independent of pH (except Cr (VI)). Normally, an increase in pH will promote the photoreduction of metal ions. However, photogenerated electrons not only participate in the photoreduction reaction of metal ions but they can also reduce protons, water and dissolved oxygen depending on specific environmental conditions.
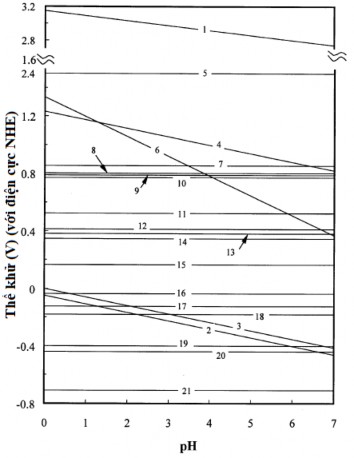
1-E VB , 2-E CB , 3- 𝜑 𝐻 ∗ ⁄ 𝐻 2 , 4- 𝜑 𝑂 2 ⁄ 𝐻 2 𝑂 , 5- 𝜑 𝐴𝑢 3+ ⁄𝐴𝑢 , 6- 𝜑 𝐶𝑟 6+ ⁄𝐶𝑟 3+ , 7- 𝜑 𝐻𝑔 2+ ⁄𝐻𝑔 ,
2
Maybe you are interested!
-
 Study on synthesis of TiO2- Fe2O3/GNP materials from ilmenite and graphite ore to orient Cr(VI) transformation in defense industrial wastewater - 6
Study on synthesis of TiO2- Fe2O3/GNP materials from ilmenite and graphite ore to orient Cr(VI) transformation in defense industrial wastewater - 6 -
 Study on synthesis of TiO2- Fe2O3/GNP materials from ilmenite and graphite ore to orient Cr(VI) transformation in defense industrial wastewater - 2
Study on synthesis of TiO2- Fe2O3/GNP materials from ilmenite and graphite ore to orient Cr(VI) transformation in defense industrial wastewater - 2 -
 The impact of social capital on business performance - a case study of the Textile and Garment industry in the Southern region of Vietnam - 36
The impact of social capital on business performance - a case study of the Textile and Garment industry in the Southern region of Vietnam - 36 -
 Testing the credit risk tolerance of Vietnamese commercial banks - Case study of Vietnam Joint Stock Commercial Bank for Industry and Trade - 21
Testing the credit risk tolerance of Vietnamese commercial banks - Case study of Vietnam Joint Stock Commercial Bank for Industry and Trade - 21 -
 Study on synthesis and characterization of MoS2/rGO catalyst modified with Mn and its application for photodegradation of rhodamine B in visible light region - 23
Study on synthesis and characterization of MoS2/rGO catalyst modified with Mn and its application for photodegradation of rhodamine B in visible light region - 23
8- 𝜑 𝐴𝑔 + ⁄𝐴𝑔 , 9- 𝜑 𝐻𝑔 2+ ⁄𝐻𝑔 , 10- 𝜑 𝐹𝑒 3+ ⁄𝐹𝑒 2+ , 11- 𝜑 𝐶𝑢 + ⁄𝐶𝑢 , 12- 𝜑 𝐻𝑔𝐶𝑙 2 ⁄ 𝐻𝑔 ,
4
13- 𝜑 𝐻𝑔𝐶𝑙 2− ⁄𝐻𝑔 , 14- 𝜑 𝐶𝑢 2+ ⁄𝐶𝑢 , 15- 𝜑 𝐶𝑢 2+ ⁄𝐶𝑢 + , 16- 𝜑 𝐹𝑒 3+ ⁄𝐹𝑒 , 17- 𝜑 𝑃𝑏 2+ ⁄𝑃𝑏 ,
18- 𝜑 𝑁𝑖 2+ ⁄𝑁𝑖 , 19- 𝜑 𝐶𝑑 2+ ⁄𝐶𝑑 , 20- 𝜑 𝐹𝑒 2+ ⁄𝐹𝑒 , 21- 𝜑 𝐶𝑟 3+ ⁄𝐶𝑟
Figure 1.5. Positions of the conduction and valence bands of TiO 2 (anatase) compared with the reduction potentials of metal ions at different pH values [29].
As shown in Figure 1.5, the photoreduction of Cr(III), Cd(II) and Fe(II) does not occur at any pH because the reduction potential of these ions is lower than the reduction potential.
photoreduction of photogenerated electrons, while the photoreduction of other metal ions such as Au(III), Cr(VI), Hg(II), Ag(I), Hg(I), Fe(III), Cu(I) and Cu(II) is thermodynamically feasible. Among these metal ions, Cr(VI) and Fe(III) can only be reduced to Cr(III) and Fe(II), respectively, while in the case of Pb(II) and Ni(II), depending on the pH, the process may or may not be thermodynamically feasible [29].
The reaction diagram of the photoreduction reaction of TiO 2 metal is as follows Step 1: Initiation
ℎ𝑣≥𝐸 𝐵𝐺
𝑇𝑖𝑂 2 → 𝑇𝑖𝑂 2
(ℎ +
+ 𝑒
− ) (1.1)
Step 2: Oxidation of organic compounds and water
𝑂𝐻 − +ℎ + → 𝐻𝑂 . (1.2)
𝑂𝐻 . +𝑅𝐻 → 𝑅 . + 𝐻 2 𝑂 → 𝐶𝑂 2 + 𝑎𝑥𝑡 𝑘ℎ𝑜á𝑛𝑔 (1.3)
2𝐻 2 𝑂 + 4ℎ + → 𝑂 2 + 4𝐻 + (1.4) Step 3: Reduction process of metal ions
𝑀 𝑛+ +𝑒 − → 𝑀 (𝑛−1)+ (1.5)
There are several parameters that affect the photoreduction process of metal ions such as: initial metal ion concentration, amount of catalyst used, presence of electron or hole consumers, light intensity.
a. Effect of initial metal ion concentration
The photocatalytic reaction kinetics are quite different from conventional advanced oxidation reactions (AOPs) and often follow the saturation effect. The initial rate of the photocatalytic reaction follows the Langmuir–Hinshelwood mechanism with respect to the initial concentration of the metal ion.
𝑟 0
= 𝑘𝐾𝐶 0
1+𝐾𝐶 0
(1.6)
According to this formula, at high concentrations (KC 0 >>1), the reaction rate is highest and is described as zero-order rate, in contrast to dilute solutions (KC 0 <<1), the reaction rate is first-order.
b. Effect of pH
Another important variable is pH, since pH determines the morphology of both the photocatalyst surface and the reduced metal ion, which further affects the photocatalytic reaction efficiency.
The general trend when evaluating the efficiency of metal ion photoreduction reaction with pH variation is that at low pH, the efficiency of photoreduction reaction is usually low and when pH increases in the acidic environment range, the efficiency of photoreduction reaction will increase, but when pH continues to increase, the efficiency of photoreduction reaction will decrease.
In aqueous environments, low pH provides a large amount of H + ions , which can react with the TiOH ( hydrated TiO 2 ) surface to form Ti . OH 2 + . The protonated Ti . OH 2 + molecules will have more difficulty releasing electrons, although most metal ions exist in forms that are easily reduced in large quantities, low photoreduction efficiency is often observed. It can be seen that the number of electrons plays a prominent role in the photoreduction process.
As the pH increases in the acidic range, the H + concentration decreases, which may increase the TiOH generation. This may increase the electron release and the metal ions are found as reduced species, so the photodegradation may increase significantly.
Finally, the number of OH groups may exceed at high pH, which may cause the negatively charged surface of TiO 2 to form TiO - . This may slow down the electron release process. On the other hand, metal ions often precipitate with hydroxide ions to form M(OH) n (M is the metal ion, n is the valence). Both of these situations have a negative effect on the photoreduction process.
Due to many different factors, the optimal pH value also depends a lot on the nature of each individual metal ion. The optimal pH value for Ag is usually from
6 - 7, with Cr(VI) is pH = 2 [83], with Cu(II) is from 2 - 3.5 [113] and with Hg(II) is pH = 4 - 4.5 [97].
c. Effect of photocatalytic material content
The initial rate of the photocatalytic reaction is proportional to the amount of photocatalytic material used. However, when the concentration increases to an optimum value, the reaction rate becomes independent of the amount of photocatalyst.
This optimum concentration depends largely on factors such as light intensity, reactor geometry, photocatalyst particle size, and phase composition. At the optimum concentration, it can be assumed that the entire surface of the photocatalyst is illuminated by the incident light source. When the catalyst concentration exceeds the optimum concentration, a shielding effect occurs between the catalyst particles, preventing light from reaching the hidden catalyst particles, making it impossible for these hidden particles to exhibit their photocatalytic activity, thus the photocatalytic activity does not increase although increasing the photocatalyst concentration increases the photocatalyst surface area per unit volume of the solution. Chen and Ray proposed a formula to explain the effect of photocatalyst concentration on the photocatalytic reaction rate.
𝑟 𝑖 = 𝑘[1 − 𝑒 −𝜀𝛽𝐶 𝑥ú𝑐 𝑡á𝑐 𝐻 ] (1.7)
Where k is the reaction rate constant, C is the catalyst concentration, the light absorption coefficient of the reaction system is Ɛ, β is a power related to the light intensity (its value lies between 0.5 and 1) and H is the thickness in the direction of light transmission [97].
c. Effects of electron and hole acceptors
In photocatalytic reactions, electrons participate in the reduction reaction while holes participate in the oxidation reaction. The use of an electron acceptor (usually dissolved oxygen) is necessary in the case of oxidation reactions. The rate of photooxidation of phenol in the presence of dissolved oxygen has been
described by Chen and Ray [97], this speed also follows the Langmuir–Hinshelwood formula:
𝑘 =
𝑘𝐾 𝑂 2 𝑝 𝑂 2 1+𝐾 𝑂 2 𝑝 𝑂 2
(1.8)
Where k is the measured kinetic constant of organic matter decomposition, 𝐾 𝑂 2 is the adsorption constant of dissolved oxygen on the surface of the photocatalyst, 𝑝 𝑂 2 is the partial pressure of dissolved oxygen.
Meanwhile, the photoreduction process requires a hole agent (usually formic acid, ethanol, methanol, EDTA, citric acid…) to participate in the reaction system during the reduction process. Usually oxygen is replaced by circulating or continuous streams of nitrogen or other inert gases [97].
d. Effect of light intensity
With the increase of light intensity, the photon flux (the number of incident photons per unit time per unit area) increases, which enhances the generation of electron-hole pairs. Therefore, at low light intensities, the photocatalytic reaction follows a first-order rate with light intensity, while at high intensities, the photocatalytic reaction rate is proportional to the square root of the intensity. According to Herrmann, at higher intensities (25 mWm -2 ) the rate of electron-hole pair formation exceeds the reaction rate, leading to the promotion of electron-hole recombination [97].
e. Effect of illumination time
The reaction time is determined by the exposure time between light and the photocatalyst and the photogenerated electrons of these substances with the reduced metal ions. The general trend observed is that the efficiency of the photoreduction reaction is enhanced as the reaction time is increased. But when the reaction time is increased beyond the optimum time, the photoreduction reaction is usually independent of the reaction time.
At the beginning and during the following time period, the contact between light and the photocatalyst becomes more efficient and results in the generation of a larger number of electrons. Then, the extended time period allows more efficient contact between the available electrons and the reduced metal ions. This can significantly enhance the photoreduction reaction. At one point, the photoreduction reaction reaches its maximum level indicating that the optimum time has been reached. When the reaction time is extended beyond the optimum time, a very large amount of product is formed which will prevent the contact between the reactants. Therefore, the photocatalyst will be hindered from releasing more electrons, which will not enhance the photoreduction reaction. Another reason is that the reduced ions in the solution have been completely reduced, leaving no ions in the solution. The optimum reaction time was determined to be approximately 5 h for Ag(I) ion, 4 h for Cr(VI) ion, 3 h for Cu(II) ion and 50–150 min for Hg(II) ion[113].
f. Method of identifying oxidizing agents in photocatalysis
The photocatalytic reaction takes place according to the radical mechanism with the formation of free radicals e - , h + , then the appearance of ∙ OH, O 2 - . The free radical ∙ OH is a strong oxidizing agent that allows non-selective mineralization of organic compounds. According to author Buxton [28], to identify the free radical ∙ OH, butanol-1 can be used as an agent. When butanol-1 is involved, the rate of conversion of organic compounds is greatly reduced because butanol-1 easily reduces the ∙ OH agents . When studying the photocatalytic mechanism of Cr(VI) and RhB, author Xue Bai [24] also used free radical quenchers such as: sodium ethylene diamine tetra acetic (EDTA-Na) to quench h + radicals , dimethyl sulfoxide (DMSO) to quench e - radicals , benzoquinone (BQ) to quench • OH radicals, isopropyl alcohol to quench • O 2 - radicals. The results showed that when using free radical quenchers, the efficiency of the photocatalytic reaction was significantly reduced.
1.3. Conclusion of chapter 1
Above are some research projects that delve into the use of graphene-based composite materials for environmental treatment. Thereby, it can be seen that the application of materials based on graphene and metal oxides is attracting a lot of attention from domestic researchers. This is a new research direction because most of the projects are from 2016 onwards, the number of publications is very diverse, in which the direction of treating heavy metals is the most focused direction. Composite materials all demonstrate much better adsorption capacity and photocatalytic activity in treating pollutants than the component materials when used individually.
Graphene used in research is mainly GO and RGO which are synthesized directly from graphite by Hummers method or improved Hummers method using strong oxidizing agents to create GO which is then reduced with reducing agents to obtain RGO. GO and RGO are widely used in research because these graphene derivatives can form strong bonds with metal oxides thanks to the oxygen-containing functional groups on the surface. However, the use of strong oxidizing agents causes defects on the graphene surface and thus reduces the properties of graphene such as reducing surface area, conductivity, and electron transfer ability. On the other hand, the synthesis process of graphene through GO has to go through many steps, is complicated, and uses strong oxidizing agents, which limits the practical application of the material. Graphene nanoplatelets (or graphene nanoplates) is a graphene material with a large surface area, few surface defects, high electrical conductivity, simple chemical fabrication, and can be applied on an industrial scale.
In addition, most studies are focusing on using adsorption processes to treat heavy metals, while the treatment of heavy metals by advanced oxidation methods, specifically the photocatalytic method, has received almost no attention. Advanced oxidation is an advanced treatment method that has proven to be very effective in treating inorganic and organic pollutants. For heavy metals, the advanced oxidation process can
Reduced chemical use, non-toxic catalysts, low secondary waste emissions, high reusability. For wastewater from the production of defense explosives, this is also a suitable method because it can simultaneously treat both heavy metals and organic substances that are difficult to decompose in it.
Composite materials based on graphene and semiconductor metal oxides have many advantages over the use of individual materials, because they can take advantage of the heavy metal adsorption capacity, electrical conductivity and electron transmission capacity of graphene, thereby significantly improving the photocatalytic effect of composite materials.
Through the overview of materials based on graphene and metal oxides, characteristics of heavy metal contaminated wastewater from industrial explosives production and generalizing the advantages and disadvantages of heavy metal treatment methods, the research direction of the thesis topic was selected to focus on the synthesis of materials based on graphene and TiO 2 , applied in the photocatalytic process of heavy metal treatment. The selected graphene precursor is graphene nanoplatelets (or graphene nanoplates) synthesized directly from natural graphite by chemical methods. The precursor to manufacture TiO 2 is from 52% Binh Dinh ilmenite concentrate. The target for treatment is Cr(VI) ion in defense production wastewater.





