g., glass-ceramics).
The following will explore some structural features of the crystalline state and the solid state.
amorphous in common inorganic materials.
a. Crystalline state in inorganic materials
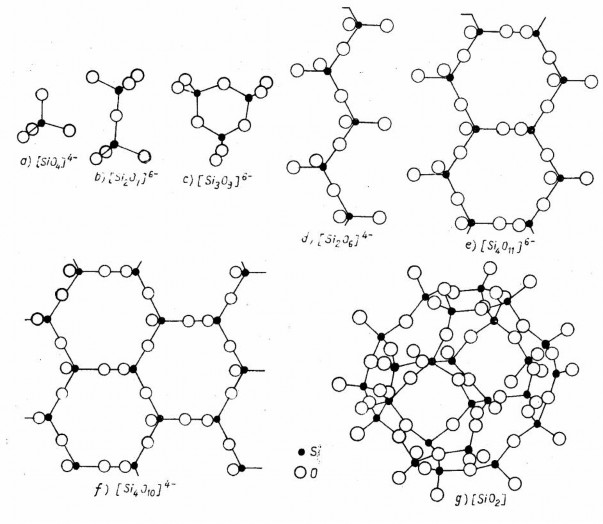
Figure 4.2. Silicate structural forms
a) Island structure; b) cluster structure; c) ring structure (e.g., triple ring); d) single-chain structure; e) double-chain structure; f) layer structure; g) skeleton structure
The crystal lattice of most inorganic materials can be considered approximately as a lattice of ions, in which cations and anions occupy the positions of the lattice nodes. But because there is always a certain proportion of covalent bonds, there is adjustment and rearrangement in the lattice, the anions X surrounding the cation Me create a coordination polyhedron of the form MeXn . The index n is the coordination number depending on the ratio of the radii between the ions ( r Me / r x ), with a value from 3 to 12 depending on the type of coordination polyhedron.
Among the inorganic oxide group of materials, the most common are the tetrahedral coordination polyhedra MeO 4 ( n = 4) and the octahedral MeO 6 ( n = 6). Other forms include MeO 3, MeO 5, MeO 8, and MeO 12 .
These polyhedra have anionic character and are called complex anions, for example [BO 3 ] 3- ,
[BO 3 ] 5- , [PO 4 ] 3- , [SiO 4 ] 4- . The coordination polyhedra are linked together to form the crystal lattice of the material. They can be linked together through the vertices, through the edges or through the faces of the coordination polyhedra. The stability of the lattice will be greatest when the polyhedra are connected through the vertices,





