Cirrhotic liver tissue makes it difficult for surgical instruments to pass through the parenchyma easily.
In this study, we applied the technique of controlling the hepatic pedicle outside the Glisson's capsule outside the liver.
|
Maybe you are interested!
-
 Studies on Measures to Prevent and Control Small Liver Flukes
Studies on Measures to Prevent and Control Small Liver Flukes -
 Clinical and paraclinical characteristics and treatment outcomes of patients with acute liver failure at the Poison Control Center of Bach Mai Hospital in 2020 - 2021 - 10
Clinical and paraclinical characteristics and treatment outcomes of patients with acute liver failure at the Poison Control Center of Bach Mai Hospital in 2020 - 2021 - 10 -
 Effectiveness of Community Intervention to Prevent and Control Small Liver Flukes in People After 2 Years of Intervention
Effectiveness of Community Intervention to Prevent and Control Small Liver Flukes in People After 2 Years of Intervention -
 Internal control of revenue and expenditure activities at the National Children's Hospital - 2
Internal control of revenue and expenditure activities at the National Children's Hospital - 2 -
 Measurement and Remote Control – Electrical Engineering Industry - 8
Measurement and Remote Control – Electrical Engineering Industry - 8
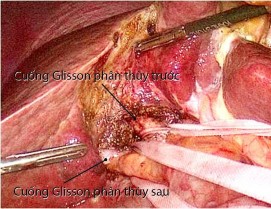
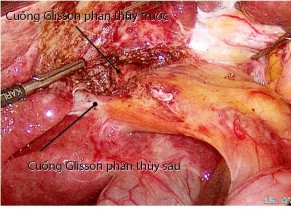
Figure 4.3. Glisson's extracapsular hepatic pedicle control technique, "Case 184, Patient: Nguyen Thi Tien M., SNV 12-0024098"
We found that this extrahepatic Glisson's capsule hepatic pedicle control technique has many advantages:
- Limit hepatic pedicle dissection, shorten surgery time.
- Minimal dissection of the hepatic pedicle helps reduce damage to the already dilated lymphatic vessels in cirrhotic patients, thereby reducing the risk of postoperative ascites.
- Dissection of the hepatic pedicle outside the Glisson capsule avoids the risk of damaging the structures in the hepatic pedicle area, which has many anatomical variations.
- More selective control of the hepatic pedicle, minimizing ischemia of the preserved liver.
- Can determine the boundaries between liver lobes, facilitating anatomical liver resection, which has many advantages: reducing blood loss when resecting liver parenchyma, better ensuring the principles of cancer treatment.
4.1.4.6. Liver parenchyma resection and hemostasis control
Control of hemostasis during parenchymal resection is a critical issue in liver resection.
In PTNS, hemostasis control is even more difficult because it is impossible to compress the cut surface or control bleeding points with the surgeon's hands. When bleeding occurs, if not handled effectively and promptly, the surgical field will become darker and more difficult to observe. If the leakage of large blood vessels is not well controlled, it is possible to increase the risk of embolism due to air... Furthermore, heavy blood loss during surgery, intraoperative and postoperative blood transfusion are among the risk factors that increase the recurrence rate after liver resection for HCC. Therefore, effective hemostasis control during parenchymal resection is one of the major challenges that must be overcome, determining the success of PTNS liver resection for HCC.
To limit blood loss when cutting parenchyma in laparoscopic surgery, good control of the hepatic pedicle, cutting liver parenchyma according to anatomical boundaries, and effective and reasonable use of combined equipment and instruments are essential factors.
Thanks to the development of science and technology, there are more and more equipment to support liver parenchyma resection. The structures of the portal pedicle and large blood vessels can be dissected and then cut with a stapler. Smaller blood vessels can be clamped with plastic or metal clips. Liver parenchyma can be cut and hemostasis can be done with cauterization hooks, scissors, ultrasonic cutters (Harmonic Scalpel, Sonosurg,...), Ligasure, CUSA, or Tissue link,... Small bleeding points can be controlled with bipolar cauterization clamps or sutures... However, up to now, there is still no standard procedure for liver parenchyma resection technique through endoscopy. Most authors perform laparoscopic liver resection according to habit, experience or available equipment conditions.
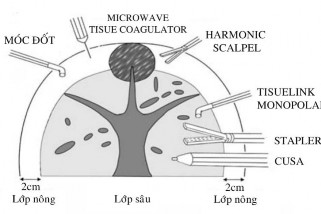
Author Kanako recommends dividing the parenchymal layers to use the appropriate instrument:...
|
Figure 4.4. Strategy for using liver parenchyma cutting instruments according to Kaneko "Source: Kaneko, 2009" [ 50]
Currently, most authors in the world mainly cut liver parenchyma using CUSA. This is a tool that helps destroy and suck liver parenchyma out, allowing exposure of small blood vessels in the parenchyma so that they can be clipped and cut. This tool allows for meticulous dissection, minimizing blood loss, very suitable for tumors in special locations, near large blood vessels that require meticulous dissection, but the disadvantage is that it prolongs the surgery time and is expensive equipment.
In our study, in most cases, we cut the liver according to the anatomical structure. The surgical plane is the plane separating the lobes and the HPT of the liver. In this plane, we encounter very few structures of the portal pedicle, so the process of cutting the parenchyma is convenient, fast, with little blood loss and minimal damage to abnormal anatomical structures. After controlling the hepatic pedicle of the liver part to be resected, we cut the parenchyma with an ultrasonic cutting knife (Harmonic Scalpel). The ultrasonic blade is also an effective tool for us to crush the liver parenchyma, playing a similar role to the Kelly clamp in the "Crushing clamp" technique, to explore and detect the vascular pedicles inside the parenchyma before cutting. This technique helps us
I limit the risk of puncturing large blood vessels causing heavy bleeding during surgery.
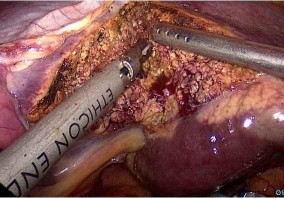
|
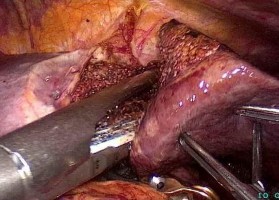
Figure 4.5. Cutting liver parenchyma with ultrasonic cautery knife.
Stapler resection of the Glisson pedicle of the left lobe. "Case 205, Patient: Le Van H., SNV 13-0001518"
The large blood vessels exposed during the process of crushing and cutting the liver parenchyma are clearly exposed, then clamped or Hem o lock and cut. Small bleeding points are easily controlled by bipolar cautery, which is an effective and safe means of hemostasis. We found that: selective and effective control of the hepatic pedicle, cutting the liver according to the anatomical boundaries between the segments, lowering the central venous pressure along with the strategy of using appropriate liver parenchyma cutting instruments are the key issues to control good hemostasis when cutting the liver parenchyma.
In this study, we only used the ultrasonic scalpel (Harmonic Scalpel) as the main means to cut liver parenchyma. This is a relatively common PTNS instrument, equipped in many hospitals to perform PTNS to cut the colon, stomach, etc. We found that the ultrasonic scalpel performed liver parenchyma resection relatively quickly, effectively stopped bleeding (needs to be combined with good control of the hepatic pedicle, liver resection according to the anatomical boundary, low central venous pressure), safe and with few complications.
4.1.5. Ability to perform various types of liver resection surgeries by PTNS
We have 260 successful cases of PTNS with various types of liver resection.
4.1.5.1. For left lobe liver resection
Left lobectomy is often the first surgery of most surgeons when starting to perform liver resection. Because this is a relatively uncomplicated surgery, no need to dissect the hepatic pedicle, the liver parenchyma in the cut area is relatively thin, the structure of the Glisson pedicle of HPT 2 and 3 is easy to access. Normally, we use Bulldog, temporarily clamping the left half of the hepatic pedicle to limit bleeding during the process of cutting the superficial parenchyma. Then the Glisson pedicles of HPT 2 and 3 are cut at the same time with a stapler. The left hepatic vein can also be cut with a stapler or cut with scissors after clamping the Hem O lok.
In our study, left lobe hepatectomy accounted for the highest proportion of 82 patients (31.5%), this is a simple surgery, can be performed easily, has a short surgery time, and has few complications and side effects. Even with large tumors, severe cirrhosis, and obese patients, surgery can still be performed easily and is somewhat more favorable than open surgery. This is demonstrated by the results of our study, in the left lobe hepatectomy group, the average surgery time was 90 minutes, blood loss was about 50ml, and no complications or side effects were recorded after surgery. Patients can walk and do daily activities 2 days after surgery (Table 3.21). We think this is the standard treatment method for cases of HCC with left lobe hepatectomy.
At the first World Liver Resection Consensus Conference, the authors also agreed on the value of left lobectomy. This technique is recommended as the standard treatment method and should be used routinely for patients requiring liver resection [18],[55],[85],[94],[96],[107].
4.1.5.2. For HPTs in the peripheral zone of the liver (2, 3, 4, 5, 6)
These are the positions where we performed the most liver resections in the study: HPT 6 (19.6%), HPT 5 (8.5%), HPT 4 (5.4%), HPT 3
(4.2%)... This type of surgery is not too complicated and can be performed safely and effectively. However, the HPT 5 liver resection technique is relatively complicated because of the multi-faceted surgical plane, the Glisson pedicle has many variations, and is near the division of the right Glisson pedicle and the middle hepatic vein tail, so caution is needed to avoid complications of bleeding or bile leakage. In 11 cases of the failed PTNS group, there were 4 cases of tumors in HPT 5, we converted to open surgery because of the risk of severe bleeding (loss of more than 500 ml).
At the world consensus conference on PTNS liver resection, most authors agreed that tumors located in the peripheral HPTs of the liver are suitable lesions for PTNS liver resection [18],[43],[85],[96].
4.1.5.3. For HPTs in the posterior superior region of the liver
The subsegments in the posterior superior region (HPT 1, 4a, 7, 8) are difficult areas for PTNS liver resection. This is understandable due to the limitation of the ability to observe through the endoscope, the difficulty in accessing the surgical field, and the difficulty in supporting or pulling the liver with laparoscopic instruments. Furthermore, the tumors in the posterior superior region are located very close to the hepatic veins and vena cava. Therefore, the problem of controlling hemostasis and ensuring the resection surface is far from the tumor becomes difficult. In the early days, most authors believed that tumors in this liver area were not an indication for PTNS liver resection. However, according to the training curve, PTNS liver resection for the posterior superior HPTs has begun to be reported recently.
Author Yoon [111] in Korea divided patients into two groups based on the location of the planned liver resection. The anterolateral region included HPTs 2, 3, 4b, 5, 6 and the posteroinferior region included HPTs 1, 4a, 6, 7. Except for the location, the two groups were similar in preoperative characteristics. The author found that
PTNS liver resection in the posterior inferior region had a longer operative time and a longer hospital stay with statistical significance. This group tended to lose more blood during surgery and had a higher conversion rate to open surgery, although not statistically significant. However, the complication rate and survival time of patients after surgery were similar in the two groups.
In our study, 7 patients (2.7%) had HPT 7 liver resection, two patients (0.8%) had HPT 8 liver resection. However, these were cases of liver resection that did not follow the anatomical boundaries precisely.
We found that, for tumors at HPT 7 or 8, if liver function is not too poor, allowing for larger liver resections, we tend to perform anterior segmental hepatectomy (instead of simple HPT 8) or posterior segmental hepatectomy (instead of simple HPT 7). We prioritize the indication of segmental hepatectomy because the Glisson pedicle is easier to dissect and control, the resection surface is flat and simple, allowing for wide resection of parenchyma along the anatomical boundary, so it better complies with the principles of treating HCC. This is also the reason why our study had 9 patients (3.5%) with posterior segmental hepatectomy and 7 patients (2.7%) with anterior segmental hepatectomy according to anatomy. This rate is much higher than the rate of resection of simple posterior-upper liver HPTs such as simple HPT 7 (2.7%) or simple HPT 8 (0.8%). Our view is similar to that of Cho [27], Yoon [111] and Han [44]. These authors recommended complete segmental resection along the anatomical boundary for tumors in the posterosuperior region, located more than 3 cm below the liver surface.
4.1.5.4. PTNS's ability to perform major liver resection
Major hepatectomy is the surgical removal of at least 3 liver HPTs [48]. One of the concerns and considerations before major hepatectomy is the issue of hepatic pedicle dissection, the ability to manipulate the large liver, and control hemostasis over a large parenchymal resection area.
In our study, we had 26 major liver resections, including: 9 cases (3.5%) of right liver resections, 15 cases (5.8%) of left liver resections, 2 cases (0.8%) of central liver resections (HPT 4, 5, 8). During the procedure, we applied the surgical technique of accessing the extracapsular Glisson pedicle and found that this technique helps simplify major liver resection with many advantages. The Glisson pedicles of the right or left liver can be clearly dissected in the hilum area. Temporarily clamping these pedicles allows identification of the corresponding liver parenchyma through the ischemic image and determines the surgical plane between the right and left livers. After checking to ensure that the remaining liver is safe - not ischemic, the liver pedicles are clamped and cut with a stapler. Next, the liver parenchyma is cut along the anatomical plane, separating the right and left livers from the front to the back, to the front of the vena cava. Then, the right or left hepatic vein is exposed and cut in half. Finally, the liver, after all the vascular pedicles have been removed, is moved away from the diaphragm and posterior abdominal wall, and taken out of the abdominal cavity. This is also the anterior approach technique proposed by Belghiti [11], which better adheres to the "No touch" principle in cancer treatment.
The total time of major liver resection in our study was 180 minutes (60-345 minutes), the average blood loss was 200 ml (100-600 ml), no case required blood transfusion during or after surgery. After surgery, 1 patient had transient liver dysfunction, 2 patients had pneumonia. All 3 patients recovered well after medical treatment.
During the period from 2001 to 2011, Korea had 1009 cases of laparoscopic liver resection, of which 265 cases involved major liver resection, including 165 patients with left hepatectomy and 53 patients with right hepatectomy. The rate of conversion to open surgery was 6.4%. The average surgical time was 399.3 minutes, with approximately 836 ml of blood loss. 24.5% of cases required intraoperative blood transfusion. Complications occurred in 20% of cases. Mortality was 0.75% [48].