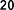
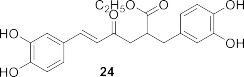
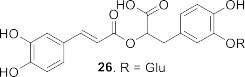


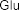



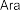


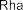
Figure 1.6. Phenolic compounds isolated from the genus Sanchezia
By mass spectrometry and high-performance liquid chromatography, Juliana Mourao Ravasi et al. [107] isolated compounds from different parts of S. oblonga grown in Brazil, including many polyphenolic compounds such as 1- O -coumaroyl-2-hydroxy propanal ( 21 ), 1- O -coumaroyl-2- O -arabinopyranosyl ( 22 ), 1- O -coumaroyl-2- O -rhamnopyranosyl propanal ( 23 ), ethyl rosmarinate ( 24 ), 4- O -
arabinopyranosyl butyl sinapate ( 25 ), rosmarinic acid-3′- O -glucopyranoside ( 26 ), 4- hydroxy-3-methoxybenzyl ( 27 ), caffeic acid glucopyranoside ( 28 ), benzyl alcohol-7- O -arabinopyranosyl ( 29 ), dihydrosinapic acid- O -glucopyranoside ( 30 ), 4- O -galloyl- sinapyl alcohol diacetate ( 31 ), sinapic acid- O -glucopyranoside ( 32 ), and 4- O -glupyranosyl-ethyl-dihydrosinapat ( 33 ).
In addition, some publications in the world and in Vietnam have shown that the aboveground part of the genus Sanchezia contains many acids and glycosides. Accordingly, in 2013, Ahmed E. Abd Ellah and colleagues isolated 5 alcohol compounds from the methanol extract of the aboveground part of S. speciosa including [22]: (1-octen-3-ol) (34) , 3- O - β -glucopyranosyl-1-octen-3-ol (35) , 3- O - β -glucopyranosyl-(1→6)- β -
glucopyranosyl-1-octen-3-ol (36) , 3- O - β -arabinopyranosyl-(1→6)- β -
glucopyranosyl-1-octen-3-ol (37) , 3- O - β -arabinopyranosyl-(1→6)- β -glucopyranosyl-(1→6)- β -glucopyranosyl-1-octen-3-ol (38) . Also from the methnol extract of leaves and roots of S. speciosa , Ahmed E and colleagues isolated two compounds: 9- O - β -glucopyranosyl- trans -cinnamyl alcohol ( 39 ) , 9- O - β -xylopyranosyl- (1→6 )- O - β -glucopyranosyl- trans -cinnamyl alcohol (40) .
According to the study of Juliana Mourao Ravasi et al. [107], using mass spectrometry and high-performance liquid chromatography-mass spectrometry, a number of fatty acids were identified from different parts of S. oblonga grown in Brazil, including organic acids: ethyl octadecanoate ( 41 ), stearic acid ( 42 ), oleic acid ( 43 ), ethyl linoleate ( 44 ), 9,12-octadecadienal ( 45 ), linoleic acid ( 46 ), ethyl palmitate.
( 47 ), palmitic acid ( 48 ), nonadecylic acid ( 49 ).
In Vietnam, according to the study of author Le Thi Hong Nhung in 2018 [ 1], from the n- hexane fraction of S. speciosa leaves using chromatography-mass spectrometry (GC-MS), 14 fatty acids were identified including stearic acid ( 42 ), oleic acid ( 43 ), palmitic acid ( 48 ), nonadecylic acid ( 49 ), lauric acid ( 50 ), myristic acid ( 51 ),
pentadecylic acid ( 52 ), margaric acid ( 53 ), arachidic acid ( 54 ), eicosenoic acid (5 5 ),
vaccenic acid ( 56 ), palmitoleic acid ( 57 ), linoleic acid ( 58 ), α -linolenic acid ( 59 ).
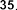

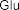

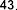
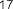
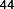
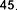





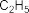
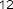
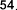
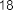
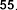
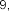
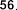
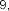
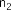
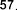
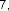


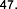




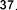



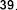

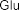


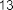


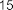
Figure 1.7. Organic acid and glycoside compounds isolated from the genus Sanchezia

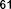
In addition to the above groups of compounds, according to some publications, the Sanchezia genus also contains a number of other terpene compounds. Accordingly, in 2017, Nusrat Shaheen et al. [155] determined from the dichloromethane extract from the root part of the S. Speciosa genus to be (+)- 3,13-clerodadien-16,15-olid-18-oic acid (60) , stigmasterol 3- O - -D-glucopyranoside (61) .

Figure 1.8. Terpene compounds isolated from the genus Sanchezia
Also according to the study of Juliana Mourao Ravasi and colleagues [107] showed that from different parts of S. oblonga species grown in Brazil, there are stigmasta-4,22- dien-3-one ( 62 ), sitosterol ( 63 ), stigmast-4-en-3-one ( 64 ) , campesterol ( 65 ).
In addition, some other publications indicate that the Sanchezia genus also contains several other groups of compounds. In 2017, Nusrat Shaheen [155] isolated an alkaloid compound from the dichloromethane extract of the roots of S. speciosa , which was 3-methyl-1H-benzoindole-4,9- dione ( 66) . Similarly, in 2019, Vu Duc Loi and colleagues [183] isolated several chemical compounds from the ethyl acetate extract of S. speciosa leaves , which were 3-methyl-1H-benzoindole-4,9- dione ( 66) , scopoletin ( 67 ), 3′- O -methyl-3,4- methylenedioxy ellagic acid ( 68 ). From the methanol extract of leaves and roots of S. nobilis , Ahmed E and colleagues isolated a neolignan glucoside compound: 6,7,8-trimethoxy-cumarin (69) .
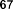

Figure 1.9. Other compounds isolated from the genus Sanchezia
Table 1. 2. Published chemical composition of Sanchezia genus in the world and Vietnam
STT
Year of publication | Author | Species/Distribution/BPD | The determining substances | TLTK number | |
In the world | |||||
1 | 2013 2014 | Ahmed E. Abd Ellah, Khaled M. Mohamed, Enaam Y. Backheet, Mahmoud H. Mohamed, | Sanchezia nobilis (Egypt) Roots, leaves, above ground parts | 2, 3, 4, 15, 16, 17, 18, 34, 35, 36, 37, 38, 39, 40 - 5 matsuke alcohol compounds - 3 flavonoid compounds - 2 cinamyl alcohol compounds - 4 phenolic compounds | [22], [213] |
2 | 2015 | Parvin S., Abu Shuaib Rafshanjani, | Sanchezia speciosa (Bangladesh) Leaf | Qualitative | [218] |
Maybe you are interested!
-
![Study on the structure and biological activity of some compounds isolated from the halong palm (Livistona halongensis TH Nguyen & Kiew) and gotu kola [Centella asiatica(Linn.) Urban] - 2](data:image/svg+xml,%3Csvg%20xmlns=%22http://www.w3.org/2000/svg%22%20viewBox=%220%200%2075%2075%22%3E%3C/svg%3E) Study on the structure and biological activity of some compounds isolated from the halong palm (Livistona halongensis TH Nguyen & Kiew) and gotu kola [Centella asiatica(Linn.) Urban] - 2
Study on the structure and biological activity of some compounds isolated from the halong palm (Livistona halongensis TH Nguyen & Kiew) and gotu kola [Centella asiatica(Linn.) Urban] - 2 -
 Study on the ability to decompose petroleum hydrocarbons of some strains of photosynthetic purple bacteria forming biofilms isolated in Vietnam - 2
Study on the ability to decompose petroleum hydrocarbons of some strains of photosynthetic purple bacteria forming biofilms isolated in Vietnam - 2 -
![Benzopyrano Ring of Flavonoid Compounds [31]](data:image/svg+xml,%3Csvg%20xmlns=%22http://www.w3.org/2000/svg%22%20viewBox=%220%200%2075%2075%22%3E%3C/svg%3E) Benzopyrano Ring of Flavonoid Compounds [31]
Benzopyrano Ring of Flavonoid Compounds [31] -
 Distribution of Families, Genus, and Species of Medicinal Plants in Pine Plantations at Kvnc
Distribution of Families, Genus, and Species of Medicinal Plants in Pine Plantations at Kvnc -
 Rate of infection and carrying genes resistant to 3rd generation Cephalosporin and Quinolone of Klebsiella strains causing respiratory infections isolated at the National Children's Hospital, 2009 - 2010 - 16
Rate of infection and carrying genes resistant to 3rd generation Cephalosporin and Quinolone of Klebsiella strains causing respiratory infections isolated at the National Children's Hospital, 2009 - 2010 - 16

Year of publication | Author | Species/Distribution/BPD | The determining substances | TLTK number | |
Kader T. Sharmin, | |||||
4 | 2015 | Seline Omondi JC Onyango, | Sanchezia speciosa University Botanical Garden Maseno, Kenya | Qualitative | [57] |
5 | 2017 | Nusrat shaheen, Muhammad Uzair, Bashir Ahmad, Alamgeer, | Sanchezia speciosa (Pakistan) Root, flower, | 1, 19, 20, 60, 61 - 1 flavonoid compound - 2 phenolic compounds - 2 terpene compounds - 1 alkaloid compound | [155] |
6 | 2020 | Juliana Mourão Ravasi, Giuseppina Negri, Antonio Salatino, Maria Luiza Faria Salatino, et al | Sanchezia oblonga (Brazil) Whole plant | 5-8; 21-33, 41-49, 62- 65 - 4 flavonoid compounds - 13 phenolic compounds - 9 fatty acid compounds - 4 terpene compounds | [107] |
7 | 2022 | Progga Paramita Paul, Pritam Kundu, Utpal Kumar Karmakar, | Sanchezia nobilis (Bangladesh) | Qualitative | [161] |
In Vietnam | |||||
8 | 2016 | Bui Thanh Tung, Vu Duc Loi, Nguyen Thanh Hai, Nguyen Tien Basin, | Sanchezia speciosa (Tuyen Quang) Leaves | 9, 10, 61,66 - 2 flavonoid compounds - 1 terpene compound - 1 alkaloid compound | [29] |
9 | 2017 | Nguyen Tien Vung, Vu Duc Loi, Nguyen Thi Mai | Sanchezia speciosa (Sanchez Light) Leaves | Qualitative | [4] |
10 | 2017 | Loi Vu Duc, Tung Bui Thanh, Ha Vu Hoang, | Sanchezia speciosa (Sanchez Quang) | 9, 10, 61,66 - 2 flavonoid compounds - 1 terpene compound - 1 alkaloid compound | [63] |
STT
Year of publication | Author | Species/Distribution/BPD | The determining substances | TLTK number | |
Tuyen Nguyen Manh, | Leaf | ||||
11 | 2018 | Le Thi Hong Nhung, | Sanchezia speciosa Leaf | 41, 42, 43, 47, 50-59 - 14 fatty acid compounds | [1] |
12 | 2019 | Vu Duc Loi, Tran Minh Ngoc, Bui Thi Xuan, | Sanchezia nobilis (Nam Dinh) Leaves | 11, 12, 13, 67, 68 - 3 flavonoid compounds; 2 other compounds | [183] |
STT
Thus, the species studied for chemical composition of the genus Sanchezia are S. speciosa, S. nobilis and S. oblonga, however these three species names are identified as synonyms [220], therefore the thesis only synthesizes the chemical composition according to the genus, not according to the species. The most common groups of compounds in the genus are flavonoids, terpenes and alkaloids. However, the number of isolated and structurally determined compounds is still limited, so continuing to isolate and determine the structure of the compounds will provide more information about the chemical composition of the plant, as well as contribute to explaining the biological effects of the plant.
1.3. Biological effects of Sanchezia genus
1.3.1. Acute toxicity
Previous studies on the acute toxicity of the genus Sanchezia focused mainly on n- hexane and ethyl acetate extracts of leaves and roots, methanol extracts of bark, wood, leaves and roots of S. speciosa using the test method on brine shrimp larvae [51], [ 218] . The analytical results showed that the n- hexane and ethyl acetate extracts from S. speciosa leaves showed increased mortality with increasing concentrations. The LC50 values of n - hexane and ethyl acetate extracts were found to be 19.95 µg/mL and 12.88 µg/mL compared to the positive control vincristine sulphate which had a significant LC50 value of 10.96 µg/mL. Therefore, the ethyl acetate fraction was more toxic than the n- hexane fraction on brine shrimp larvae but was safer than vincristine sulfate [218].
Nusrat Shaheen et al. (2017) conducted acute toxicity tests from dichloromethane and methanol extracts of S. speciosa bark, wood, leaves and roots grown in Multan on shrimp larvae at different dose levels. The results showed that the lethality
Different toxic effects were observed when exposed to different test doses and mortality was found to be proportional to the concentration of the tested extract. Among them, the dichloromethane extract of the root exhibited significant toxic effects with an IC 50 of 2.52 µg/mL compared to the control etoposide with an IC 50 of 7.46 µg/mL [ 51 ].
1.3.2. Anti-inflammatory effect
The methanol extract of S. speciosa bark and root was tested by Nurat haseen [155] for anti-inflammatory effects on two models, carrageenan-induced mouse paw edema and cotton-pellet-induced mouse paw edema. At a dose of 50 mg/kg, there was no anti-inflammatory effect on the carrageenan-induced mouse paw edema model, but it showed a mild anti-inflammatory effect on the cotton-pellet model. At doses of 100 mg/kg (52.79% reduction in edema) and 200 mg/kg (68.75% reduction in edema) after 3 hours, both showed statistically significant anti-inflammatory effects compared to the control group and were close to the effect of indomethacin at a dose of 5 mg/kg (76.34% reduction in edema) on the carrageenan-induced mouse paw edema model . Similar results were also obtained in the cotton-pellet model. The indomethacin group at a dose of 5 mg/kg reduced inflammation by 65.35%, while the methanol extract of S. speciosa bark and root at a dose of 100 mg/kg reduced inflammation by 46.12% and the dose of 200 mg/kg reduced inflammation by 59.32%.
In Vietnam, according to the study of Vu Duc Loi and colleagues in 2016 [63], from the ethyl acetate fraction of ethanol extract of cultivated S. speciosa leaves , 4 compounds were isolated, the compounds were evaluated for their acute anti-inflammatory effects on the mouse paw edema model induced by carragenan. Level of effect: Compound 3-methyl-1H-benzoindole-4,9-dione ( 66 ) > daucosterol ( 61 ) > hyperosid ( 10 ) > quercitrin ( 9 ). Compound 3 -methyl-1H-benzoindole-4,9-dione ( 66 ) had the strongest inhibition, with an IC 50 value of 193.70 ± 5.24 μg /mL. These four compounds were also evaluated in vitro anti-inflammatory model by albumin denaturation test by Bui Thanh Tung et al. [29], the results showed that all four compounds effectively inhibited thermal albumin denaturation at different concentrations in the above order. The anti-inflammatory activity of all compounds was concentration dependent.
In 2018, Le Thi Hong Nhung also tested the anti-inflammatory activity of n -hexane, ethyl acetate and butanol extracts from S. speciosa by determining the inhibitory activity on nitric oxide (NO) production on RAW264.7 cells. The results showed that in 3 residues
In the experimental extraction, n- hexane and ethyl acetate residues showed good inhibitory activity against NO production. Notably, n -hexane residue had stronger activity at low concentrations (IC 50 10.82 ±1.80
µg/mL) [1].
1.3. 3. Pain relief effect
Nurat haseen [155] used methanol extract from the bark and roots of S. speciosa to evaluate the analgesic effect on three models: acetic acid, hot plate and formalin. On the acetic acid-induced colic model, the methanol extract showed a maximum pain inhibitory effect of 79.21% when using a dose of 200 mg/kg body weight compared to aspirin which showed an inhibition of 88.01%. The extract with doses of 100 and 200 mg/kg showed significant effects after 1 hour on the hot plate model. The results were clearly shown at a dose of 200 mg/kg of 20.28 ± 4.6 (seconds) which was comparable to the reference drug tramadol (22.60 ± 4.3 seconds). The analgesic effect in the formalin-induced paw licking test, measured by time (29.6 ± 3.1 seconds) at a dose of 200 mg/kg, was close to that of the reference drug indomethacin (39.3 ± 2.9 seconds). Thus, the results showed that the methanol extract from the bark and roots of S. speciosa had a fairly good central analgesic effect.
Another study by Progga et al. [161] used ethanol extract of S. nobilis to evaluate the analgesic effect on acetic acid model. The study sample was used at doses of 250 and 500 mg/kg, the positive control was sodium diclofenac at a dose of 25 mg/kg. The results showed that ethanol extract of S. nobilis showed pain inhibition by 32.7% (p < 0.03) and 41.78% (p < 0.02) at doses of 250 and 500 mg/kg, respectively, while the positive control sodium diclofenac showed significant inhibition of pain reflex by 74.23% (p < 0.008).
1.3.4. Antimicrobial effects
In 2014, Abu Shuaib and colleagues evaluated the antibacterial, antifungal and insecticidal effects of S. nobilis by disc diffusion method on 15 strains of Gram (+) and Gram (-) bacteria, 6 strains of fungi and one strain of Tribolium castaneum . The results showed that, in the 3 fractions obtained from the ethanol extract of S. nobilis, which were petroleum ether, chloroform and ethyl acetate, the chloroform fraction

![Study on the structure and biological activity of some compounds isolated from the halong palm (Livistona halongensis TH Nguyen & Kiew) and gotu kola [Centella asiatica(Linn.) Urban] - 2](https://tailieuthamkhao.com/en/uploads/2025/03/16/study-on-the-structure-and-biological-activity-of-some-compounds-isolated-from-the-halong-palm-120x90.jpg)

![Benzopyrano Ring of Flavonoid Compounds [31]](https://tailieuthamkhao.com/uploads/2022/05/09/luanghien-cuu-hoa-hoc-va-tham-do-hoat-tinh-sinh-hoc-cua-loai-thong-la-det-4-19-120x90.gif)

