Figure 1.16. Structure of plasmid pITB-AST
CHAPTER 2. MATERIALS AND METHODS
2.1. Materials
2.1.1. Soybean varieties
Seven soybean varieties were collected and used, including 6 Vietnamese varieties (domesticated hybrids) and one Japanese variety (Table 2.1). Specifically, the soybean varieties include MTD 176, MTD 760-3, MTD 860-3, MTD 885-1 from the Department of Agricultural Genetics and Breeding, Faculty of Agriculture & Applied Biology - Can Tho University; HL 07-15; OMĐN 29 from Hung Loc Agricultural Experimental Research Center, Southern Institute of Agricultural Science and Technology; Edamame is a Japanese soybean variety from Yen Khe Company Limited.
Table 2 1 Origin and some characteristics of soybean varieties used .
Alike
Soybean
MTD 176 | MTD 760-4 | MTD 860-3 | MTD 885-1 | Edamame | HL 07- 15 | OMDN 29 | |
Source | DH4 x CES 97- 13 | MTD176 x A70 | TWK* x MTD176 | DT 2000 x MTĐ65 | No information available | HL 203 x HL 92 | OMQDN 1 x Kettum |
TG born adult (day) | 80-85 | 85-90 | 80-85 | 85 | 78-85 | 80-85 | |
Productivity (ton/ha) | 2.0-3.2 | 2.8-3.8 | 1.9-2.8 | 2.5-3.5 | 2.19-2.37 | 2-2.5 | |
Germination rate | >95% | >95% | >95% | >95% | >95% | >95% | >95% |
Place of supply | Department of Agricultural Genetics and Breeding, Faculty Agriculture & Applied Biology - Can Tho University | LLC Yen Khe | Research Center Hung Loc Agricultural Experiment | ||||
Maybe you are interested!
-
 Solution 2: Manufacturing Enterprises Must Regularly Maintain Transaction Activities With Partners To Increase Proficiency, Create
Solution 2: Manufacturing Enterprises Must Regularly Maintain Transaction Activities With Partners To Increase Proficiency, Create -
 Starting a business Opening a coffee and ornamental plant shop - 2
Starting a business Opening a coffee and ornamental plant shop - 2 -
 Structural Characteristics of the Forest Stand with Distribution of 5-Needle Pine
Structural Characteristics of the Forest Stand with Distribution of 5-Needle Pine -
 Create a Table of Area and Dimensions of Constructions.
Create a Table of Area and Dimensions of Constructions. -
 Monitoring Report Based on Approved Plan Item: Operation Management Area of Song Bung 4 Hydropower Plant
Monitoring Report Based on Approved Plan Item: Operation Management Area of Song Bung 4 Hydropower Plant
2.1.2. Bacterial strains, plasmids
Agrobacterium tumefaciens EHA 105 strain used for gene transformation into soybean
Plasmid pCAMBIA3301 (CAMBIA - International Centre for Molecular Biology Applications in Agriculture, Australia) is approximately 11.3 kb in size. The T-DNA region carries the gene combination P CaMV35S - bar - T nos / P CaMV35S - gus -T nos .
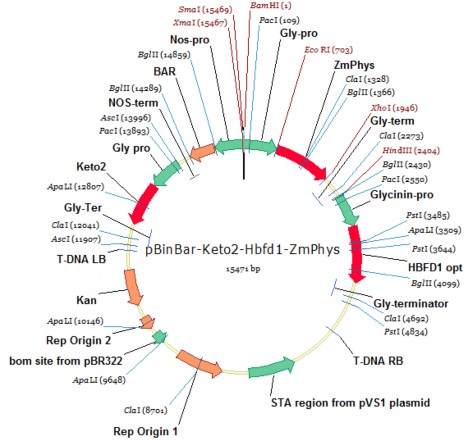
Plasmid pITB-AST (Genetic Technology Department - Institute of Structural Tropical Biology) is a binary plasmid in the binary system used for gene transfer of Agrobacterium tumefaciens with a size of about 15.5 kb, the T-DNA of the plasmid carries the gene combination P glycinin-cbfd2- T glycinin/ P glycinin-hbfd1- T glycinin/ P glycinin - Zm-psy -T glycinin and the selected gene P nos-bar- T nos expressed in plants. In which, the gene Zm-psy expresses the enzyme phytoene synthase that enhances the synthesis of phytoene, promoting increased synthesis of β-carotene in seeds; the genes cbfd2, hbfd1 express enzymes that can use the substrate β-carotene in soybean seeds to synthesize astaxanthin.
2.1.3. Needle puncture to create wound for transgenic sample
The acupuncture needle is prepared from a 10 cm piece of wire with an inner core consisting of 30 small copper wires (the copper wire size is 0.1 mm). The cross-sectional diameter of the wire is 3.15 mm, with a core of 1.7 mm. About 0.5 cm of the plastic cover at one end is removed to expose the copper wire inside, then cut off, leaving only 10 copper wires, which are sharpened with sandpaper (Figure 3.17 b).
2.1.4. Tissue culture environment
The tissue culture medium, presented in Table 2.2, used the basic components, minerals, and vitamins of MS medium [153] and Gamborg B5 (B5) [154], with additional components from Paz et al. (2006), Ma and Wu (2008) [111] [102]. The medium components were produced by Ducefa (Netherlands), except for sucrose (Bien Hoa refined sugar, Vietnam); agar (Nam Ha Long, Vietnam).
Table 2.2. Composition of tissue culture and gene
Environment
Mineral | Vitamin | Growth regulator | Other ingredients | |
Seed germination medium (GM) | 1X MS | B5 | 30 g/l Sucrose; 10 g/l agar ; pH 5.7 | |
Shoot regeneration medium (SI) | 1X B5 | B5 | (0; 0.5; 1; 1.5; 2; 2.5; 3) mg/l BA | 30 g/l Sucrose; 0.59 g/l MES; and 10 g/l agar; 500 mg/l cefotaxime. pH 5.7 |
Growth environment | 1X MS | B5 | 0.1 mg/l IAA; 0.5 mg/l GA 3 ; | 30 g/l Sucrose; 0.59 g/l MES; 10 g/l agar; 500 |
body (SE)
1 mg/l Zeatin-R | mg/l cefotaxime; 50 mg/l Asparagine; 100 mg/l L-Pyroglutamic Acid. pH 5.7 | |||
Infectious environment (IF) | 1/10 X B5 | 1/10X B5 | 0.25 mg/l GA 3 ; 2 mg/l BA | 30 g/l Sucrose; 3.9 g/l MES; 100 µM acetocyringone. pH 5.4 |
Common culture medium (CC) | 1/10X B5 | 1/10X B5 | 0.25 mg/l GA 3 ; 2 mg/l BA | 30 g/l Sucrose, 3.9 g/l MES and 10 g/l agar, 400 mg/l Cysteine; 154.2 mg/l Dithiothreitol; 100 µM acetosyringone. pH 5.4 |
Environment plant growth (RM) | 1X MS | B5 | (0; 0.5; 1; 1.5; 2) mg/l IBA | 20 g/l Sucrose; 0.59 g/l MES; 10 g/l agar pH 5.6. |
2.1.5. Chemicals in other experiments
- DNA extraction from plants
Method of Dellaporta et al. (1983) [155].
+ 5 M NaCl. + 10% SDS.
+ β Mercaptoethanol. + Potassium acetate 5M.
+ Tris-HCl 1 M. + RNAse.
+ EDTA 0.5 M. + Ethanol 80%.
+ Isopropanol. + Phenol/Chloroform 1:1.
DNeasy Plant Mini Kit (Qiagen)
- PCR reaction
Promega PCR kit includes: dNTPs (ATP, CTP, GTP, TTP), 10X PCR buffer, Taq Polymerase, PCR water
+ Forward primer (Primer F) and reverse primer (Primer R).
+ Plant DNA sample.
- GUS histochemistry
+ X-gluc 0.02 M stock solution: 70 mg X-gluc, 2 ml DMSO, 8 ml sterile distilled water.
+ X-gluc solution for staining samples: 5 ml stock X-gluc, 10 ml Na 2 HPO 4 buffer solution (pH 7), 4 ml 0.25 M EDTA (pH 7), 1 ml 10% triton-X 100.
- Agarose gel electrophoresis
+ Agarose.
+ Gelred.
+ TAE buffer solution (10X stock): Tris base 48.4 g/l, glacial acetic acid 11.4 ml, EDTA Disodium 3.72 g/l pH 8 800 ml, water to volume 1 liter.
- Gene transfer experiment
+ Phosphinothricin (PPT): mixed in distilled water, sterile filtered, stock concentration 2.5 mg/ml used for selection for bar gene .
+ Kanamycin: stock concentration 10 mg/ml, mixed in distilled water, sterile filtered. Use to select A. tumefaciens bacteria carrying the gene transfer plasmid.
+ Rifamicin: stock 10 mg/ml, mixed in distilled water and sterile filtered. Used for selection of A. tumefaciens bacteria carrying helper plasmid.
+ Acetosyringone: 2 mg/ml dissolved in alcohol, add distilled water, sterile filter.
+ Cefotaxime: 500 mg/ml mixed in distilled water, sterile filtered, used for sterilization.
- Southern bot
Amersham ECL Direct Nucleic Acid Labelling And Detection Systems kit from GE Healthcare UK.
+ Depuration solution: HCl 250 mM.
+ Denaturation solution: NaCl 1.5 M ; NaOH 0.5 M.
+ Neutralizing solution: NaCl 1.5 M; Tris HCl 0.5 M.
+ 20x SSC: Na 3 citrate 0.3 M; 3 M NaCl.
+ Primary wash buffer: Urea 6 M; SDS 0.4%; 20 x SSC.
+ Secondary wash buffer: 20 x SSC.
- Testing herbicide resistance of transgenic plants
+ Basta herbicide: Contains DL-Phosphinothricin (glufosinate ammonium) 200 g/l.
2.1.6. Equipment used
- PCR Techne ® machine - Sterile culture cabinet (Sanyo, Japan)
- Water distiller (Schott) - Shaker (Innova 2100, Flatform Shaker)
- Autoclave (Hirayama) - Incubator (SANYO)
- Innova 4230 shaker - Biorad ® electrophoresis machine
- Biorad gel imager - Eppendorf ® , 5417R refrigerated centrifuge
- BTX ECM pulse machine - Thermo OD meter, USA
- Ultrasonic wave generator - Negative pressure chamber of gene blaster (Bio-Rad, USA)
2.2. Content and Method
Content 1: Building a complete in vitro plantlet generation system from cotyledons and half seeds.
2.2.1. Soybean variety selection
Selection of domesticated soybean varieties, high yield, good resistance to pests and diseases, widely grown.
The β-carotene content of selected soybean varieties was tested by HPLC because this is the necessary substrate for enzymes (encoded by transformed genes) to metabolize and create astaxanthin.
Soybean lines with the presence of β-carotene were tested for germination to ensure stability, accuracy, and efficiency during the experiment. Seed germination was tested by sowing seeds on wet blotting paper placed in petri dishes. Each dish was sown with 20 seeds, repeated 3 times. After 5 days, the number of germinated seeds was checked.
2.2.2. Seed sterilization
Chlorine gas disinfection
Place soybean seeds in a single layer of 90 x 15 mm petri dishes. Arrange the dishes, with the lids open, and a 250 ml measuring cup in a sealed desiccator. Add 100 ml of Javel water (38 g/l chlorine content) to the measuring cup and add 3.5 ml of HCl (12 N) by pouring down the side of the cup. Close the desiccator and keep for different times (16, 24 h). After the chlorine exposure time, close the petri dishes containing the seeds and place them in a sterile incubator. Open the dishes to allow the chlorine gas to completely escape within 30 min [111].
Disinfect with Javel solution
Soybean seeds were sterilized by soaking in 50% Javel water solution (chlorine content 38 g/l) for different times of 30, 40, 50 minutes, then
51
Rinse the sample three times with sterile distilled water [102][150].
Seeds after being sterilized by agents were cultured on solid medium including MS mineral, vitamin B5, pH 5.6 [111][154]. The experiments were carried out with 12 samples/medium bottle, repeated 3 times. The experiment was arranged in a completely randomized single-factor design.
Monitoring indicators: Rate of uninfected seeds, rate of uninfected germinated seeds.
2.2.3. Effect of BA on shoot regeneration of cotyledon and half seed
Investigate the effect of BA on the ability to form adventitious buds of cotyledons and half seeds. Thereby determining the appropriate environment to stimulate shoot formation.
The seed sterilization method is applied according to the optimal procedure obtained from the section.
2.2.2. Cotyledon samples were prepared according to the procedures of Hinchee et al. (1988), Olhoft et al. (2003) [121][126] (QC 1) with the following steps: sowing bean seeds on MS-vitamin B5 medium for 5 days for germination. Then, the germinated plants had their roots removed, keeping the hypocotyledonous column about 3-5 mm long, attached to the cotyledon. Using a 15-gauge scalpel, separate the two cotyledons by splitting longitudinally from the node to the end of the hypocotyledonous column, then removing the apical bud and lateral buds. Next, gently cut 7 times perpendicular to the hypocotyledon to create a wound for the axillary meristem region. The cotyledonous sample, including the cotyledon, hypocotyledonous column, and axillary meristem region, was used to induce buds (Figure 2.1).
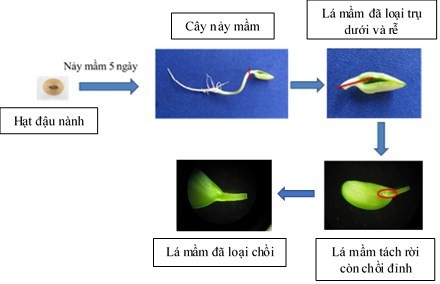
Figure 2.1. Diagram of steps for preparing cotyledon samples
Half-grain samples were prepared according to the procedure of Paz et al. (2006)
[111] (QC 2) with steps including soaking beans in sterile distilled water
about 20 hours. Then, separate the two cotyledons along the hilum. Next, remove the embryo axis (Figure 2.2).



Soybeans Soybeans soaked in water for 20 hours
Half of the seed still has the embryo axis.
Half of the seed has had its embryo axis removed.
Figure 2.2. Diagram of steps for preparing half-grain sample
Cotyledons and half of the seeds prepared in the above ways were placed on SI regeneration medium supplemented with BA (0; 0.5; 1; 1.5; 2; 2.5; 3) mg/l. Each treatment consisted of 10 samples, repeated 3 times. The experiment was arranged in a single-factor, completely randomized manner. The samples were grown on light shelves (18 hours of light/day) with an intensity of 2,000 -
3,000 lux, temperature 25±2 o C. Record results after 3 weeks.
Monitoring indicators: Rate of shoot regeneration, average number of regenerated shoots (number of regenerated shoots/number of surveyed samples), normal and uniform shoot morphology.
Shoot clusters formed on optimal shoot growth medium were transferred to SE stem growth medium. Shoots about 3 cm high were separated and cultured on rooting medium .
2.2.4. Effect of IBA on rooting of in vitro shoots
Experiment to determine the appropriate IBA concentration to stimulate root formation for shoots
in vitro to create complete plantlets.
In vitro shoots about 3 cm high were cultured on MS mineral medium, vitamin B5, 0.59 g/l MES, supplemented with IBA at different concentrations (0; 0.5; 1; 1.5; 2) mg/l, PH 5.6. Results were recorded after 3 weeks of culture.
Each experiment consisted of 10 samples, repeated 3 times. The experiment was arranged in a single-factor, completely randomized design.
Culture conditions: samples were cultured on light shelves (18 hours of light/day) with intensity of 2,000 - 3,000 lux, temperature 25±2 o C.
Monitoring indicators: rooting shoot ratio, average number of roots (number of roots formed/number of samples surveyed).





