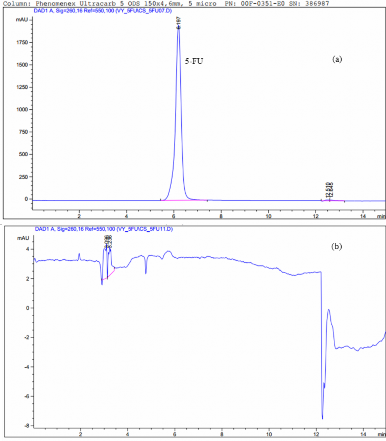
Figure 3.31. HPLC spectrum of 5-FU (a) and HPLC spectrum of PNS-GPTMS-CS-mPEG system carrying released 5-FU (b)
General comments
Here we can see that the silica nanoparticles after being modified with Chitosan (PNS-GPTMS-CS- mPEG) have a DOX drug loading efficiency of 71.75 ± 1.89 and 5-FU of 70.04 ± 2.60 %, higher than the unmodified PNS system, which contributes to affirming the significance of modifying porous nano silica to increase better drug loading efficiency.
During the release study, DOX was released well in pH 4.5 (an environment similar to cancer cells) and released little in pH 7.4 (a healthy cell environment). However, 5-FU was not released from the material. Therefore, this modified system is more suitable for DOX release than 5-FU.
3.3.3.3. DOX drug loading results of PNS-GPTMS-CS-mPEG
By determining the amount of drug that has not been carried, we can easily analyze the drug loading efficiency and drug capacity of the particle. After measuring the amount of excess drug that has not been carried by UV-Vis, we have the following results:
Table 3.10. Measurement results of free DOX of the PNS-GPTMS-CS-mPEG system
STT
Time | Mean free drug content (mg) | |
1 | 6 hours | 0.33 ± 0.02 |
2 | 12 hours | 0.10 ± 0.03 |
3 | 24 hours | 0.05 ± 0.01 |
Maybe you are interested!
-
 1 H, 13 C-Nmr Spectrum Data Of At3 (Cd 3 Od, 500/125Mhz)
1 H, 13 C-Nmr Spectrum Data Of At3 (Cd 3 Od, 500/125Mhz) -
 De Gruyter, C., Currie, G., Rose, G. (2017). Sustainability Measures Of Urban Public Transport In Cities: A World Review And Focus On The Asia/middle East Region . Sustainability, Vol. 9, No. 1,
De Gruyter, C., Currie, G., Rose, G. (2017). Sustainability Measures Of Urban Public Transport In Cities: A World Review And Focus On The Asia/middle East Region . Sustainability, Vol. 9, No. 1, -
 Epidemiology of autism spectrum disorder in children aged 18 - 30 months and barriers to accessing autism spectrum disorder diagnosis and intervention services in Vietnam, 2017 - 2019 - 2
Epidemiology of autism spectrum disorder in children aged 18 - 30 months and barriers to accessing autism spectrum disorder diagnosis and intervention services in Vietnam, 2017 - 2019 - 2 -
 Esi-Ms-Negative Spectrum of Ph11
Esi-Ms-Negative Spectrum of Ph11 -
 1H Spectrum of Cde4 (Myricitrin)
1H Spectrum of Cde4 (Myricitrin)
The amount of free drug is 0.48 (mg), so the amount of drug carried in the PNS-GPTMS-CS-mPEG system is 1.7 (mg). From there, we can calculate the drug loading efficiency and drug carrying capacity of the carrier.
Table 3.11. Comparison of DLE and DLC data of PNS-GPTMS-CS-mPEG and PNS
Sample
DLE(%) | DLC(%) | |
PNS-GPTMS-CS-mPEG with DOX drug | 71.75 ± 1.89 | 12.55 ± 0.27 |
PNS carrying DOX drug | 23.01 ± 2.16 | 2.09 ± 0.32 |
Comment:
Here we can see that the nano silica particles after being modified with chitosan have a higher drug loading efficiency and drug storage capacity of 71.75 ± 1.89% of the modified material compared to the unmodified nano silica material systems and the previously modified system. This good result is due to the imine bond of the drug DOX and the carrier PNS-GPTMS-CS- mPEG[22].
3.3.3.4. Investigation of the DOX drug release ability of the PNS-GPTMS-CS-mPEG system
After examining the DOX release ability of the PNS-GPTMS-CS-mPEG carrier in two neutral and acidic environments, we have the following data table:
Table 3.12. Data showing the drug release ability of DOX of the PNS-GPTMS-CS- mPEG system at pH=7.4 and pH=4.5
Release time (hours)
Release rate (%) | ||
pH = 7.4 | pH = 4.5 | |
0 | 0 | 0 |
1 | 5.18 | 21.47 |
2 | 9.38 | 35.65 |
3 | 11.92 | 39.62 |
6
14.30 | 47.43 | |
12 | 16.00 | 62.12 |
24 | 18.55 | 66.62 |
48 | 23.86 | 67.91 |
72 | 29.21 | 69.26 |
84 | 30.30 | 70.04 |
96 | 30.22 | 70.65 |
- In the first 6 hours, the seeds released 47.43% of the drug carried in the seeds in a pH 4.5 environment. Also in the first 6 hours, but in a pH 7.4 environment, the amount of released drug was only 14.3%. So the initial results show that the drug releases well in a pH 4.5 environment (similar to the pH of cancer cells).
- In the period from 6 to 96 hours, the granules released 70.65% at pH 4.5 while at pH 7.4 it was 30.22%. We can see that the drug released less in pH 7.4 conditions (an environment similar to the pH of healthy cells).
- The results of table 3.12 show that at pH = 4.5, the silica system modified with chitosan releases DOX well, especially from 3 to 24 hours. This has great practical significance in drug release in cancer cells.
- Results 3.12 and figure 3.35 clearly show that in the pH 7.4 environment, the modified silica system released relatively little drug, 30.22% of the drug, less than the amount of drug released by the system in the pH 4.5 environment. This data contributes to proving that the drug carrier system will be less toxic to the healthy cell environment.

Figure 3.32. Graph describing the DOX drug release ability of the PNS-GPTMS-CS-mPEG system
Comment
With the research results, we can see that in the pH 7.4 environment after 96 hours of release, the amount of drug released is 44.43% less than in the pH 4.5 environment. And compared to all systems, the PNS-GPTMS-CS-mPEG system is considered the best drug retention system when circulating in the pH 7.4 environment (the environment of healthy cells).
3.3.4. Loading and release results of PNS-APTES (drug carrier 3)
3.3.4.1. 5-FU loading results of PNS-APTES
Similar to the method of carrying 5-FU drug performed with unmodified porous silica nanomaterial, we have the following results:
Table 3.13. Mass of free drug and drug carried in 60mg of PNS-APTES carrier
Sample
Free drug content (mg) | Amount of drug carried (mg) | |
PNS-APTES | 12.07 | 2.93 |
From the data, the drug loading efficiency and drug capacity of the carrier can be calculated.
Table 3.14. Drug loading efficiency and 5-FU drug loading capacity of modified porous silica nanoparticles (PNS-APTES) compared with unmodified PNS
Sample
DLE(%) | DLC(%) | |
PNS-APTES carrying 5-FU | 19.53 ± 1.20 | 4.65 ± 0.19 |
PNS with 5-FU | 15.60 ± 1.85 | 3.75 ± 0.12 |
Although the drug carrying efficiency increased slightly due to the electrostatic interaction between the drug group and the modified carrier, the efficiency was still not high, so further modification was needed. In addition, when releasing 5-FU, it was tightly held in the PNS-APTES carrier and could not be released in the two buffer environments.
3.3.4.2. 5-FU release results of PNS-APTES
After carrying the drug, we also released 5-FU within 48 hours with 2 environments: phosphate buffer solution (PBS) (pH 7.4) and acetate buffer (pH 4.5). The HPLC results (Figure 3.36 b) showed that the amount of drug released was 0 (mg).
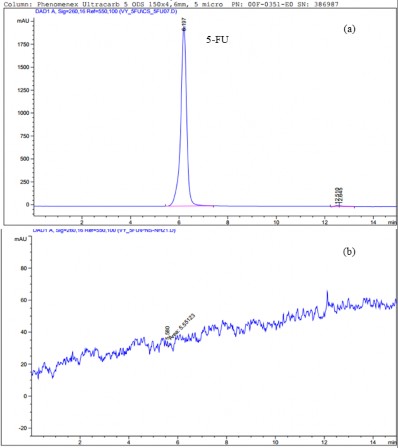
Figure 3.33. HPLC spectrum of 5-FU (a) and HPLC spectrum of PNS-APTES system carrying 5-FU
release (b)
Explain
With the method of carrying 5-FU tightly compressed into the material as presented in section 3.3.1.5 , the drug carrying efficiency will increase but the drug is tightly held in the material so it cannot be released.
3.3.4.3. DOX loading results of PNS-APTES
Calculating the efficiency and drug carrying capacity of PNS-APTES through the presentation in section 3.3.1.4, we get the following results: DLE = 54.03 ± 2.41 (%) and DLC = 7.40 ± 0.43 (%).
Here we see that the performance and drug carrying capacity of nano silica particles after being modified with PNS-APTES is slightly improved compared to the unmodified nanoporous material (PNS), which is due to the DOX drug forming a chemical bond with the carrier, which contributes to affirming the significance of nano silica modification [90]. However, to further increase the drug carrying efficiency, we continue to modify the nanoporous silica system with higher functional groups.
3.3.4.4. DOX release results of PNS-APTES
PNS-APTES carrier was investigated for drug release in 96 hours at pH 7.4 and 4.5 (Table 3.15 and Figure 3.37).
Table 3.15. DOX release data of PNS-APTES at pH 7.4 and 4.5
Release time (hours)
Release rate (%) | ||
pH = 7.4 | pH = 4.5 | |
0 | 0 | 0 |
1 | 7.69 | 32.79 |
2 | 13.06 | 43.11 |
3 | 18.03 | 53.80 |
6 | 26.95 | 61.37 |
12 | 31.31 | 71.20 |
24 | 37.43 | 77.08 |
48 | 48.69 | 79.20 |
72 | 54.15 | 80.09 |
84 | 54.85 | 80.28 |
96 | 54.86 | 80.32 |

Figure 3.34. Graph describing the release capacity of PNS-APTES For pH 7.4 environment:
During the first 6 hours, the drug release rate was slow and the amount of drug released was not much.
- In the first hour of the study, samples were taken three times and the average amount of drug released was 7.69%.
- At 6:00 to 12:00, the study also took samples 3 times and measured the amount of drug released as 31.31% of the drug carried.
- At 72 hours, the total amount of drug released was 54.15% of the amount of drug carried. It is clearly seen that compared to previous times, the drug release process began to weaken, satisfying the desired condition of slow release.
For pH 4.5 environment:
During the first 6 hours, the release rate of the system was slow, and the amount of drug released by the system was quite large compared to the 7.4 system.
- In the first hour of the study, samples were taken three times and the average amount of drug released was 32.79%.
- At 6:00 to 12:00, the study also took samples 3 times with the amount of drug released by the system being 71.20 % of the amount of drug carried. At this time, the amount of drug released by the system was quite good.
- At 72 hours, the total amount of drug released was 80.10% of the amount of drug carried. It is clear that, compared to previous times, the release process began to increase slowly and was highest at 96 hours at 80.32.
After examining the DOX release of the PNS-APTES system, the data showed that in the pH 7.4 environment, the APTES-modified silica system released the drug very weakly, only 54.85% after 96 hours, less than the amount of drug released by the system in the pH 4.5 environment, which was 80.32% at 96 hours. So the condition is good release in the poor pH condition of pH 4.5 (similar to the pH of cancer cells) and low release in the pH 7.4 environment (similar to the pH of healthy cells).
3.3.5. Results of PNS-APTES-Succinic Anhydride-Gelatin loading and release (PNS- APTES-COOH-GE is the drug carrier 4)
3.3.5.1. Result of 5-FU loading of PNS-APTES-COOH-GE
To determine the mass of the drug in the sample, it is necessary to use the HPLC measurement method and the established standard curve to determine the drug content in two solutions A (the solution before carrying the drug) and solution B (the solution after carrying the drug). After obtaining the concentration of 5-FU in the two solutions, we can calculate the mass of the drug, called m A and m B. Thereby, the mass of the drug contained in the particle is calculated as follows m A - m B. The initial mass of the drug is m A and
The total mass of sample and drug in the sample will include the mass of drug in the sample and the initial 50mg of sample. The measurement results are as follows:
Table 3.16. Measurement results of solutions A and B (before and after carrying 5-FU drug of the PNS-APTES-Succinic Anhydride-GE system)
Solution
Peak area (mAU*s) | Concentration (ppm) | Mass (mg) | |
A | 40256.2 | 1256,443 | 13,8209 |
B | 35232.3 | 1099,598 | 10,9960 |
From the above results, we will determine the two values of DLE and DLC using the formula in section 3.3.1.4.
DLE (%) = 20.44 (%) and DLC (%) = 5.35 (%).
After conducting release experiments in two different environments and HPLC measurements, it was found that the amount of 5-FU that was stuck in the carrier could not be released in both environments, pH 7.4 and pH 4.5.
Comment
Although the modified PNS-APTES-COOH-GE system has a higher 5-FU drug loading efficiency than the modified PNS system, it is still lower than the 5-FU drug loading efficiency of the PNS-GPTMS-Hydrazine and PNS-GPTMS-CS-mPEG systems, because the modified Gelatin system has a large structure that can hinder the electrostatic interaction of the 5-FU drug and the carrier.
3.3.5.2. 5-FU release results of PNS-APTES-COOH-GE
Similar to the previous modified systems carrying 5-FU, the PNS-APTES-COOH-GE system also carries 5-FU by adding 60 mg of carrier into a dropper, then pouring a solution containing 15 mg of 5-FU through, collecting it below and letting it flow through the tube many times. Next, the dialysis measures the drug carried and released in 1 hour, 2 hours, 3 hours with 2 environments of phosphate buffer solution (PBS) (pH 7.4) and acetate buffer (pH 4.5). The HPLC results show that the amount of drug released is 0 (mg). So 5-FU was tightly held in PNS-APTES-COOH- GE and could not be released in the 2 buffer environments.





