AMINO ACID, HEMOGLOBIN, NUCLEOTID ACID METABOLISM AND PROTEIN BIOSYNTHESIS
LEARNING OBJECTIVES: After completing this lesson, students will be able to
1. Correctly describe the process of digestion and absorption of protein.
2. Correctly describe the process of protein degradation.
Maybe you are interested!
-
 Questions: Circle the Correct Answer to the Following Questions
Questions: Circle the Correct Answer to the Following Questions -
 Protein Docking Simulation with 60 Ligands and Evidence
Protein Docking Simulation with 60 Ligands and Evidence -
 Presentation of Inventory Accounting Information on Financial Statements
Presentation of Inventory Accounting Information on Financial Statements -
 Requirements to Ensure Correct Application of Criminal Law on the Crime of Using Computer Networks, Telecommunications Networks, and Electronic Means to Commit Acts of Appropriation
Requirements to Ensure Correct Application of Criminal Law on the Crime of Using Computer Networks, Telecommunications Networks, and Electronic Means to Commit Acts of Appropriation -
 Steps to Create a Presentation Using Storymap Tool on Arcgis Online Platform
Steps to Create a Presentation Using Storymap Tool on Arcgis Online Platform
3. Correctly describe the process of protein synthesis.
4. Correctly describe the process of Hemoglobin degradation.
5. Correctly describe protein metabolism disorders.
CONTENT
1. General diagram:
Transform (copy) Decode (translate)
Self-replicating DNA RNA Protein (1) (2)
2. Body needs:
- 1 gram protein/1kg/1 day


- Synthesis of structural and functional proteins: Actin, myosin, collagen...
• Is the main constituent of protoplasm, an important component in building organelles, plasma membranes... the diverse structure of proteins determines all characteristics, morphology, and anatomy of the body:
• Create enzymes that catalyze biochemical reactions. About 3,500 types of enzymes are now known. Each type participates in a specific reaction.
• Creates hormones that regulate metabolism in cells and the body.
• Form antibodies, which protect the body against disease-causing bacteria.
• Participates in the motor function of cells and the body.
• Protein breakdown creates energy for the vital activities of cells and the body.
In short, proteins perform many functions related to all life activities of cells, regulating the characteristics of living organisms.
- Biological protein synthesis: Enzymes, some hormones Factors involved in protein synthesis:
Includes the following 5 elements:
1/ The role of DNA
2/ mRNA (messenger RNA) 3/ tRNA (transfer RNA) 4/ rRNA (ribosomal RNA)
5/ Opening, extending and closing elements
1/ Origin:
Exogenous: Animal protein, plant protein, microorganisms, yeast
Endogenous: Cellular proteins are hydrolyzed into amino acids called endogenous amino acids called catepsins, which have properties and mechanisms similar to peptidase enzymes. These catepsins are located in lysosomes. When the cell dies and the lysosomal membrane ruptures, they are released and act on proteins, causing autolysis.
3. Digestion and absorption:
- Occurs in the digestive tract thanks to digestive enzymes
- Enzymes that hydrolyze polypeptides or proteins are called peptidases or proteinases.
2 types:
+ Endopeptidase: hydrolyzes in the Pepsin and Trypsin chains
+ Exopeptidase: hydrolyzes the two ends of the chain
- Exopeptidase: hydrolyzes the two ends of the chain Carboxypeptidase, Aminopeptidase
3.1. In the stomach
- Pepsin (pH 2-3) requires an acidic environment, thanks to the secretion of HCl by parietal cells.
- Ability to digest collagen (connective tissue)
- Protein digestion in the stomach accounts for ~ 15%, producing proteoses, peptones and polypeptides.
3.2. In the small intestine:
- The resulting products are poured into the duodenum, stimulating the small intestine to release cholecystokinin, stimulating the pancreas to secrete proenzymes, the proenzymes follow the ducts in the pancreas into the small intestine and are activated into active forms.
Pancreas secretion
- Bicarbonate: neutralizes gastric acid
- Trypsinogen
- Chymotrypsinogen
- Procarboxypeptidase
- Proelastase
- These are inactive forms that have the effect of protecting the pancreas itself. At the same time, there is always a trypsin inhibitor in the pancreas.
- When proenzymes are poured into the small intestine, they are activated into active forms that help further hydrolyze proteins.
Small intestine excretes:
- Aminopeptidase
- Dipeptidase
- Globular protein (animal) is completely hydrolyzed
- Partially digested fibrous proteins (collagen, keratin)
- Cereal protein is incompletely digested (due to the cellulose layer).
4. ABSORPTION: AA is absorbed into the capillary network of the small intestine, then to the portal vein and from there to the liver and other tissues.
5. Protein degradation:
5.1 Intracellular protein degradation:
- Cell proteins are constantly renewed.
5.2 Deamination
- Separate the amino group from the amino acid in the form of NH 3
5.3 Amination
- Is the reaction of synthesizing amino acids (aa) from α ketonic acid and NH 3
- Amino exchange reaction:
Usually occurs in the heart, liver, brain, and kidneys. There is no release of NH 3 , but only the transfer of an amino group from this aa to an α ketonic acid to form a new aa, and the substance becomes another α ketonic acid that enters the Kreds cycle.
6. Final product of aa transformation:
6.1 . NH3
6.2 GLUTAMIN
Nature:
- Non-toxic, non-charged, so easy to transport through membranes.
- Is the main NH 4+ transporter /plasma
- Source of amino groups for amino acid synthesis
- In the liver, NH 4+ released from glutamine will be synthesized into urea and excreted in the urine.
- In the kidney, glutamine hydrolyzes to NH 4+ , creating ammonium salts and excreting them.
urine, helps excrete H + acid-base balance
- Glutamine hydrolysis/renal increase in acidosis
APPLICATION : Increased NH 3 in the blood causes:
- Reduce blood pH
- Nerve cell damage:
NH3 crosses the blood-brain barrier, leading to reduced glutamate to form glutamine, causing a decrease in brain α ketoglutarate, leading to a decrease in oxaloacetate, affecting the Krebs cycle, lack of energy for the brain, and cell damage.
Increases glutamine in the brain, reduces glutamate reserves (glu needed to create neurotransmitters such as ϒ-aminobutyrate (GABA).
Increased brain glutamine, changes in glial cell osmolytes, causing cerebral edema.
- Severe liver cell failure: end-stage cirrhosis, poisoning, viral infection causing acute necrosis...
- Connect main door
- Genetic deficiency of urea cycle enzymes.
Clinical applications:
Increased blood urea:
Outside the kidney: Reduced blood flow to the kidney, reduced glomerular filtration (congestive heart failure, dehydration, salt loss, blood loss...). Urinary system obstruction due to stones, prostate tumors. Increased protein degradation: trauma, burns, fever...
In the kidney: Acute glomerulonephritis, acute tubular nephritis (poisoning, blood transfusion error)... Chronic nephritis
Reduce blood urea:
- High glucose infusion. Diabetes. Severe liver damage.
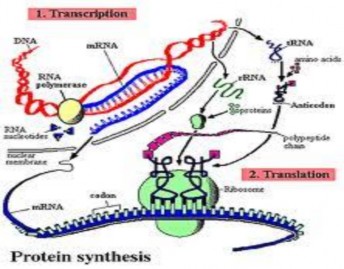
7. Protein synthesis process: Includes 2 stages:
Stage 1: RNA synthesis to transfer genetic information from genes to protein products (see RNA synthesis)
Stage 2: Protein synthesis in the cytoplasm consists of 4 basic steps
+ Step 1: Activation of amino acids. Free amino acids in the cytoplasm are activated by binding to the energy-rich compound adenosine triphosphate (ATP) under the action of certain enzymes. Then, thanks to another specific enzyme, the activated amino acid re-binds to the corresponding tRNA to form an amino acid-tRNA complex (aa-tRNA) .
+ Step 2: The polypeptide chain begins with the participation of the ribosome, the AUG start triplet, the starting amino acid tRNA enters the ribosome, its anticodon matches the start codon on the mRNA according to NTBS. End of the initiation phase
+ Step 3: The polypeptide chain is elongated, the tRNA transporting the first amino acid enters the ribosome, its anticodon matches the start codon of the mRNA according to the complementary principle. aa 1 - tRNA moves to the adjacent position, its anticodon matches the code of the first amino acid on the mRNA according to the complementary principle. The enzyme catalyzes the formation of a peptide bond between the start amino acid and the first amino acid. The ribosome moves one triplet on the mRNA (translocation) causing the start tRNA to leave the ribosome. Next, aa 2 - tRNA enters the ribosome, its anticodon matches the code of the second amino acid on the mRNA according to the complementary principle.
A peptide bond is formed between aa1 and aa2. Translocation occurs again, and so on until the ribosome contacts a triplet adjacent to the terminator, and the polypeptide chain now has the structure
aaMD – aa 1 – aa 2 ... aan is still attached to the nth amino acid tRNA.
+ Step 4: At the end of the polypeptide chain, the Ribosome moves to the termination codon, at this time the translation process stops, the two subunits of the ribosome separate to form tRNA, the last amino acid is separated from the polypeptide chain. Another enzyme removes the starting amino acid, releasing the polypeptide chain.
It should be noted that on each mRNA at the same time, many ribosomes can slide past at a distance of 51Å ® 102Å. This means that on each mRNA, many proteins of the same type can be synthesized.
Protein synthesis helps ensure that proteins perform the function of expressing traits and providing raw materials to form organelles and perform many different functions.
8. Hemoglobin metabolism
8.1 Heme synthesis
+ Globin synthesis
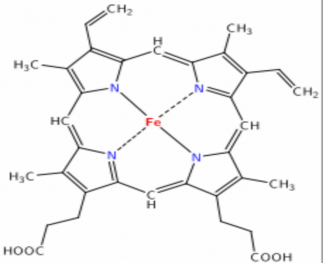
▪Hemoglobin is a porphyrinoprotein consisting of 2 components:
• The pure protein part is called Globin, made up of 4 polypeptide chains including two α chains and two β chains.
•The exogroup part is Hem, which is formed by the protoporphyrin IX nucleus and Fe 2+ ion.
GLOBIN
8.2 Hemoglobin degeneration:
8.2.1 Digestion: Hemoglobin or myoglobin of food when entering the intestine under the effect of digestive enzymes is hydrolyzed into two parts: Hem and globin
+ Heme is oxidized into hematin, which is difficult to absorb, so most of it is excreted in the feces, a small part is transformed under the influence of intestinal bacteria.
+ Globin is hydrolyzed into peptides and amino acids, and is further metabolized through the protein metabolism pathway.
8.2.2 Hemoglobin Degeneration:
The average life span of a red blood cell is about 120 days. After red blood cells die, they mainly occur in the reticuloendothelial system, i.e. in the liver, bone marrow, and especially the spleen. Hemoglobin is released at a rate of about 8 to 9 grams per day.
+ Hemoglobin degradation diagram: includes all parts below 8.2.2.1 Biliverdin production
8.2.2.2 Creating free bilirubin: yellow, insoluble in water (2 propionate radicals create intramolecular H-bonds)
Free: not combined with glucuronic acid Neurotoxic
Slow diazo reaction → indirect bilirubin 250-300 mg produced/day
Source:
- 85% from the destruction of old red blood cells, 15% from ineffective erythropoiesis, metabolism of heme-containing proteins (myoglobin, cytochrome, peroxidase) transported in the blood by albumin. At the liver cell membrane, bilirubin separates from the complex with albumin, is actively transported into the cell
- Excess: → extravascular tissue: jaundice; brain: brain damage. 8.2.2.3 Creation of conjugated bilirubin:
+ 85% diglucuronate, 15% monoglucoronate, water soluble, non-toxic, fast diazo reaction → direct bilirubin.
- Diazo reaction (Van Den Bergh reaction)
- Bilirubin transformation in the intestine
- Hemoglobin metabolism disorders