This result shows the interference of compounds existing in the analyzed sample and the role of electrochemical treatment of organic compounds in the detection of Hg(II) in real samples. The electrochemical treatment conditions were also investigated. The results are presented below:
(a)
(b) | |
(c) |
(d) |
Maybe you are interested!
-
 Car body electrical practice - 8
zt2i3t4l5ee
zt2a3gs
zt2a3ge
zc2o3n4t5e6n7ts
If the voltage is out of specification, replace the wire or connector.
If the voltage is within specification, install the front fog light relay and follow step 5.
Step 5 Check the front fog light switch
- Remove the D4 connector of the fog light switch
- Use a multimeter to measure the resistance of the front fog light switch.
Measurement location
Condition
Standard
D4-3 (BFG) -D4-4 (LFG)
Light switchFront Fog OFF
>10kΩ
D4-3 (BFG) -D4-4 (LFG)
Front fog light switchON
<1 Ω
- Standard resistor
D4 connector is located on the combination switch assembly.
If the resistance is out of specification, replace the combination switch (the fog light switch is located in the combination switch).
If the resistance is within specification, follow step 6.
Step 6 Check wiring and connectors (front fog light relay-light selector switch)
- Disconnect connector D4 of the combination switch assembly
- Use a voltmeter to measure the voltage value of jack D4 on the wire side.
Measurement location
Control modecontrol
Standard
D4-3 (BFG) - (-) AQ
TAIL
11 to 14 V
D4 connector for the wiring of the combination switch assembly
If the voltage does not meet the standard, replace the wire or connector.
If the voltage is within standard, there may have been an error in the previous measurements.
Step 7 Check the front fog lights
- Remove the front fog light electrical connector.
- Supply battery voltage to the fog lamp terminals
Jack 8, B9 of front fog lamp on the electrical side
blind first.
Power supply location
Terms and Conditions
Battery positive terminal - Terminal 2Battery negative terminal - Terminal 1
Fog lightsbefore morning
- If the light does not come on, replace the bulb.
If the light is on, re-plug the jack and continue to step 8.
Step 8 Check wiring and connectors (relay and front fog lights)
- Disconnect the B8 and B9 connectors of the front fog lights.
- Use a voltmeter to measure voltage at the following locations:
Measurement location
Switch location
Terms and Conditions
B8-2 - (-) AQ
Electric lock ON TAIL size switchFog switch ON
11 to 14 V
B9-2 - (-) AQ
Electric lock ONTAIL size switch Fog switch ON
11 to 14 V
B8 and B9 connectors on the front fog lamp wiring side
Voltage is not up to standard, repair or replace the jack. If up to standard, there may have been an error in the measurement process.
2.2.4. Procedure for removing, installing and adjusting fog lights 1. Procedure for removing
- Remove the front inner ear pads
Use a screwdriver to remove the 3 screws and remove the front part of the front inner ear liner
-Remove the fog light assembly
+ Disconnect the connector.
+ Use a screwdriver to remove 3 screws to remove the fog light cover
2. Installation sequence
-Rotate the fog lamp bulb in the direction indicated by the arrow as shown in the figure and remove the fog lamp from the fog lamp assembly.
-Rotate the fog light bulb in the direction indicated by the arrow as shown in the figure and install the light into the fog light assembly.
- Use a screwdriver to install the fog light cover
-Install the electrical connector
Attention: Be careful not to damage the plastic thread on the lamp assembly.
- Install the front inner ear pads
Use a screwdriver to install the front inner bumper with 3 screws.
3. Prepare the vehicle to adjust the fog light convergence. Prepare the vehicle:
- Make sure there is no damage or deformation to the vehicle body around the fog lights.
- Add fuel to the fuel tank
- Add oil to standard level.
- Add engine coolant to standard level.
- Inflate the tire to standard pressure.
- Place spare tire, tools and jack in original design position
- Do not leave any load in the luggage compartment.
- Let a person weighing about 75 kg sit in the driver's seat.
4. Prepare to check the fog light convergence
a/ Prepare the vehicle status as follows:
- Place the car in a dark enough place to see the lines. The lines are the dividing line, below which the light from the fog lights can be seen but above which it cannot.
- Place the car perpendicular to the wall.
- Keep a distance of 7.62 m between the center of the fog lamp and the wall.
- Park the car on level ground.
- Press the car down a few times to stabilize the suspension.
Note: A distance of approximately 7.62 m is required between the vehicle (fog lamp center) and the wall to adjust the convergence correctly. If the distance of 7.62 m cannot be achieved, set the correct distance of 3 m to check and adjust the fog lamp convergence. (Since the target area varies with the distance, please follow the instructions as shown in the figure.)
b/ Prepare a piece of thick white paper about 2 m high and 4 m wide to use as a screen.
c/ Draw a vertical line through the center of the screen (line V).
d/ Set the screen as shown in the picture. Note:
- Keep the screen perpendicular to the ground.
- Align the V line on the screen with the center of the vehicle.
e/Draw the reference lines (H, V LH and V RH lines) on the screen as shown in the figure.HINT:
Mark the center of the fog lamp on the screen. If the center mark cannot be seen on the fog lamp, use the center of the fog lamp or the manufacturer's name mark on the fog lamp as the center mark.
H line (fog light height):
Draw a line across the screen so that it passes through the center mark. Line H should be at the same height as the center mark of the fog light bulb.
Line V LH, V RH (center mark position of left fog lamp LH and right fog lamp RH):
Draw two lines so that they intersect line H at the center marks.
5. Check the fog light convergence
a/ Cover the fog lamp or remove the connector of the other side fog lamp to prevent light from the unchecked fog lamp from affecting the fog lamp convergence test.
b/ Start the engine.
c/ Turn on the fog lights and make sure that the dividing line is outside the standard area as shown in the drawing.
6. Adjust the fog light convergence
Use a screwdriver to adjust the fog light to the standard area by turning the toe adjustment screw.
Note: If the screw is adjusted too far, loosen it and then tighten it again, so that the last rotation of the light adjustment screw is clockwise.
3. Self-study questions
1. Describe the operating principle of the lighting system with automatic headlight function
2. Describe the operating principle of the lighting system with the function of rotating headlights when turning
3. Draw diagram and connect lighting system on Hyundai Porter car
4. Draw diagram and connect lighting system on Honda Accord 1992
5. Draw the lighting circuit on a 1993 Toyota Lexus
LESSON 3 MAINTENANCE AND REPAIR OF SIGNAL SYSTEM
I. IMPLEMENTATION GOAL
After completing this lesson, students will be able to:
- Distinguish between types of signals on cars
- Correctly describe common symptoms and suspected areas causing damage.
- Connecting signal circuits ensures technical requirements
- Disassemble, install, check, maintain and repair the signal system to ensure technical requirements.
- Ensure safety in work and industrial hygiene
II. LESSON CONTENT
1. General description
The signal system equipped on cars aims to create signals to notify other vehicles participating in traffic about the vehicle's operating status such as: stopping, parking, braking, reversing, turning...
Signals are used either by light such as headlamps, brake lights, turn signals….. or by sound such as horns, reverse music….
Just like the lighting system. A signal system circuit usually consists of: battery, fuse, wire, relay, electrical load and control switch. Only some switches of the signal system are on the combination switch. The switches of other signals are usually located in different locations such as in the gearbox or brake pedal……
2. Maintenance and repair
2.1. Turn signals and hazard lights
The installation location of the turn signal is shown in Figure 3.1. The turn signal control switch is located in the combination switch under the steering wheel. Turning this switch to the right or left will make the turn signal turn right or left.
The hazard light switch is used when the vehicle has a problem while participating in traffic. When the hazard light switch is turned on, all the turn signals on the vehicle will light up at a certain frequency. The hazard light switch is usually placed separately from the turn signal switch (some old cars integrate the hazard and turn signal switches on the same combination switch cluster).
Figure 3.1 Turn signal switch Figure 3.2 Hazard switch
The part that generates the flashing frequency for the lights is called a turn signal relay. The turn signal relay usually has 3 terminals: B (positive power supply); E (negative power supply); L (providing the turn signal switch to distribute to the
lamp)
2.1.1. Circuit diagram
To generate the frequency for the turn signal, a turn signal relay is used in the turn signal circuit. The current from the turn signal relay will be sent to the turn signal switch assembly to distribute the current to the turn signal lights for the driver's purpose.
Figure 3.3. Schematic diagram of a turn signal circuit without a hazard switch
1. Battery; 2. Electric lock; 3. Turn signal relay; 4. Turn signal switch; 5. Turn signal lamp; 6. Turn signal lamp; 7. Hazard switch
Figure 3.4 Schematic diagram of turn signal circuit with hazard switch
1. Battery; 2. Combination switch cluster; 3. Turn signal;
4. Turn signal light; 5. Turn signal relay
Today's cars no longer use three-pin turn signal relays (B, L, E) but use eight-pin turn signal relays (figure 3.5) (pin number 8 is used for hazard lights).
For this type, the current supplying the turn signal lights is supplied directly from the turn signal relay to the lights.
div.maincontent .p { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 14pt; margin:0pt; } div.maincontent p { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 14pt; margin:0pt; } div.maincontent .s1 { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 13pt; } div.maincontent .s2 { color: black; font-family:"Times New Roman", serif; font-style: italic; font-weight: normal; text-decoration: none; font-size: 14pt; } div.maincontent .s3 { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 14pt; } div.maincontent .s4 { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 13pt; } div.maincontent .s5 { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 13pt; vertical-align: 1pt; } div.maincontent .s6 { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 11pt; } div.maincontent .s7 { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 14pt; vertical-align: -9pt; } div.maincontent .s8 { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 11pt; } div.maincontent .s9 { color: #008000; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 14pt; } div.maincontent .s10 { color: black; font-family:"Times New Roman", serif; font-style: italic; font-weight: normal; te
Car body electrical practice - 8
zt2i3t4l5ee
zt2a3gs
zt2a3ge
zc2o3n4t5e6n7ts
If the voltage is out of specification, replace the wire or connector.
If the voltage is within specification, install the front fog light relay and follow step 5.
Step 5 Check the front fog light switch
- Remove the D4 connector of the fog light switch
- Use a multimeter to measure the resistance of the front fog light switch.
Measurement location
Condition
Standard
D4-3 (BFG) -D4-4 (LFG)
Light switchFront Fog OFF
>10kΩ
D4-3 (BFG) -D4-4 (LFG)
Front fog light switchON
<1 Ω
- Standard resistor
D4 connector is located on the combination switch assembly.
If the resistance is out of specification, replace the combination switch (the fog light switch is located in the combination switch).
If the resistance is within specification, follow step 6.
Step 6 Check wiring and connectors (front fog light relay-light selector switch)
- Disconnect connector D4 of the combination switch assembly
- Use a voltmeter to measure the voltage value of jack D4 on the wire side.
Measurement location
Control modecontrol
Standard
D4-3 (BFG) - (-) AQ
TAIL
11 to 14 V
D4 connector for the wiring of the combination switch assembly
If the voltage does not meet the standard, replace the wire or connector.
If the voltage is within standard, there may have been an error in the previous measurements.
Step 7 Check the front fog lights
- Remove the front fog light electrical connector.
- Supply battery voltage to the fog lamp terminals
Jack 8, B9 of front fog lamp on the electrical side
blind first.
Power supply location
Terms and Conditions
Battery positive terminal - Terminal 2Battery negative terminal - Terminal 1
Fog lightsbefore morning
- If the light does not come on, replace the bulb.
If the light is on, re-plug the jack and continue to step 8.
Step 8 Check wiring and connectors (relay and front fog lights)
- Disconnect the B8 and B9 connectors of the front fog lights.
- Use a voltmeter to measure voltage at the following locations:
Measurement location
Switch location
Terms and Conditions
B8-2 - (-) AQ
Electric lock ON TAIL size switchFog switch ON
11 to 14 V
B9-2 - (-) AQ
Electric lock ONTAIL size switch Fog switch ON
11 to 14 V
B8 and B9 connectors on the front fog lamp wiring side
Voltage is not up to standard, repair or replace the jack. If up to standard, there may have been an error in the measurement process.
2.2.4. Procedure for removing, installing and adjusting fog lights 1. Procedure for removing
- Remove the front inner ear pads
Use a screwdriver to remove the 3 screws and remove the front part of the front inner ear liner
-Remove the fog light assembly
+ Disconnect the connector.
+ Use a screwdriver to remove 3 screws to remove the fog light cover
2. Installation sequence
-Rotate the fog lamp bulb in the direction indicated by the arrow as shown in the figure and remove the fog lamp from the fog lamp assembly.
-Rotate the fog light bulb in the direction indicated by the arrow as shown in the figure and install the light into the fog light assembly.
- Use a screwdriver to install the fog light cover
-Install the electrical connector
Attention: Be careful not to damage the plastic thread on the lamp assembly.
- Install the front inner ear pads
Use a screwdriver to install the front inner bumper with 3 screws.
3. Prepare the vehicle to adjust the fog light convergence. Prepare the vehicle:
- Make sure there is no damage or deformation to the vehicle body around the fog lights.
- Add fuel to the fuel tank
- Add oil to standard level.
- Add engine coolant to standard level.
- Inflate the tire to standard pressure.
- Place spare tire, tools and jack in original design position
- Do not leave any load in the luggage compartment.
- Let a person weighing about 75 kg sit in the driver's seat.
4. Prepare to check the fog light convergence
a/ Prepare the vehicle status as follows:
- Place the car in a dark enough place to see the lines. The lines are the dividing line, below which the light from the fog lights can be seen but above which it cannot.
- Place the car perpendicular to the wall.
- Keep a distance of 7.62 m between the center of the fog lamp and the wall.
- Park the car on level ground.
- Press the car down a few times to stabilize the suspension.
Note: A distance of approximately 7.62 m is required between the vehicle (fog lamp center) and the wall to adjust the convergence correctly. If the distance of 7.62 m cannot be achieved, set the correct distance of 3 m to check and adjust the fog lamp convergence. (Since the target area varies with the distance, please follow the instructions as shown in the figure.)
b/ Prepare a piece of thick white paper about 2 m high and 4 m wide to use as a screen.
c/ Draw a vertical line through the center of the screen (line V).
d/ Set the screen as shown in the picture. Note:
- Keep the screen perpendicular to the ground.
- Align the V line on the screen with the center of the vehicle.
e/Draw the reference lines (H, V LH and V RH lines) on the screen as shown in the figure.HINT:
Mark the center of the fog lamp on the screen. If the center mark cannot be seen on the fog lamp, use the center of the fog lamp or the manufacturer's name mark on the fog lamp as the center mark.
H line (fog light height):
Draw a line across the screen so that it passes through the center mark. Line H should be at the same height as the center mark of the fog light bulb.
Line V LH, V RH (center mark position of left fog lamp LH and right fog lamp RH):
Draw two lines so that they intersect line H at the center marks.
5. Check the fog light convergence
a/ Cover the fog lamp or remove the connector of the other side fog lamp to prevent light from the unchecked fog lamp from affecting the fog lamp convergence test.
b/ Start the engine.
c/ Turn on the fog lights and make sure that the dividing line is outside the standard area as shown in the drawing.
6. Adjust the fog light convergence
Use a screwdriver to adjust the fog light to the standard area by turning the toe adjustment screw.
Note: If the screw is adjusted too far, loosen it and then tighten it again, so that the last rotation of the light adjustment screw is clockwise.
3. Self-study questions
1. Describe the operating principle of the lighting system with automatic headlight function
2. Describe the operating principle of the lighting system with the function of rotating headlights when turning
3. Draw diagram and connect lighting system on Hyundai Porter car
4. Draw diagram and connect lighting system on Honda Accord 1992
5. Draw the lighting circuit on a 1993 Toyota Lexus
LESSON 3 MAINTENANCE AND REPAIR OF SIGNAL SYSTEM
I. IMPLEMENTATION GOAL
After completing this lesson, students will be able to:
- Distinguish between types of signals on cars
- Correctly describe common symptoms and suspected areas causing damage.
- Connecting signal circuits ensures technical requirements
- Disassemble, install, check, maintain and repair the signal system to ensure technical requirements.
- Ensure safety in work and industrial hygiene
II. LESSON CONTENT
1. General description
The signal system equipped on cars aims to create signals to notify other vehicles participating in traffic about the vehicle's operating status such as: stopping, parking, braking, reversing, turning...
Signals are used either by light such as headlamps, brake lights, turn signals….. or by sound such as horns, reverse music….
Just like the lighting system. A signal system circuit usually consists of: battery, fuse, wire, relay, electrical load and control switch. Only some switches of the signal system are on the combination switch. The switches of other signals are usually located in different locations such as in the gearbox or brake pedal……
2. Maintenance and repair
2.1. Turn signals and hazard lights
The installation location of the turn signal is shown in Figure 3.1. The turn signal control switch is located in the combination switch under the steering wheel. Turning this switch to the right or left will make the turn signal turn right or left.
The hazard light switch is used when the vehicle has a problem while participating in traffic. When the hazard light switch is turned on, all the turn signals on the vehicle will light up at a certain frequency. The hazard light switch is usually placed separately from the turn signal switch (some old cars integrate the hazard and turn signal switches on the same combination switch cluster).
Figure 3.1 Turn signal switch Figure 3.2 Hazard switch
The part that generates the flashing frequency for the lights is called a turn signal relay. The turn signal relay usually has 3 terminals: B (positive power supply); E (negative power supply); L (providing the turn signal switch to distribute to the
lamp)
2.1.1. Circuit diagram
To generate the frequency for the turn signal, a turn signal relay is used in the turn signal circuit. The current from the turn signal relay will be sent to the turn signal switch assembly to distribute the current to the turn signal lights for the driver's purpose.
Figure 3.3. Schematic diagram of a turn signal circuit without a hazard switch
1. Battery; 2. Electric lock; 3. Turn signal relay; 4. Turn signal switch; 5. Turn signal lamp; 6. Turn signal lamp; 7. Hazard switch
Figure 3.4 Schematic diagram of turn signal circuit with hazard switch
1. Battery; 2. Combination switch cluster; 3. Turn signal;
4. Turn signal light; 5. Turn signal relay
Today's cars no longer use three-pin turn signal relays (B, L, E) but use eight-pin turn signal relays (figure 3.5) (pin number 8 is used for hazard lights).
For this type, the current supplying the turn signal lights is supplied directly from the turn signal relay to the lights.
div.maincontent .p { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 14pt; margin:0pt; } div.maincontent p { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 14pt; margin:0pt; } div.maincontent .s1 { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 13pt; } div.maincontent .s2 { color: black; font-family:"Times New Roman", serif; font-style: italic; font-weight: normal; text-decoration: none; font-size: 14pt; } div.maincontent .s3 { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 14pt; } div.maincontent .s4 { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 13pt; } div.maincontent .s5 { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 13pt; vertical-align: 1pt; } div.maincontent .s6 { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 11pt; } div.maincontent .s7 { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 14pt; vertical-align: -9pt; } div.maincontent .s8 { color: black; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 11pt; } div.maincontent .s9 { color: #008000; font-family:"Times New Roman", serif; font-style: normal; font-weight: normal; text-decoration: none; font-size: 14pt; } div.maincontent .s10 { color: black; font-family:"Times New Roman", serif; font-style: italic; font-weight: normal; te -
 Comparison of the effects of two methods of complete intravenous anesthesia with Propofol with and without target concentration control - 2
Comparison of the effects of two methods of complete intravenous anesthesia with Propofol with and without target concentration control - 2 -
 When Should a Healthy Child Get the First Dose of Hepatitis B Vaccine? (Check Only One)
When Should a Healthy Child Get the First Dose of Hepatitis B Vaccine? (Check Only One) -
 Two Elements A And B Are In Two Consecutive Main Groups In The Periodic System. In Their Pure Substance State, A And B Do Not React With Each Other. Total Number Of Protons
Two Elements A And B Are In Two Consecutive Main Groups In The Periodic System. In Their Pure Substance State, A And B Do Not React With Each Other. Total Number Of Protons -
 Equations Expressing the Relationship Dt/d1,3 In the Form: Dt = A + B.d1,3 At Different Ages
Equations Expressing the Relationship Dt/d1,3 In the Form: Dt = A + B.d1,3 At Different Ages
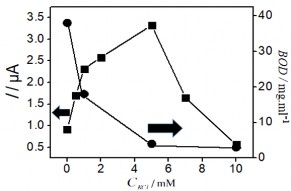
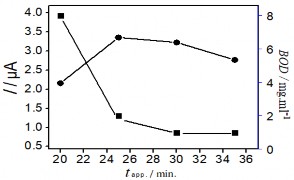
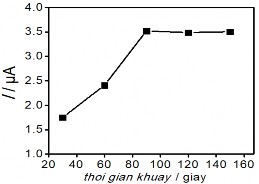
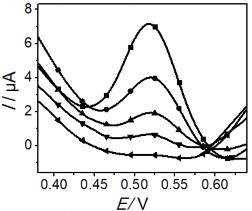
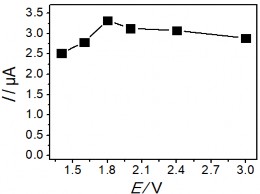
Figure 3.41. Effects of voltage (a); KCl concentration (b); Electrolysis time (c); and stirring time (d) on the Hg(II) signal
The obtained results show that initially the Hg (II) current signal variation in the figure
3.41 a, b, c corresponding to voltage, KCl concentration and electrolysis time all increase to a plateau, where the current signal obtained is the largest. In contrast, Figure 3.41 b, c shows a significant reduction in BOD indicating a decrease in the amount of organic compounds in the sample. This result shows the role of electrochemical treatment to eliminate the influence of organic compounds in the determination of Hg(II). After that, the peak current tends to decrease gradually past the plateau when continuing to increase voltage, KCl concentration and electrolysis time. The voltage for electrolysis to occur reaches a maximum value of +1.8 V, further increase in voltage may affect the electrolysis efficiency. When the KCl concentration is greater than 5 mM, the Hg(II) current signal decreases rapidly indicating the influence of the oxidant
to the SAM surface. The Au-S bond is easily oxidized under ambient conditions, and adversely affects the quality and structure of the SAM [187]. This result is also consistent with the study of another author [188]. Increasing the electrolysis time to 25 minutes leads to a decreasing trend in the current signal, so 25 minutes is enough time to oxidize the substances in the sample. Figure 3.41d examines the stirring time, which is the period of time that the solution is stirred at a rotation speed of 60 rpm after the end of the electrolysis process. The variation of the Hg (II) current signal shows that extending the time to 80 minutes is the optimal condition, sufficient to remove oxidized organic compounds in the real sample. The analysis results of Hg (II) were determined by the standard addition method. The height of the Hg (II) peak was determined from the voltammetric spectrum as shown below:
|
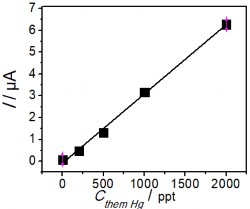
Figure 3.42. Standard addition graph (left) and DPASV curve (right) for Hg determination
(II) in treated To Lich river water samples
Figure 3.42 is the DPASV spectrum using PET-AET-SAM/AuNP + GO electrode to determine Hg(II) in To Lich river water sample with different concentrations. In this figure, it can be observed that the peak appeared at 0.52 V and the peak current gradually increased when adding Hg(II) standard solution. The results obtained have a good linear correlation between the current signal and concentration with correlation coefficient R=0.996 and the Hg(II) content was determined to be 24 ppt.
The results obtained showed that it is possible to treat real samples containing organic compounds under optimal conditions and using SAM modified electrodes to determine Hg(II) at trace levels.
CONCLUDE
The studies in this thesis focused on the modification of the glassy carbon electrode (GCE) surface using nanomaterials such as spherical gold particles (AuNP), graphene oxide (GO) with 4-pyridine ethanethiol (PET) and 2-aminoethanethiol (AET) to create a self-assembled monolayer (SAM), to improve the sensitivity and selectivity for the analysis of trace amounts of Hg(II) in water samples. The main results include:
1. Successfully fabricated AuNP coating as well as AuNP-GO composite on GCE surface by electrochemical method as substrate material for modification with PET and AET. The results showed that the presence of AuNP was linked with PET and AET molecules, as well as the presence of GO increased the electrochemically active surface area of the modified electrode to 26.98%.
2. The use of electrodes modified with single-component SAMs PET, AET as well as two-component SAMs (PET-AET) was studied to increase the selectivity for Hg(II) during the enrichment process. The differential pulse voltammetry results showed that the peak current height corresponding to the Hg(II) signal ( I HgII ) increased significantly for the PET-AET-SAM/AuNP-GO modified electrodes, respectively: 0.3 A (AET-SAM/AuNP), 1.0 A (PET- SAM/AuNP), 1.80 A (PET-AET-SAM/AuNP), 3.50 A (PET-AET- SAM/AuNP+GO). The Hg(II) detection limit of the PET-AET- SAM/AuNP+GO modified electrode was 18.99 ppt, much smaller than the values of the PET-SAM/AuNP and PET-AET-SAM/AuNP modified electrodes which were 38.42 ppt and 31.97 ppt, respectively.
3. The ability to increase the sensitivity of Hg(II) detection for PET-AET- SAM/AuNP-GO modified electrodes was studied due to not only the increase in electrochemically active surface area due to the presence of GO on the substrate material, but also the change in orientation of PET, AET molecules when GO is present on the surface thanks to hydrogen bonding between functional groups as well as interactions between electrons of the benzene ring. Thanks to that, PET-AET- SAM on the surface of AuNP-GO composite has a stable structure and increases the ability to bind with Hg(II) at functional groups.
4. The advantage of the PET-AET-SAM/AuNP+GO modified electrode is also shown in the increase in the linear range between the peak current and the Hg(II) concentration. This range is 30 ppt – 3500 ppt with a correlation coefficient R 2 = 0.999 compared to 100 ppt – 1000 ppt and 50
ppt – 2000 ppt for PET-SAM/AuNP, PET-AET- SAM/AuNP modified electrodes respectively. Furthermore, the number of uses of PET-AET-SAM/AuNP- GO modified electrode for Hg(II) analysis was up to 112 times with a repeatability of RSD 5.19% compared to 30 times and 51 times for PET-SAM/AuNP and PET-AET- SAM/AuNP modified electrodes respectively.
5. The applicability of the modified electrode has also been carried out for some seawater and industrial wastewater samples. The results obtained have acceptable accuracy compared with the atomic absorption method, showing that the modified electrode can be used for the analysis of trace amounts of Hg(II) in some environmental samples.
NEW CONTRIBUTIONS OF THE THESIS
1. AuNP and AuNP-GO composite electrodes modified with self-assembled monolayers (SAMs) of 2-aminoethanethiol hydrochloride (AET) and 4-pyridine ethanethiol hydrochloride (PET) were fabricated.
2. The fabricated electrodes showed that the AuNP-GO electrode modified by two components PET and AET had the best ability to determine Hg (II), with a detection limit of 18.99 ppt, a linear range of 30 ppt – 3500 ppt with a correlation coefficient of 0.999.
LIST OF PUBLISHED WORKS RELATED TO THE THESIS
1. Dang Thanh Huyen, Pham Thi Hai Yen, Nguyen Hoang Anh, Pham Hong Phong. Initial application of nanostructured electrochemical sensor to determine trace amounts of mercury in seawater. Journal of Chemistry, 52 (6A), 181-185, 2014.
2. Pham Hong Phong, Dang Thanh Huyen, Nguyen Hoang Anh, Vu Thi Thu Ha. Effects of Introducing 2-aminoethanethiol into 4-pyridineethanethiol Self-assembled Monolayer Applicable to Enhance Sensitivity of Hg(II) Electrochemical Analysis, Journal of New Materials for Electrochemical System, 18, 207-212, 2015.
3. Dang Thanh Huyen, Vu Duc Loi, Pham Hong Phong. Study of an electrochemical behaviour of modified binary self-assembled monolayers for enhancing sensitivity of Hg(II) detection. Journal of Chemistry, 54(2), 223-227, 2016.
4. Dang Thanh Huyen, Pham Hong Phong. Study of 4-pyridineethanthiol modified composite graphene oxide-AuNPs electrode for inhancing the sensitivity of Hg(II) determination, Journal of Chemistry, 54(6e2), 160-164, 2016.
5. Dang Thanh Huyen, Vu Duc Loi, Vu Thi Thu Ha, Pham Hong Phong. Effect elimination of organic compounds by electrolysis to enhance the electrochemical signal of Hg (II) determination using two-component self-assembled thin layer modified the composite on glassy carbon electrode. Journal of Chemistry, 56(4e), 136- 140, 2018.
6. Pham Hong Phong, Dang Thanh Huyen, Vu Duc Loi, Vu Thi Thu Ha, Nguyen Thi Cam Ha, Nguyen Ngoc Ha, Le Minh Thanh. Conformational changes in self- assembled monolayer of 4-pyridineethanethiol and 2-aminoethanethiol on the gold nanoparticles-graphene oxide composite and advantages in Hg(II) determination. Journal of Nanoparticle Research, 21,159, 2019.
REFERENCES
1. Guidelines for drinking-water quality, fourth edition , World health organization, (2011).
2. MS Jeoung & HS Choi, Spectrophotometric determination of trace Hg(II) in cetyltrimethylammonium bromide media, Bulletin of the Korean Chemical Society, 25 (12), 1877-1880, (2004).
3. EK Pavlos & GKK Nikolaos, Selective mercury determination after membrane complexation and total reflection X-ray fluorescence analysis, , Analytical Chemistry, 76 (15), 4315-4319, (2004).
4. JM Ombaba, Total Mercury determination in Biological and Environmental Standard Samples by Gold Amalgamation Followed by Cold Vapor Atomic Absorption Spectrometry, Micro. Chem. J, 53 (2), 195-200, (1996).
5. P. Jones J. Murphy, SJ Hill, Determination of total mercury in environmental and biological samples by flow injection cold vapor atomic absorption spectrometry, Spectrochimica Acta Part B: Atomic Spectroscopy, 51 (14), 1867-1873, (1996) .
6. H. Bagheri & A. Gholami, Determination of very low levels of dissolved mercury(II) and methylmercury in river waters by continuous flow with on- line UV decomposition and cold-vapor atomic fluorescence spectrometry after pre-concentration on a silica gel-2-mercaptobenzimidazol sorbent, Talanta, 55 (6), 1141-1150, (2001).
7. S. Zampieri P. Ugo, L.M. Moretto, Determination of mercury in process and lagoon waters by inductively coupled plasma-mass spectrometric analysis after electrochemical preconcentration: comparison with anodic stripping at gold and polymer coated electrodes, Analytica Chimica Acta, 434 (2 ), 291- 300, (2001).
8. AC Barbosa & GA East GR Boaventura, Multivessel system for cold-vapor mercury generation determination of mercury in hair and fish, Biological Trace Element Research, 60 (1-2), 153-161, (1997).
9. M. de la Guárdia JS dos Santos, A. Pastor, Determination of organic and inorganic mercury species in water and sediment samples by HPLC on-line coupled with ICP-MS, Talanta, 80 (1), 207-211, ( 2009).
10. Z. Cheng T. Zhang, Y. Wang, Z. Li, C. Wang, Y. Li, Y Fang, Self-assembled 1-octadecanethiol monolayers on graphene for mercury detection, Nano Lett, 10 (11), 4738 -4741, (2010).
11. Giovana L. Okajima Newton L. Dias Filho, Geovanna Pires, Voltammetry of Mercury(II) Based on an Organo-Clay Modified Graphite Electrode, Portugaliae Electrochimica Acta, 26 163-179, (2008).
12. Antje Widmann & Constant MG van den Berg, Mercury Detection in Seawater Using a Mercaptoacetic Acid Modified Gold Microwire Electrode, Electroanalysis, 17 (10), 825-831, (2005).
13. Nor Azah Yusof Nurulhaidah Daud, Tan Wee Tee, Development of Electrochemical Sensor for Detection of Mercury by Exploiting His-Phe-His- Ala-His-Phe-Ala-Phe Modified Electrode, Int. J. Electrochemistry. Sci., 6 2798 - 2807, (2011).
14. Vu Duc Loi, Research on determining some forms of mercury in biological and environmental samples, PhD thesis, Institute of Chemistry, Vietnam Academy of Science and Technology, 2008).
15. Phung Thi Thu Huyen, Determination of trace amounts of mercury by optical solid phase extraction method , Master's thesis, Place of test, University of Science, Vietnam National University, Hanoi, (2012).
16. Trinh Xuan Gian & Tong Thi Thanh Thuy Duong Thi Tu Anh, Research on simultaneous determination of trace As(III) and Hg(II) content by anodic stripping voltammetry method using gold membrane electrode, Journal of Analytical Chemistry, Physics and Biology, 14 (4), 44-48, (2009).
17. VTT Ha PTH Yen, PK Duy, VH Dang, Investigation of electrochemical properties of homemade nano gold electrodes and application in determination of Hg (II) at the trace levels, Vietnam Journal of Chemistry, 53 (5), 657).
18. K.; Morikawa Lee, Y.; Langreth, DC , Adsorption of n-Butane on Cu(100), Cu(111), Au(111), and Pt(111): van der Waals Density Functional Study, Phys. Rev., 82 155461, (2010b).
19. Apparao Chaitanya Kumar, Yakub, Corrosion protection of copper by self-assembled monolayers, Indian Journal of Chemical Technology, 16 (1), 25- 31, (2009).
20. El-Ads EH Atta NF, Galal A, Self-Assembled Monolayers on Nanostructured Composites for Electrochemical Sensing Applications , (2016).
21. RW Cernosek, Palladium as a Substrate for Self-Assembled Monolayers Used in Biotechnology, Anal. Chem, 76 (20), 6116-6121, (2004).
22. M.Osińska T.Łuczak, New self-assembled layers composed with gold nanoparticles, cysteamine and dihydrolipoic acid deposited on bare gold template for highly sensitive and selective simultaneous sensing of dopamine in the presence of interfering ascorbic and uric acids, Solid State Electrochemistry , 21 (3), 747-758, (2016).
23. MA Hines A. Dhirani, AJ Fisher, O. Ismail, and P. Guyot-Sionnest, Structure of Self-Assembled Decanethiol on Ag(111): A Molecular Resolution Scanning Tunneling Microscopy Study, Langmuir, 11 (7), 2609 -2614, (1995).
24. Zutphen Wöhrmann Print Services, The Netherlands, Self-assembled monolayers on metal oxides : applications in nanotechnology , (2010).
25. Christine L.; Diehl McGuiness, Gregory A.; Blasini, Daniel; Smilgies, D.M.; Zhu, M.; Samarth, Nitin; Weidner, Tobias; Ballav, Nirmalya; Zharnikov, Michael; Allara, David L., Molecular self-assembly at bare semiconductor surfaces: Cooperative substrate-molecule effects in octadecanethiolate monolayer assemblies on GaAs(111), (110), and (100), ACS Nano, 4 (6), 3447-3465 , (2010).
26. Lara A. Estroff J. Christopher Love, Jennah K. Kriebel, Ralph G. Nuzzo, George M. Whitesides Self-Assembled Monolayers of Thiolates on Metals as a Form of Nanotechnology, Chem. Rev., 105 1103-1169, (2005).
27. PA Lewis RK Smith, PS Weiss, Patterning self-assembled monolayers,
Prog. Surf. Sci, 75 1-68, (2004,).