contact. In which the metal is heated to a temperature higher than its melting point. The metal droplets that appear after being melted are covered on the electrode surface . Between the liquid metal and the gas environment, diffusion occurs and the physical and chemical effects on each other such as surface stress, internal energy, temperature and thermal conductivity, and shrinkage affect the internal structure and other properties of the liquid metal.
During the melting process, some chemical components in the metal also burn.
liquid
The growth of some molten metal flowing from the tip of the wire exists until the dynamic force of the compressed air stream is greater than the surface stress of the molten metal, which will separate the metal droplets . The metal droplets under the pressure of the compressed air stream will separate into many small particles, these particles create metal spray.
The entire process of flow and dispersion occurs very rapidly. The dispersion lasts only about 1/10,000 to 1/100,000 of a second. And every second about 7,000 metal droplets are produced. The shape of the particles that emerge from the separation of metal droplets depends on the type of metal. This can be explained as follows:
Maybe you are interested!
-
 Research on the fabrication and gas sensitivity of heterostructures between SnO2 nanowires and some semiconducting metal oxides - 16
Research on the fabrication and gas sensitivity of heterostructures between SnO2 nanowires and some semiconducting metal oxides - 16 -
 Impact of Improper Solid Waste Disposal
Impact of Improper Solid Waste Disposal -
 Study on fabrication and gas sensitivity of heterostructure between SnO2 nanowires and some semiconducting metal oxides - 17
Study on fabrication and gas sensitivity of heterostructure between SnO2 nanowires and some semiconducting metal oxides - 17 -
 Theoretical mechanics of Metal cutting profession - Vocational college Part 2 - General Department of Vocational Training - 9
Theoretical mechanics of Metal cutting profession - Vocational college Part 2 - General Department of Vocational Training - 9 -
 Proportion of Highly Liquid Assets Before and After Deducting Loans from State Bank and Commercial Banks in the Market of Some Commercial Banks
Proportion of Highly Liquid Assets Before and After Deducting Loans from State Bank and Commercial Banks in the Market of Some Commercial Banks
Each grain, in addition to metal, also contains a certain percentage of oxides, which can be divided into two types:
The liquid surrounds the grain and forms a spherical grain – for example in steel.
Solids freeze very quickly and liquid metal droplets cannot form a sphere – for example aluminium and zinc.
1.3.2.2 Flight process of particles
The entire flight process of the particles, from the time they are separated from the metal droplet to the time they impact on the metal spray surface, occurs very briefly, about 0.002÷0.008 seconds. During the flight of the particles, oxidation mainly occurs, so the metal spray particles are covered with an oxide layer, which will grow with the flight distance.
The molten metal particles move in a compressed air stream at very high speeds. In addition, the particles are affected by many factors, which manifest themselves in heterogeneous reactions . When spraying metal , we must take into account the following issues:
Metals separate in liquid or solid state.
The spray elements are constantly changing their flight speed in the acceleration field.
The particles always react with the surrounding environment containing oxygen, nitrogen, hydrogen, water vapor.
and other chemical components.
The solubility of a gas depends on the temperature as well as the partial pressure of the gas.
1.3.2.3 Formation of metal coating
The process of forming a coating by metal spraying is relatively complicated. Based on the results of many experiments and calculations, it has been determined that: metal particles during impact on the sprayed surface are in a liquid state and are greatly deformed.
To understand the formation of coatings, one must pay attention to the phenomena that occur when particles collide.
element onto the metal surface (base material) to be sprayed.
The first problem is that the kinetic energy of the particles impacting the sprayed surface causes a very fast and strong impact force and deformation. This kinetic energy is determined by the speed of the particles and their mass Ek = 1.2 mV2. Therefore, particles of different sizes will have different kinetic energies (when they have the same speed ). The flight speed of the particles is a factor in determining the deformation of the particles . Arnold calculated the speed required for some metals when impacting the sprayed surface according to the following equation:
mV 2
2 427
C t
2 t 1 S
(1.2)
Here m=G/g. When G=1g then the velocity required for shattering on impact is:
C t 2t 1 S
V 91
m - mass of spray particles (g). V - velocity of particles during impact (ms) C - specific heat (cal.g -1°C-1).
(1.3)
(°C)
t1 - temperature of metal element at the moment of impact on the surface of the part
t2 - melting temperature of metal elements (°C)
S - latent heat (cal)
The second issue that needs to be considered in determining the deformation of the elements is the ability to
The deformation capacity of the elements. The oxide layer on the elements has a great influence on this property . It must be affirmed that : at the time of impact, the oxide layer is liquid, in this case it cannot maintain the deformation of the elements and vice versa, in elements with a hard oxide layer, its deformation capacity is mainly determined by this coating.
The ability of steel elements with oxide coating in liquid state to deform depends on the deformation of the previous elements , and it does not end immediately but further deformation occurs due to the action of the following elements, similar to the action of forging . The deformation of the elements occurs very quickly. Therefore, when the following elements collide with the previous elements that are still in liquid or viscous state, metallic bonding easily occurs between them.
1.3.2.4 Structure of metal coating
The metal coating has properties and composition that are completely different from the original material. The structure of the metal coating has the characteristics of structures that have been suddenly cooled. The cooling of the elements occurs very quickly and the cooling rate is very large, so when solidifying, dislocation centers will appear in the network, these centers affect the adhesion of the coating.
Due to the rapid crystallization of the particles in the coating, a metastable structure can be obtained.
stable in the form of saturated solid solutions and amorphous states of the metal. The post-spray heating stage will separate the stable phases in the form of small-structured impurities, evenly distributed in the coating volume. Such a structure will create a durable coating with good heat resistance.
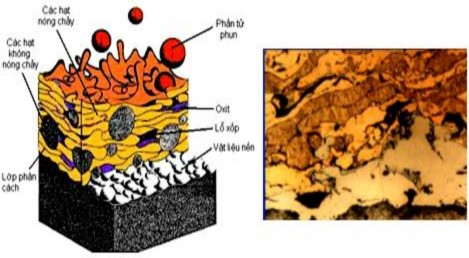
Figure 1.28. Structure of metal coating
In metal coatings there can be two types of oxides, one type of oxide is formed separately, the other type is wrapped around the deformed metal elements. The first type is often considered disadvantageous , worsening the mechanical properties of the coating. The second type has the task of linking the separate metal elements . Besides the above structures, in the composition of the coating structure, there must be a large number of pores. These pores are formed due to the loose connection of the metal elements when deformed. The pores in the coating structure will give the coating good properties when the coating works under lubricating conditions.
The coating forms in air, so the pores are filled with gas, thus deteriorating the contact properties, especially between the layers with the greatest possibility of gas saturation. Other defects can also form such as: no adhesion between the coating and the part, segregation, microcracking due to tensile stress on the deformed elements, cracks perpendicular to the surface due to the coating shrinking when cooling and being restrained by adhesion, non-melting particles. The chemical composition of the metallic coating is also different from that of the material.
initial coating. In general, the chemical composition (metallic elements) of the coating is usually
decrease , especially alloys.
The coating has a layered structure, consisting of highly deformed particles, connected to each other along the contact surface. It is characterized by metal sheets with layering due to the sheets formed at different times sliding over each other. These sheets have different deformations and are separated from each other by a thin oxide layer with a thickness of 1 m. The boundary between the coating and the metal substrate determines the adhesion between them. The properties of the coating itself are expressed by the adhesion between the particles. The boundary between the layers is formed due to different time intervals between sprayings. After each spraying, the surface is very quickly contaminated,
oxidation
This makes the contact processes between particles more difficult and thus the boundaries between the sprayed layers appear . The layer thickness varies greatly and depends on the technology used.
The structure and properties of the coating depend on the interaction processes between metal particles and gas flow and the coating formation process on the base metal surface, which is determined by the technology and spraying technology mode.
1.4 Adhesion of metal coatings
1.4.1. Adhesion force of metal particles
1.4.1.1 Adhesion force of liquid metal particles on solid surfaces
Assuming that the sprayed particles impact the surface of the part in a liquid state, there will be an adhesion phenomenon of a liquid droplet to the surface of solids based on the surface tension of the liquid metal droplet and the surrounding contact environment.
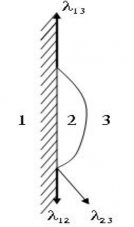
Figure 1.29. Adhesion of liquid drops on solid objects
1- solid; 2- liquid; 3- air Figure 1.29 describes the adhesion of a liquid drop onto a solid. If we call:
α 12 Is the tension between the liquid droplet and the solid α 23 The tension between the liquid droplet and the air α 13 The tension between the solid and the air The angle θ between the liquid droplet and the solid is called the tension angle
To keep the liquid drop in equilibrium, we have:
13 12 23 cos
cos 13 12
(1.4)
Hence:
23
(1.5)
The angle θ depends on the nature of the three phases, it varies with the state and purity of the
b surface. If the tension angle of the solid and gas is larger than that of the liquid, then the angle 0<θ<90 0 .
When cos θ>0 the liquid has a folded shape (Figure 1.30a ) and then the liquid drop wets the surface.
solid surface
When θ >90 0 , that is , α 13 >α 12 (that is, angle θ is obtuse), the liquid drop has a curved shape (figure 30b).
In this case, the liquid drop does not wet the solid surface. Since cosθ is not large, we have:
13 12
23
(1.6)
This is the wetting condition for a smooth surface .
For roughened surfaces, equation (1.4) (must be of the form (1.8) taking into account the coefficient K.
This coefficient is the ratio between the rough surface volume and the surface area.
K S n
S p
Sn - rough surface area
Sp - smooth surface area.
From (1.4) we have
K 13 K 12 23 cos
Then θ has the value θ 1
K 13 K 12 23 cos 1
K ( 13 12 ) 23 cos 1
From (1.5) we deduce:
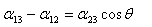
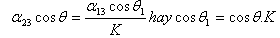 1 then θ1 wet glass is better (1.7) (1.8) (1.9) (1.10) 4 4" class="lazyload">When K θ sticks to 4" class="lazyload">1 then θ1 wets better (1.7) (1.8) (1.9) (1.10) 4" class="lazyload">
1 then θ1 wet glass is better (1.7) (1.8) (1.9) (1.10) 4 4" class="lazyload">When K θ sticks to 4" class="lazyload">1 then θ1 wets better (1.7) (1.8) (1.9) (1.10) 4" class="lazyload">
When K<1 then θ1>θ wets less well
When K>1 then θ1<θ d wetting is better
(1.7)
(1.8)
(1.9)
(1.10)
(1.11)
(1.12)
Due to roughening, the wetted area increases K times, so K is always greater than 1.
and the smaller θ angle should facilitate better adhesion.
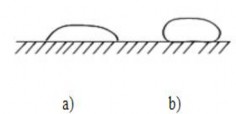
Figure 1.30. Shape of liquid drop
a-inward form; b - outward form
1.4.1.2 Van der Waals force
When a particle hits a metal surface , there is a Van der Waals binding force between them. This force acts on the dipole moments of two atoms or molecules. When the center of gravity of the electron due to fluctuations does not coincide with the center of charge of the atomic nucleus, then the atom becomes a dipole moment. The value and direction of the moment change over time and the average value over time is zero. If a molecule or atom with a dipole moment falls into the field of another atom that also has a dipole moment, there is a force acting between them. According to Debai, the energy exchanged between them is:
E 2 / 3. 12 22 . 1
w r 6 KT
(1.13)
In which μ1, μ2 – dipole moment
r6- distance between two atoms T- temperature
But when atoms or molecules react with the metal surface, the energy is transferred.
The change is difficult to determine because the atoms are in a network bonded to other atoms.
1.4.1.3 Binding force due to the influence of the electric double layer
During thermal motion, conductive electrons can move beyond the boundaries of the surface and form an electron cloud on the metal surface. Between the electron cloud, the atoms form a double layer of area, one pole is the electron cloud, the other pole is the atoms on the surface. This layer is called the double electron layer. When two metals are in contact with each other, due to the difference in electric potential at the two ends of the surface, there is an electric force acting between them. That force is calculated according to the formula:
F 2 2 S
(1.14)
- specific area per unit area of the electron cloud layer
S- contact area. This area depends on the force that flattens the metal particles at
contact area
This pressure depends largely on the nature of the metal and the surface factors of the metal.
contact type
1.4.1.4 Metallic bonds
When two metals come into contact with each other, the atoms of one metal, under the influence of thermodynamic factors , diffuse into the lattice of the other metal, forming transition regions. That is the solid solution between the two metals. Then we say that there is a metallic bond between them . This bond depends a lot on the contact temperature and the surface state of the metal .
Through experimental work , researchers have supplemented these perceptions.
on the adhesion of the coating to the base metal by the following methods:
By diffusion of two metals together (including micro diffusion).
By welding or micro-welding together .
By the bonding of chemical reactions. By the force of metal contraction during crystallization. By mechanical adhesion .
The theory of diffusion of metals as well as the welding process of metals together and the connection of chemical reactions has so far achieved quite deep and complete understanding. Here we consider some other perceptions related to and affecting the adhesion of coatings.
Mechanical adhesion is expressed as the holding of the metal particles against the irregularities of the base metal surface . This adhesion is an important factor for the overall adhesion of the coating.
In the case of considering the tendency of the surface layer to be completely clean before coating, we see that: the surface is not clean of metal dust, oxides, other dirt or different roughness etc., which can lead to different bonding by chemical or mechanical bonding force. Under suitable spraying conditions and suitable surface morphology, in some metals it is possible to achieve point-to-point welding between the base metal and the auxiliary elements.
When studying the adhesion of metal coatings and objects by spraying, author NN Rykalin proved that each surface area can be divided into the following 3 stages:
- Create a contact surface, meaning create a close displacement of the metal atoms
type to a distance sufficient to have a chemical reaction.
- The activity and chemical reaction of metal atoms close to each other will lead to
form strong chemical bonds.
- The recovery processes (recrystallization, diffusion to create new phases , ...) that occur next have
can enhance or decrease the strength of the bond.
In which the first two stages represent surface transformation, the third stage represents internal property change. To create a strong bond between the coating and the coated material, it is necessary to perform the first two stages well. Diffusion does not necessarily have to be performed in the third stage. This stage is often the result of high temperature heating (or annealing after coating).
1.4.2 Factors affecting adhesion
1.4.2.1 Effect of metal contraction force.
The force from the metal shrinkage has a significant influence on the coating adhesion. The metal particles after impacting the base surface will gradually cool down after a period of time and the coating will have stress due to the shrinkage of the metal coating. The stress caused by the shrinkage can also increase the adhesion of the coating to the base metal, and conversely, it can also reduce the adhesion of the coating to the base.
When spraying cylindrical outer surfaces , such as shafts, the forces generated by the shrinkage of the coating are internal forces and are therefore very weak, so their influence is negligible (see Figure 1.31a).
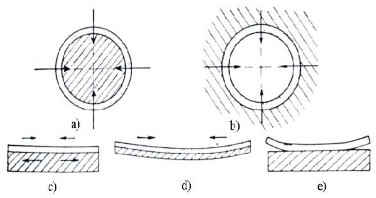
Figure 1.31. Stresses in the coating
In cases of spraying with a full layer and the metal to be sprayed has a large shrinkage (e.g. steel).
0.1% carbon) the influence of internal forces can lead to longitudinal cracking in the coating.
Internal forces generated by shrinkage on the surfaces in Figure 31b have a negative impact on the adhesion of the coating. In this case, the shrinkage acts in a radial direction, causing the coating to separate from the base surface. There are many ways to overcome these problems, such as preheating the part , cooling the coating slowly, or spraying with thin layers of 0.05 to 0.1 mm and cooling slowly for each layer.
For coatings on flat surfaces, the stress generated by the shrinkage of the coating metal can lead to deformation of the coating, the base metal or coating peeling (Figure 31 c, d, e). Consider a small coating element AB with length l, height H and cross-sectional area
∆S, we can consider as a bar attached to line AB (figure 1.32)