CHLORHEXIDINE GLUCONATE
Recipe:
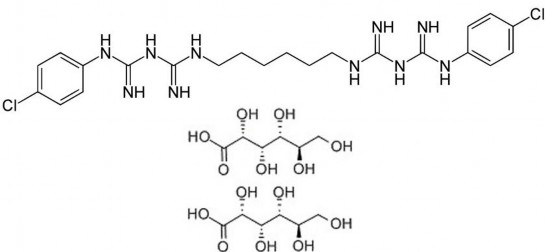
C 22 H 30 Cl 2 N 10 .2C 6 H 12 O 7 ptl: 897.77
Scientific name: 1,6-di(4'-chlorophenyl diguanidino)hexane di D-gluconate
Preparation:
Reflux a mixture of hexamethylene bis[dicyandiamide], [NCNHC (:NH):NH- (CH 2 ) 3 ] 2 and p-chloroaniline hydrochloride in 2-ethoxyethanol, at 130 – 140 o C for 2 hours. Neutralize with D-gluconic acid.
The preparation is in the form of a 5% or 20% solution, colorless or slightly yellow; very miscible with water, miscible with 5 volumes of alcohol, 3 volumes of acetone.
Nature
Chlorhexidine gluconate is not a quaternary ammonium salt, but due to the strong alkalinity of the two guanidine groups, at near physiological pH it will ionize and exist in the form of dications, with chemical properties and surface activity similar to cationic surfactants, and antibacterial effects by the same mechanism.
Uses
Broad spectrum of activity on two grams of bacteria, except for fast alcohol-resistant strains, fungal spores and viruses. Does not penetrate the skin and mucous membranes, so it is not toxic. The residue left on the surface where it is applied remains effective for 1-2 days. The activity is not affected by body fluids, but is lost when exposed to anionic detergents, complex ions such as phosphates, carbonates, silicates. Soap does not reduce the effect.
Assign
Commonly used in surgery. 4% solution for skin disinfection, cleaning instruments before surgery (better than povidone-iodine or hexachlorophene solution, may be inferior to povidone-iodine on some strains of gram-negative bacteria)
150
1% solution for erratic infections. Also widely used for wound cleansing, dressings, burn treatment, mouthwash, bathing infants and other hygiene tasks.
Alcoholic solutions are more potent than aqueous solutions, for example a 0.5% alcohol solution is as potent as a 4% aqueous solution.
Note: Do not use on eye wounds, do not put medicine in ears. Some people may be sensitive to this antiseptic.
151
LESSON 19. DRUGS TO KILL TRICHOMONAS AND AMIB
LEARNING OBJECTIVES
After completing this lesson, students will be able to:
1. Describe commonly used drugs to treat Trichomonas and amoeba, including drug names and structural formulas.
2. Describe the physical and chemical properties and their application in testing; instructions for use of each drug.
GENERAL CONTENT
Diseases caused by amoebic parasites or protozoans such as Trichomonas, Giardia... account for a fairly large proportion in tropical countries. Women are more susceptible to diseases caused by parasites in the genital tract than men. Statistics show that Trichomonas is the most common cause of vaginal and vulvar infections after Candida albicans.
These antiparasitic drugs also have many other uses in the treatment of genital infections caused by anaerobic bacteria as well as in combination with antibiotics (usually Clarithromycin) and proton pump inhibitors (usually Lansoprazole) to treat Helicobacter pylori.

SOME COMMON DRUGS
1. 5-Nitroimidazole derivative
1.1 General structure
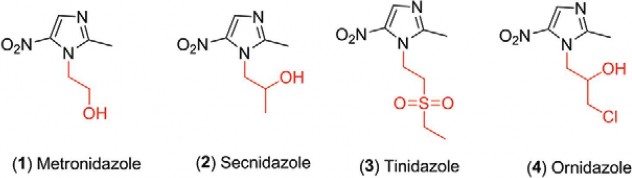
152
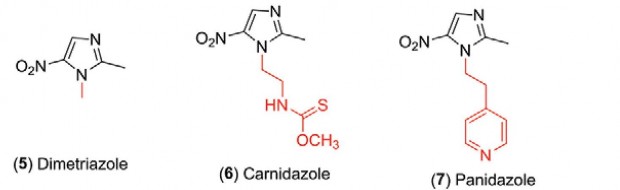
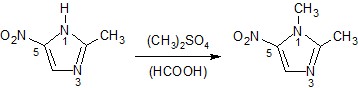
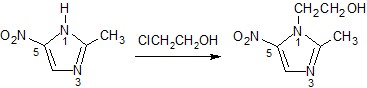
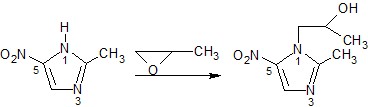
1.2 Preparation of some substances
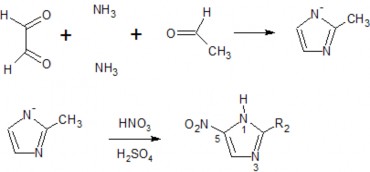
Dimetridazole
Metronidazole
Ornidazole
153
Tinidazole
1.3 Physical and chemical properties
DCI
Nomenclature | Recipe - M | Solubility | Flow | UV-capacitive lip | |
Carnidazole | N-[2-(2-methyl- 5-nitro-1H- imidazol-1- yl)ethyl thiocarbamate | C 8 H 12 N 4 O 3 S 244.3 | 142.5 (EtOH) | ||
Dimetridazole | 1,2-dimethyl-5- nitro-1H- imidazole | C5H7N3O2 141.1 | Soluble/alcohol Slightly soluble/water, ether | 138-142 (Water) | 276nm (HCl 0.1M) |
Metronidazole | 2-(2-methyl-5- nitro-1H- imidazol-1- y)ethan-1-ol | C6H9N3O3 171.2 | Water (1), EtOH (0.5), CHCl3, ether (0.05) | 159-163 | 277nm (HCl 0.1M) |
Nimorazol | 4-[2-(nitro-1H- imidazol-1- y)ethyl]morpholi n | C9H14N4O3 226.2 | Slightly soluble in water, soluble in alcohol. acetone | 110-111 (water) | |
Ornidazole | 1-chloro-3-(2-methyl-5-nitro- 1H-imidazole-1- | C 7 H 10 ClN 3 O 3 219.6 | 77-78 (Toluene) | 289-312nm (Propanol) |
Maybe you are interested!
-
 Describe the Differences Between Antipsychotic and Antidepressant Drugs, the Efficacy and Common Side Effects of Each Drug.
Describe the Differences Between Antipsychotic and Antidepressant Drugs, the Efficacy and Common Side Effects of Each Drug. -
 Comparison of the effects of two methods of complete intravenous anesthesia with Propofol with and without target concentration control - 2
Comparison of the effects of two methods of complete intravenous anesthesia with Propofol with and without target concentration control - 2 -
 Study on botanical characteristics, chemical composition and acetylcholinesterase enzyme inhibitory effects of two species Piper thomsonii (C. DC.) Hook. f. var. thomsonii and Piper hymenophyllum Miq., Piperaceae family - 24
Study on botanical characteristics, chemical composition and acetylcholinesterase enzyme inhibitory effects of two species Piper thomsonii (C. DC.) Hook. f. var. thomsonii and Piper hymenophyllum Miq., Piperaceae family - 24 -
 Effects of Factors on Reproductive Performance of Dvn1 and Dvn2 Sows
Effects of Factors on Reproductive Performance of Dvn1 and Dvn2 Sows -
 Study on chemical composition, toxicity and some biological effects supporting the treatment of gastric and duodenal ulcers of Sanchezia nobilis Hook.F. leaves - 2
Study on chemical composition, toxicity and some biological effects supporting the treatment of gastric and duodenal ulcers of Sanchezia nobilis Hook.F. leaves - 2
154
y)propan-2-olSecnidazole | 1-(2-methyl-5- nitro-1H- imidazol-1- y)propan-2-ol | C 7 H 11 N 3 O 3 185.2 | 76 (Toluene) | ||
Tinidazole | 1-[2- (ethylsulfonyl)et hyl]2-methyl-5- nitro-1H- imidazole | C 13 H 17 N 3 O 2 247.3 | Soluble/water , benzen | 125,128 | 310nm (MeOH) |
1.4 Inspection
Qualitative
- Flow measurement
- UV spectrum
- Color reactions
Quantitative
Dry medium in acetic acid and titrate with 0.1N HClO 4
1.5 Mechanism of action
- Because these derivatives have strong reducing properties, they participate in the oxidation-reduction process in the metabolism of anaerobic bacteria such as Bacteroides fragilis.
- Nitroimidazole derivatives form free radicals, affecting the multi-enzyme system, thereby interfering with the replication process and breaking the DNA chain of unicellular organisms, especially protozoa containing a lot of adenine and thymine such as Trichomonas and Entamoeba.
1.6 Treatment indications
1.6.1 Unicellular
- Trichomonas vaginalis in women (causing vaginitis and urinary tract infections), dose 2g/day x 10 days for Metronidazole and single dose 2g for Secnidazole and Tinidazole.
- Lamblia (flagellates): 2g/day x 5 days (Metronidazole) and 1-2g single dose (remaining derivatives)
- Entamoeba (Amoeba in intestine and liver): 2g single dose in adults and 30mg/kg in children.
1.6.2 Anaerobic bacteria
- 1.5-2g/day for oral use or intravenous solution.
- Prevention of anaerobic infections after surgery.
1.6.3 Treatment of Helicobacter pylori
155
Combined with Clarythromycin (500mg/day) + Lansoprazole (30mg/day) + Tinidazole (1g/day) x 7 days.
1.7 Side effects - contraindications Hives, headache, dizziness, numbness... Stool and urine may be reddish brown
Contraindicated in sensitive patients, pregnant women. Note: there is antabuse phenomenon, so avoid using when drinking alcohol.
1.8 Structural relationship and pharmacodynamic effects
- NO2 group is required
- Substituent at N position is necessary
- Position number 2 can be methyl or other substituents such as phenyl, imidazolidinendione, carbamate, oxygen-containing groups.
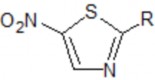
2. Thiazole derivatives
2.1 General structure
5-nitrothiazole derivative
R
Nomenclature | DCI | |
NHCOMe | N-(5-nitrothiazol-2-yl) acetamide | Ternidazole |
NHCOH | N-(5-nitrothiazol-2-yl) formamide | Nimorazol |
N-(5-nitrothiazol-2-yl) thiophen-2-carboxamide | Panidazole | |
| N-(5-nitrothiazol-2-yl) imidazolidin-2-one | Nitridazole |
2.2 Treatment indications
Nitridazole: urinary tract and digestive system schistosomiasis caused by Schistosoma haematobium 25mg/kg/day divided into 2 doses morning and evening x 5-10 days.
Tetonitrozole: has good effects on Trichomonas and Candida albicans, so it should be used in cases of vulvovaginal infections caused by these two agents at a dose of 200mg, 2-3 times/day x 3-10 days.
Aminitrozol: used to treat Trichomonas vaginalis.
156
3. Emetine and derivatives
3.1 Structure
Emetine Dehydroemetine
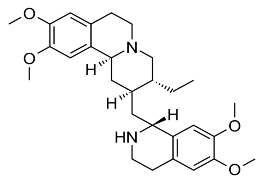
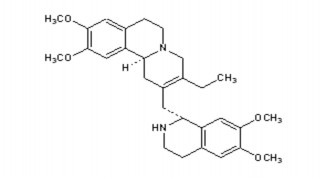
These are alkaloids extracted from the Uragoga ipecacuanha / Rubiaceae plant that have been used to treat amoeba since the 40s and 50s in many countries around the world.
3.2 Properties
Because of the 2N structure, these substances are dibases and can form 2 salts with strong acids such as HCl and HBr. These salts are soluble in water and alcohol, while the base form is soluble in organic solvents.
3.3 Inspection
Qualitative:
- UV spectrum: emetine dihydrochloride absorbs at 230 and 280nm.
- IR spectrum.
Quantitative : dry environment
3.4 Treatment indications
Today, emetine has been completely replaced by dihydroemetine and 5-nitroimidazoles. These agents can be used alternately to treat intestinal amoeba in severe cases and amoebic liver abscesses.
3.5 Side effects
Diarrhea, nausea, vomiting, pain at injection site, tachycardia, hypotension, myocarditis, neuralgia, myalgia…
4. Paromomycin
4.1 Structure
It is an aminosid antibiotic, in addition to antibacterial activity, Paromomycin also has special activity against some protozoa extracted from many Streptomyces species, especially S.rimous, var.paromomycinus. Paromomycin is also known as hydroxymycin, aminosidine...
157