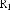
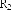
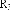
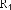







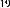
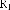



In addition, the Ph.D.Ty research group also relatively determined the content of some lupane-framed triterpenes in this species:
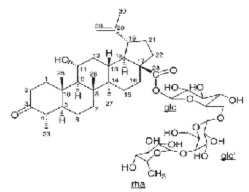
Continuing research on this plant species in Vietnam, in some recent publications, Phan Van Kiem and colleagues showed that in addition to the above compounds, the plant Ngũ gia bì hương collected in Northern Vietnam also contains many other compounds such as quercitrin ( 22 ), eleutheroside E ( 23 ), 1-(3-D-glucopyranosyl-2,6- dimethoxy-4-propenylphenol (acantrifoside E) ( 24 ), 1-[ β -D-glucopyranosyl- (1→6)- β -D-glucopyranosyl]-2,6-dimethoxy-4-propenylphenol (acantrifoside F) ( 25 ), syringin ( 26 ), (2R,3R)-2,3-di-(3,4-methylenedioxybenzyl)-butyrolactone ( 27 ), 3 α -acetoxy-30-hydroxylup-20(29)-ene-23,28-dioic acid (acantrifoic acid A) ( 28 ), 3 α -acetoxy-30-hydroxylup-20(29)-ene-23,28-dioic acid 28- O - α- L- rhamnopyranosyl-(1→4)- β -D - glucopyranosyl-(1→6)- β -D - glucopyranosyl ester (acantrifoside C) ( 29 ), 24-nor-11 α -hydroxy-3-oxo-lup-20(29)-ene-28-oic acid 28-O- α -L-rhamnopyranosyl-(l→4)- β -D-glucopyranosyl-(1→6)- β -D- glucopyranosyl ester (acantrifoside B) ( 30 ), ent -kaur-16-ene-19-oic acid ( 31 ), ent -pimara-8(14),15-dien-19-oic acid ( 32 ). Of these, compounds 24, 25, 28, 29 and 30 were the first compounds isolated from nature and compound 27 was the first isolated from this genus [21], [22],
[23], [24], [25].
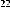
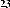
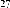
30

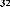
In addition, two pentacyclic triterpenes belonging to the taraxerane framework were also isolated by the research team from this species [21]:
Thus, the research results on the chemical composition of A. trifoliatus ( A. trifoliatus , Araliaceae family) have shown that this plant contains many triterpenes, especially lupane-framed triterpenes, many of which have high content.
b. Biological activity
In addition to the study of chemical composition, Phan Van Kiem and his colleagues also conducted a study on the activity of a series of substances isolated from this plant. The results showed that substances 22 and 28 have quite high anti-MAO activity even at low concentrations with IC 50 values of 0.12 and 0.18 mM, respectively. Substances 31 and 32 have anti-COX-1 enzyme activity with IC 50 values of 0.15 and 0.19 μg/ml, thus having very good anti-inflammatory activity. Testing the ability to resist tested bacteria, substances 24 and 31 can resist Gr (+) bacteria such as B. subtilis and S. aureus strains . In particular, the new phenylpropenyl glycoside compound, acantrifoside E ( 24 ) has strong antitumor activity against all three human cancer cell lines: human epithelial cancer cells (KB) IC 50 = 1.22 μg/ml, human pericardial cancer cells (RD) IC 50 = 2.06 μg/ml, and human hepatoma cells (Hep-2) IC 50 = 0.75 μg/ml [26].
In 2004, Xing Fu Cai and colleagues also conducted a survey on the activity of some lupane triterpene compounds found in the roots of A. koreanum . The results showed that impressic acid (20) , a compound found in A. trifoliatus with high content, exhibited transcription factor inhibitory activity at IC 50 = 12.65 µM [27].
In a recent publication in 2013, a research group tested the anti-inflammatory and anti-edematous properties in mice with three extracts of n -hexane, dichloroform, and methanol from A. trifoliatus leaves . The results showed that the methanol extract had significant anti-inflammatory properties, with the highest concentration of 500 mg/kg being able to resist up to 77.24% [28]. In addition, another research group tested the ability to improve memory and fight depression with the aqueous extract from the leaves of this species and also gave good results [29].
From the published results on the two studied species, it was shown that their main constituents are mainly flavonoid compounds and lupane-framed triterpenes. Because
So here is an overview of the chemistry and biological activities of these two main classes of compounds from the two studied plants.
1.2. Flavonoid compounds
Flavonoids are a class of phenolic compounds that are commonly found in plants. It can be said that flavonoids are exclusively produced in plants, only plants have the ability to biosynthesize flavonoids and are found in almost all plant species studied. According to scientists' calculations, each year about 2% of the carbon photosynthesized by plants (about 10 9 tons) is converted into flavonoids and related compounds.
[30]. Flavonoids have been studied by scientists since the 1940s. To date, more than 9,000 flavonoids have been isolated and their structures and biological activities have been determined. The term “Flavonoid” comes from the Latin word flavus meaning yellow – the color of most flavonoid compounds. However, some compounds are blue, purple-red, and some are colorless, also belonging to this class [31].
1.2.1. Chemical structure
Flavonoids are natural compounds with a carbon skeleton structure in the form of C6 – C3 –C6 or in other words , the basic skeleton consists of two benzene rings A, B connected together through a 3C chain. Except for some cases where the 3C chain is open such as chalcone, in most cases the 3C chain closes the ring with the A ring and forms a C heterocycle containing O such as [32]:
Figure 1.3: Benzopyrano ring of flavonoid compound [31]
Based on the oxidation level of the 3C chain and the position of the aryl radical (B ring) linked to the benzopyrano ring, flavonoids are divided into different types such as:
Euflavonoids: Flavonoids with an aryl group attached at the C2 position such as anthocyanidin, flavan, flavan 3-ol, flavan 4-ol, flavan 3,4-diol, flavanone, 3-hydroxy flavone, flavone flavonol, dihydro chalcone, chalcone, aurone.
Isoflavonoids: Flavonoids with an aryl group attached at the C3 position such as isoflavan, isoflav-3-ene, isoflavan-4-ol, isoflavanone, isoflavone, rotenoid, pterocarpan, coumestan, 3-arylcoumarin, coumaronochromen, coumaronochromone, dihydroisochalcone, homo-isoflavone.
Neoflavonoids: Flavonoids with an aryl group attached at the C4 position. This group of compounds has only been found in a few plant species such as 4-arylchroman, 4-aryl couramin, dalbergion, 3,4-dihydro-4-arylcouramin, neoflavene.
In addition, people also classify them into flavonoids, biflavonoids (dimeric flavonoids), triflavonoids (composed of 3 flavonoid monomers), and flavonlignans (flavonoids whose molecules have a lignan structure).
In plants flavonoids exist in various forms of substituents such as hydroxyl addition, methylation and most importantly glycosylation. Occasionally, other substituents such as aromatic, aliphatic acid, sulfate, prenyl, methylendioxy or isoprenyl groups are also attached to the flavonoid nucleus and its glycosides.
3' | O OH Flavanol | ||||
7 | 8 A | 2' 9 O 2 1' C | B 6' | 4' 5' | |
6 | 5 | 4 3 10 | |||
O | |||||
Flavones | |||||
Flavanone | Anthocyanidin | ||||
Isoflavones |
Chalcone | ||||
Maybe you are interested!
-
 Phenolic Compounds Isolated from the Genus Sanchezia
Phenolic Compounds Isolated from the Genus Sanchezia -
 Evaluation of the treatment results of Schatzker type V-VI tibial plateau fractures by combining minimal bone fusion and external ring fixation under fluoroscopy - 13
Evaluation of the treatment results of Schatzker type V-VI tibial plateau fractures by combining minimal bone fusion and external ring fixation under fluoroscopy - 13 -
 Results From Acanthopanax Trifoliatus L. Merr.
Results From Acanthopanax Trifoliatus L. Merr. -
 The “3-Ring Control” Model of Risk Management for Commercial Banks
The “3-Ring Control” Model of Risk Management for Commercial Banks -
![Study on the structure and biological activity of some compounds isolated from the halong palm (Livistona halongensis TH Nguyen & Kiew) and gotu kola [Centella asiatica(Linn.) Urban] - 2](data:image/svg+xml,%3Csvg%20xmlns=%22http://www.w3.org/2000/svg%22%20viewBox=%220%200%2075%2075%22%3E%3C/svg%3E) Study on the structure and biological activity of some compounds isolated from the halong palm (Livistona halongensis TH Nguyen & Kiew) and gotu kola [Centella asiatica(Linn.) Urban] - 2
Study on the structure and biological activity of some compounds isolated from the halong palm (Livistona halongensis TH Nguyen & Kiew) and gotu kola [Centella asiatica(Linn.) Urban] - 2
Table 1.3. Some common flavonoid frameworks
1.2.2. Biological activity
Flavonoids are a group of compounds that have been called “nature’s biochemical repairmen” due to their ability to modify the body’s response to other compounds in allergens, viruses, and carcinogens. This provides valuable activities.
It can be said that antioxidant activity is the characteristic activity of flavonoid compounds. According to Rice-Avans and colleagues [33], antioxidant capacity can be explained based on the characteristics of their molecular structure such as: The molecule contains hydroxy groups directly linked to aromatic rings that can donate hydrogen, helping flavonoids to participate in oxidation-reduction reactions, capturing free radicals; contains aromatic rings (benzene rings, heteroatom rings) and multiple bonds (C=C, C=O bonds) that form a conjugated system that helps stabilize free radicals formed when they capture active oxygen molecules; contains groups that can form transition complexes with metal ions such as catechol... to help reduce the production of active oxygen molecules. For example, three major flavonoids were isolated from the roots of Scutellaria baicalensis : wogonin (5,7-dihydroxy-8-methoxyflavonoid) ( 35 ), baicalein (5,6,7-trihydroxy-flavone) ( 36 ), and its 7-glucuronide derivative (baicalin) ( 37 ). The results of the antioxidant activity tests of these compounds showed that baicalin had the best antioxidant activity (72%). Thus, the addition of a glucuronide group at C-7 significantly increased the antioxidant capacity [34].
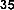
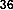
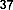
In a study, Das and Pereira showed that flavonoid molecules containing carbonyl groups at C-4 and double bonds between C-2 and C-3 have high antioxidant capacity like butein ( 38 ) [35]:
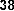
Many studies have shown the miracle of flavonoid compounds in the treatment of cancer. Two flavones 39 , 40 were found from the leaves of Baeckea frutescens (Myrtaceae) with very strong cytotoxicity (IC 50 = 0.25μg/ml) against the leukemia cell line (L1210) [36]:
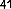
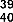
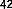
Anticancer activity by altering cell morphology leading to inhibition of cell proliferation of cancer cell lines was found in the compounds naringenin ( 41 ) and kaempferol 3- O- (2'',6''-di- Op-trans- coumaroyl) glucoside ( 42 ), isolated from the flowers of Melastoma malabathricum on breast cancer cell line (MCF-7) with values of 0.28 and 1.3μM, respectively [37].
In addition, flavonoid compounds also show the ability to resist HIV virus. Baicalin (5,6,7-trihydroxy-flavon 7-glucuronide) ( 37 ) isolated from Scutellaria baicalensis has the ability to directly inhibit HIV virus; or two biflavones, hinokiflavone ( 43 ) and robustafavone ( 44 ), can inhibit HIV-1 reverse transcriptase with IC 50 = 65µM [38], [39].
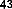
In addition, flavonoid compounds also exhibit activities such as anti-inflammatory, antibacterial, antidiabetic, etc. Due to the increasing number of people with diabetes and the dangerous complications caused by this disease, their activities on diabetes are of particular interest to scientists. Research results have shown that flavonoids themselves act as insulin stimulants or mimic the function of insulin; in addition, they also affect the activity of enzymes in the process of sugar metabolism...[40],[41]. From the roots of Dorstenia psilurus (Mulberry family, Moraceae), Tabopda and colleagues isolated the prenyl flavonol compound dorsilurin F ( 45 ) with good α -glucosidase enzyme inhibitory activity with IC 50 = 4.13μM. Notably, also with the α -glucosidase enzyme, a derivative of luteolin, 6-hydroxyluteolin ( 46 ) can inhibit up to 92% at concentrations
500μM [42].
OH
HO
OH
OH O
45
OH
O
OH
COUGH
COUGH
OH O
46
O
O
OH
HO H OH O
47
From the plant Eysenhardtia platycarpa (Fabaceae), 5,4,1”-trihydroxyy-6,7- (3”,3”-dimethylchromano) flavone ( 47 ) was isolated. This substance has the ability to resist hyperglycemia in mice at a dose of 30 mg/kg body weight [43].






![Study on the structure and biological activity of some compounds isolated from the halong palm (Livistona halongensis TH Nguyen & Kiew) and gotu kola [Centella asiatica(Linn.) Urban] - 2](https://tailieuthamkhao.com/en/uploads/2025/03/16/study-on-the-structure-and-biological-activity-of-some-compounds-isolated-from-the-halong-palm-120x90.jpg)