(1→4)-β- D- thevetopyranosyl-(14)-β- D- oleandropyranosyl-(14)-β- D- cymaropyranoside and attached to the C-3 position of the aglycone.
Table 3.5. NMR spectral data of compound GS5 and reference substance
C δ C # δ C a,b δ H a,c (multiplicity, J=Hz)
C | δ C # δ C a,b δ H a,c (multiplicity, J=Hz) | ||||||
1 | 39.8 | 39.8 | 1.08, (m) /1.80 (m) | 2 | 36.6 | 36.7 | 1.53 (m)/2.05 (m) |
2 | 30.2 | 30.2 | 1.58 (m)/1.84 (m) | 3 | 78.5 | 78.5 | 3.81 (m) |
3 | 79.2 | 79.3 | 3.50 (m) | 4 | 83.8 | 83.8 | 3.24 (m) |
4 | 39.8 | 39.8 | 2.20 (m)/2.33 (m) | 5 | 69.9 | 69.9 | 3.80 (m) |
5 | 140.0 | 140.0 | - | 6 | 18.5 | 18.4 | 1.19 (d, 6.4) |
6 | 120.0 | 120.0 | 5.32 (br s) | 3-OMe | 58.5 | 58.5 | 3.42 (s) |
7 | 35.2 | 35.2 | 2.11 (m) | Ole | |||
8 | 74.9 | 74.9 | - | 1 | 102.6 | 102.6 | 4.57 (br d, 9.2) |
9 | 44.7 | 44.7 | 1.49 (m) | 2 | 37.6 | 37.6 | 1.40 (m)/2.30 (m) |
10 | 38.0 | 38.0 | - | 3 | 80.2 | 80.3 | 3.35 (m) |
11 | 26.0 | 25.9 | 1.63 (m)/2.00 (m) | 4 | 84.1 | 84.1 | 3.18 (m) |
12 | 75.1 | 75.1 | 4.68 (m) | 5 | 72.5 | 72.6 | 3.38 (m) |
13 | 57.6 | 57.6 | - | 6 | 18.9 | 18.8 | 1.36 (d, 6.0) |
14 | 89.3 | 89.3 | - | 3-OMe | 57.6 | 57.6 | 3.39 (s) |
15 | 34.3 | 34.3 | 1.83 (m)/1.90 (m) | Thv | |||
16 | 33.5 | 33.5 | 1.75 (m) | 1 | 104.3 | 104.2 | 4.43 (d, 7.6) |
17 | 89.1 | 89.1 | - | 2 | 75.6 | 75.2 | 3.23 (m) |
18 | 11.2 | 11.2 | 1.53 (s) | 3 | 87.7 | 86.3 | 3.17 (m) |
19 | 18.5 | 18.6 | 1.12 (s) | 4 | 76.6 | 82.9 | 3.30 (m) |
20 | 71.5 | 71.5 | 3.45 (m) | 5 | 73.2 | 72.5 | 3.38 (m) |
21 | 18.9 | 18.9 | 1.05 (d, 6.4) | 6 | 18.1 | 18.5 | 1.36 (d, 7.2) |
Tig | 3-OMe | 61.0 | 61.2 | 3.60 (s) | |||
1 | 169.1 | 169.1 | - | Glc | |||
2 | 130.0 | 130.1 | - | 1 | 104.3 | 4.41 (d, 7.6) | |
3 | 139.5 | 139.5 | 6.98 (q, 7.2) | 2 | 75.6 | 3.15 (m) | |
4 | 14.5 | 14.5 | 1.80 (d, 7.2) | 3 | 78.3 | 3.23 (m) | |
5 | 12.2 | 12.2 | 1.85 (s) | 4 | 71.8 | 3.20 (m) | |
Cym | 5 | 77.9 | 3.31 (m) | ||||
1 | 97.2 | 97.2 | 4.84 (br d, 9.6) | 6 | 63.2 | 3.62 (m)/3.84 (m) | |
Maybe you are interested!
-
 Study on botanical characteristics, chemical composition and acetylcholinesterase enzyme inhibitory effects of two species Piper thomsonii (C. DC.) Hook. f. var. thomsonii and Piper hymenophyllum Miq., Piperaceae family - 24
Study on botanical characteristics, chemical composition and acetylcholinesterase enzyme inhibitory effects of two species Piper thomsonii (C. DC.) Hook. f. var. thomsonii and Piper hymenophyllum Miq., Piperaceae family - 24 -
 Study on chemical composition, toxicity and some biological effects supporting the treatment of gastric and duodenal ulcers of Sanchezia nobilis Hook.F. leaves - 2
Study on chemical composition, toxicity and some biological effects supporting the treatment of gastric and duodenal ulcers of Sanchezia nobilis Hook.F. leaves - 2 -
 Study on botanical characteristics, chemical composition and acetylcholinesterase enzyme inhibitory effects of two species Piper thomsonii (C. DC.) Hook. f. var. thomsonii and Piper hymenophyllum Miq., Piperaceae family - 23
Study on botanical characteristics, chemical composition and acetylcholinesterase enzyme inhibitory effects of two species Piper thomsonii (C. DC.) Hook. f. var. thomsonii and Piper hymenophyllum Miq., Piperaceae family - 23 -
 Study on botanical characteristics, chemical composition and acetylcholinesterase enzyme inhibitory effects of two species Piper thomsonii (C. DC.) Hook. f. var. thomsonii and Piper hymenophyllum Miq., Piperaceae family - 21
Study on botanical characteristics, chemical composition and acetylcholinesterase enzyme inhibitory effects of two species Piper thomsonii (C. DC.) Hook. f. var. thomsonii and Piper hymenophyllum Miq., Piperaceae family - 21 -
 Study on chemical composition and evaluation of anti-cancer effect of stem and leaves of Stephania dielsiana YC Wu - 1
Study on chemical composition and evaluation of anti-cancer effect of stem and leaves of Stephania dielsiana YC Wu - 1

# abc
δ C of gymsyloside A (GS1), measured in methanol-d 4 , 100 MHz, 400 MHz
Therefore, the structure of compound GS5 was concluded to be (20S)-12β-tigloyloxy- 3β,8β,14β,17β,20-pentahydroxypregn-5-ene 3-O-β- D- glucopyranosyl-(1→4)-β- D- thevetopyranosyl-(14)-β- D- oleandropyranosyl-(14)-β- D- cymaropyranoside, which is a new compound named gymsyloside E.
Figure 3.21. Main HMBC and COSY interactions of compound GS5
3.1.2.6. Compound GS6: gymnepregoside R
Figure 3.22. Chemical structure of compound GS6
Compound GS6 was isolated as a white amorphous powder. Comparison of the NMR spectral data of compound GS6 and the corresponding data of compound GS4 revealed similarities. The signals of the pregn-5-ene framework were easily recognized by the presence of three singlet methyl groups at δ H 1.05, 1.13, 1.52, an olefin proton at δ H 5.32 and two olefin carbons at δ C 119.9 and 140.0. A tigloyl group was also recognized via the olefin proton of the double bond at δ H 6.98 along with two olefin carbons at δ C 130.0 and 139.5 and a carbony carbon at δ C 169.0. The aglycon framework of GS6 was determined to be similar to GS4 by the close match of the NMR data sets of the two compounds. In the sugar chain, there are 5 sugar units identified through the data set of 5 pairs of protons and anomer carbons: Cym I (δ H (4.87)/δ C (97.2)), Cym II (δ H (4.77)/δ C (101.2)), Ole (δ H (4.56)/δ C (102.6)) and All (δ H (4.69)/δ C (102.2)), Glc (δ H (4.33)/δ C 106.2).
Table 3.6. NMR spectral data of compound GS6 and reference substance
# a, three, c # a, three, c
C δ C δ C δ H (multiplicity, J=Hz) C δ C δ C δ H (multiplicity, J=Hz)
1
39.8 | 39.8 | 1.09 (m)/1.78 (m) | 6 | 18.3 | 18.5 | 1.17 (d, 6.4) | |
2 | 30.2 | 30.2 | 1.57 (m)/1.83 (m) | 3-OMe | 58.4 | 58.4 | 3.40 (s) |
3 | 79.3 | 79.3 | 3.50 (m) | Cym II | |||
4 | 39.8 | 39.8 | 2.20 (m)/2.33 (m) | 1 | 101.2 | 101.2 | 4.77 (br d, 9.6) |
5 | 140.0 | 140.0 | - | 2 | 36.4 | 36.4 | 1.54 (m)/2.05 (m) |
6 | 120.0 | 119.9 | 5.32 (br s) | 3 | 78.4 | 78.4 | 3.82 (m) |
7 | 35.2 | 35.2 | 2.10 (m) | 4 | 83.8 | 83.8 | 3.24 (m) |
8 | 74.9 | 74.9 | - | 5 | 69.9 | 69.9 | 3.79 (m) |
9 | 44.7 | 44.7 | 1.49 (m) | 6 | 18.6 | 18.5 | 1.20 (d, 6.0) |
10 | 38.0 | 38.0 | - | 3-OMe | 58.5 | 58.5 | 3.40 (s) |
11 | 26.0 | 26.0 | 1.62 (m)/1.98 (m) | Ole | |||
12 | 75.1 | 75.1 | 4.68 (m) | 1 | 102.6 | 102.6 | 4.56 (br d, 10.8) |
13 | 57.6 | 57.6 | - | 2 | 37.5 | 37.6 | 1.40 (m)/2.30 (m) |
14 | 89.3 | 89.3 | - | 3 | 80.3 | 80.4 | 3.35 (m) |
15 | 34.3 | 34.3 | 1.83 (m)/1.90 (m) | 4 | 83.7 | 83.9 | 3.17 (m) |
16 | 33.5 | 33.5 | 1.76 (m) | 5 | 72.5 | 72.5 | 3.34 (m) |
17 | 89.1 | 89.1 | - | 6 | 18.9 | 18.9 | 1.34 (d, 6.0) |
18 | 11.2 | 11.2 | 1.52 (s) | 3-OMe | 57.4 | 57.5 | 3.39 (s) |
19 | 18.6 | 18.6 | 1.13 (s) | All | |||
20 | 71.5 | 71.5 | 3.45 (m) | 1 | 102.2 | 102.1 | 4.69 (d, 8.0) |
21 | 19.0 | 19.0 | 1.05 (d, 5.6) | 2 | 73.5 | 72.9 | 3.30 (m) |
Tig | 3 | 83.9 | 83.2 | 3.93 (m) | |||
1 | 169.1 | 169.0 | - | 4 | 75.0 | 83.9 | 3.30 (m) |
2 | 130.1 | 130.0 | - | 5 | 71.2 | 70.2 | 3.79 (m) |
3 | 139.5 | 139.5 | 6.98 (q, 7.2) | 6 | 18.6 | 18.1 | 1.28 (d, 6.0) |
4 | 14.6 | 14.5 | 1.80 (d, 7.2) | 3-OMe | 62.5 | 62.0 | 3.58 (s) |
5 | 12.2 | 12.1 | 1.85 (s) | Glc | |||
Cym I | 1 | 106.2 | 4.33 (d, 7.2) | ||||
1 | 97.2 | 97.2 | 4.84 (br d, 9.6) | 2 | 75.4 | 3.16 (m) | |
2 | 36.6 | 36.6 | 1.54 (m)/2.05 (m) | 3 | 78.0 | 3.26 (m) | |
3 | 78.5 | 78.5 | 3.82 (m) | 4 | 71.8 | 3.22 (m) | |
4 | 83.8 | 83.8 | 3.24 (m) | 5 | 77.9 | 3.32 (m) | |
5 | 69.8 | 69.8 | 3.79 (m) 6 63.1 3.63 (dd, 6.0 11.8) | ||||
3.89 (d, 11.8)
δ C # of compound GS4 , a measured in methanol-d4 , b 100 MHz, c 400 MHz
Compared with compound GS4, it was found that compound GS6 had an additional glucose unit (δ C 106.2, 75.4, 78.0, 71.8, 77.9 (5 x CH) and 63.1 (CH 2 ). This was further confirmed by high-resolution mass spectrometry results. On the HR-ESI-MS spectrum, a pseudomolecular ion peak appeared at m/z 1241.6309 [M + Na] + (theoretical calculation for the formula
The molecular formula [C 60 H 98 O 25 Na] + is 1241.6289), corresponding to the molecular formula of GS6 is C 60 H 98 O 25 . This molecular mass is 162 mcg larger than that of compound GS4 (C 6 H 10 O 5 ), completely consistent with the assumption of a glucose unit. In addition, hydrolysis of compound GS6 in acidic medium yields the monosaccharides D -cymarose, D - oleandrose and 6-deoxy- 3-O- methyl - D - allose and D - glucose.
The sugar orders were determined by HSQC, HH COSY and HMBC spectra similar to those of compounds GS1-GS5. The HMBC interactions between Glc H-1 (δ H 4.33) and All C-4 (δ C 83.9); All H-1 (δ H 4.69) and Ole C-4 (δ C 83.9); Ole H-1 (δ H 4.56) and Cym II C-4
(δ C 83.8); Cym II H-1 (δ H 4.77) and Cym I C-4 (δ C 83.8) confirmed that the sugar chain is 3-O-β- D- glucopyranosyl-(1→4)-6-deoxy- 3-O-methyl-β- D- allopyranosyl-(14)-β- D- oleandropyranosyl-(14)-β- D- cymaropyranosyl-(14)-β- D- cymaropyranoside.
Therefore, the structure of the leaked compound GS6 was 12β-tigloyloxy-3β,8β,14β,17β,20S- pentahydroxypregn-5-ene 3-O-β- D -glucopyranosyl-(1→4)-6-deoxy-3-O-methyl-β- D -allopyranosyl-(1→4)-β- D -oleandropyranosyl-(1→4)-β- D -cymaropyranosyl- (1→4)-β- D -cymaropyranoside - a new compound named gymnepregoside R.

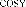
Figure 3.23. Main HMBC and COSY interactions of compound GS6
3.1.2.7. Compound GS7: gymnepregoside T
Figure 3.24. Chemical structure of compound GS7
Compound GS7 was obtained as a white amorphous powder. The molecular formula of compound GS7 was determined as C 51 H 82 O 20 based on the pseudomolecular ion peak at m/z 1037.5299 [M + Na] + (theoretical calculation for the formula [C 51 H 82 O 20 Na] + is 1037.5292). The 1 H NMR spectrum of compound GS7 showed the presence of 3 singlet methyl groups at 1.12, 1.47, and 2.20 Å, 1 olefin proton at δ H 5.32 (1H, br s), and 5 anomer protons at [δ H 4.83 (br d, J = 9.6 Hz), 4.77 (br d, J = 9.6 Hz), 4.57 (br d, J = 8.4 Hz), 4.69 (br
d, J = 8.0 Hz) suggests that the structure of GS7 is a pentasaccharide pregnane. However, unlike the previous compounds, GS7 no longer has signals of tigloyl and benzoyl groups, but has 2 additional methyl singlet groups at δ H 1.90 and 2.20 along with 2 carbonyl carbons at δ C 171.8 and 212.2, suggesting the presence of two acetyl radicals. In addition, the loss of the methyl doublet signal like the previous pregnanes at δ H 1.02-1.05 allows predicting that the hydroxy group at C-21 has been oxidized to a ketone, generating an acetyl radical here. , 4 Hz) confirm the existence of 4 deoxy sugar units . The 13 C NMR and DEPT spectral data of GS7 showed conformation with a pregnane skeleton structure (δ C 39.8, 30.1, 79.2, 39.8, 140.2, 119.7, 35.1, 74.9, 45.1, 38.1, 25.2,
74.9, 57.4, 89.9, 34.2, 33.1, 93.1, 10.2, 18.6, 212.2 and 27.7 ), 1 acetyl group (2 carbons)
and 4 deoxy sugar units (28 carbons) [77]. The positions of the substituents as well as the order of the sugars were also determined through the HMBC spectrum. The position of the acetyl group
The C-12 position was determined by HMBC interactions from H-12 (δ H 4.46) to Ac C- 1 (δ C 171.8). The acetyl group position at C-17 was determined by HMBC interactions from H-20 (δ H 2.20) to C-17 (δ C 93.1)/C-20 (δ C 212.2). The HMBC interactions between All H-1 (δ H 4.70) and Ole C-4 (δ C 84.0); Ole H-1 (δ H 4.56) and Cym II C-4 (δ C 83.8); Cym II H-1 (δ H 4.77) and Cym I C-4 (δ C 83.8) and Cym H-1 (δ H 4.83) with C-3 (δ C 79.3) were identified.
The sugar chain 3-O-6-deoxy-3-O-methyl-β-D-allopyranosyl-(14)-β-D-oleandropyranosyl-(14)-β-D-cymaropyranosyl-(14)-β-D-cymaropyranoside is attached to the C-3 position of the aglycone. Combining the above arguments, the structure of the leaked compound GS7 was 12β-acetyloxy-3β,8β,14β,17β-tetrahydroxypregn-5-en-20-one 3- O-6-deoxy-3-O-methyl-β- D -allopyranosyl- ( 14)-β- D -oleandropyranosyl-(14)-β- D -cymaropyranosyl-(14)-β- D -cymaropyranoside, a new compound was named gymnepregoside T.
Table 3.7. NMR spectral data of compound GS7
C δ C a,b δ H a,c (multiplicity, J=Hz) C δ C a,b δ H a,c (multiplicity, J=Hz)
1
39.8 | 1.08 (m)/1.78 (m) | 4 | 83.8 | 3.22 (m) | |
2 | 30.1 | 1.56 (m)/1.83 (m) | 5 | 69.8 | 3.78 (m) |
3 | 79.2 | 3.50 (m) | 6 | 18.3 | 1.16 (d, 6.0) |
4 | 39.8 | 2.20 (m)/2.33 (m) | 3-OMe | 58.5 | 3.41 (s) |
5 | 140.2 | - | Cym II | ||
6 | 119.7 | 5.32 (br s) | 1 | 101.2 | 4.77 (br d, 9.6) |
7 | 35.1 | 2.10 (m) | 2 | 36.4 | 1.54 (m)/2.10 (m) |
8 | 74.9 | - | 3 | 78.5 | 3.82 (m) |
9 | 45.1 | 1.47 (m) | 4 | 83.8 | 3.22 (m) |
10 | 38.1 | - | 5 | 70.0 | 3.78 (m) |
11 | 25.2 | 1.68 (m)/1.84 (m) | 6 | 18.5 | 1.19 (d, 6.0) |
12 | 74.4 | 4.46 (dd, 3.2, 10.8) | 3-OMe | 58.5 | 3.41 (s) |
13 | 57.4 | - | Ole | ||
14 | 89.8 | - | 1 | 102.6 | 4.56 (br d, 8.4) |
15 | 34.2 | 1.85 (m)/1.95 (m) | 2 | 37.5 | 1.39 (m)/2.29 (m) |
16 | 33.1 | 1.69 (m)/2.81 (m) | 3 | 80.4 | 3.36 (m) |
17 | 93.1 | - | 4 | 84.0 | 3.18 (m) |
18 | 10.2 | 1.47 (s) | 5 | 72.6 | 3.35 (m) |
19 | 18.6 | 1.12 (s) | 6 | 19.0 | 1.35 (d, 6.4) |
20 | 212.2 | - | 3-OMe | 58.4 | 3.39 (s) |
21 | 27.7 | 2.20 (s) | All | ||
12-OAc | 1 | 102.2 | 4.70 (d, 8.0) | ||
1 | 171.8 | - | 2 | 73.6 | 3.29 (m) |
2 | 20.8 | 1.90 (s) | 3 | 83.8 | 3.60 (m) |
Cym I | 4 | 75.0 | 3.16 (m) | ||
1 | 97.2 | 4.83 (br d, 9.6) | 5 | 71.2 | 3.63 (m) |
2 | 36.6 | 1.54 (m)/2.10 (m) | 6 | 18.5 | 1.21 (d, 6.0) |
3 | 78.5 | 3.82 (m) | 3-OMe | 62.5 | 3.58 (s) |
a measured in methanol-d 4 , b 100 MHz, c 400 MHz
Figure 3.25. Main HMBC and COSY interactions of compound GS7

3.1.2.8. Compound GS8: vetircilloside M
Figure 3.26. Chemical structure of compound GS8
Compound GS8 was obtained as a white amorphous powder. The molecular formula of compound GS8 was determined as C 62 H 96 O 25 based on the ion peak at m/z 1263.6148 [M
+ Na] + (theoretical calculation for the formula [C 62 H 96 O 25 Na] + is 1263.6133). Similar to compound GS6, the 1 H and 13 C NMR spectra of compound GS8 revealed the presence of a pregnane skeleton and five sugar units Cym I (δ H (4.83)/δ C (97.2)), Cym II (δ H (4.77)/δ C (101.2)), Ole (δ H (4.56)/δ C (102.6)) All (δ H (4.69)/δ C (102.1)) and Glc ((δ H (4.33)/δ C
(106.2)). However, the tigloyl group signal of GS6 was absent in the NMR spectrum of GS8, and instead, a very characteristic aromatic ring appeared at δ C 131.8 (C), 130.9 x 2CH , 129.5 x 2CH and 134.2 (C) and δ H 7.46-8.12 (5H). The above signals together with the presence of a carbonyl carbon (δ C 167.8) suggested the existence of a radical
benzoyl. This prediction was further confirmed by the interactions in the HMBC spectrum.
Table 3.8. NMR spectral data of compound GS8
a, three, ca, three, c
C δ C δ H (multiplicity, J=Hz) C δ C δ H (multiplicity, J=Hz)
1
39.8 | 1.11 (m)/1.82 (m) | 6 | 18.5 | 1.17 (d, 6.4) | |
2 | 30.2 | 1.58 (m)/1.84 (m) | 3-OMe | 58.4 | 3.40 (s) |
3 | 79.3 | 3.51 (m) | Cym II | ||
4 | 39.8 | 2.20 (m)/2.33 (m) | 1 | 101.2 | 4.77 (br d, 9.6) |
5 | 140.0 | - | 2 | 36.4 | 1.54 (m)/2.07 (m) |
6 | 120.0 | 5.33 (br s) | 3 | 78.4 | 3.82 (m) |
7 | 35.2 | 2.14 (m) | 4 | 83.8 | 3.22 (m) |
8 | 74.9 | - | 5 | 69.9 | 3.80 (m) |
9 | 44.8 | 1.56 (m) | 6 | 18.6 | 1.20 (d, 6.0) |
10 | 38.0 | - | 3-OMe | 58.4 | 3.41 (s) |
11 | 26.0 | 1.77 (m)/2.10 (m) | Ole | ||
12 | 75.9 | 4.68 (m) | 1 | 102.6 | 4.56 (br d, 7.2) |
13 | 57.7 | - | 2 | 37.6 | 1.40 (m)/2.30 (m) |
14 | 89.3 | - | 3 | 80.5 | 3.34 (m) |
15 | 34.4 | 1.84 (m)/1.96 (m) | 4 | 83.9 | 3.17 (m) |
16 | 33.5 | 1.78 (m) | 5 | 72.5 | 3.34 (m) |
17 | 89.1 | - | 6 | 18.9 | 1.35 (d, 6.0) |
18 | 11.3 | 1.52 (s) | 3-OMe | 57.7 | 3.40 (s) |
19 | 18.5 | 1.13 (s) | All | ||
20 | 71.6 | 3.54 (m) | 1 | 102.1 | 4.69 (d, 8.0) |
21 | 19.0 | 1.02 (d, 6.0) | 2 | 72.9 | 3.30 (m) |
Bz | 3 | 83.2 | 3.93 (m) | ||
1 | 167.8 | - | 4 | 83.9 | 3.32 (m) |
2 | 131.8 | - | 5 | 70.1 | 3.80 (m) |
3.5 | 130.9 | 8.12 (d, 7.6) | 6 | 18.2 | 1.28 (d, 6.4) |
4.6 | 129.5 | 7.46 (t, 7.6) | 3-OMe | 61.9 | 3.57 (s) |
5 | 134.2 | 7.58 (t, 7.6) | Glc | ||
Cym I | 1 | 106.2 | 4.33 (d, 7.2) | ||
1 | 97.2 | 4.84 (br d, 9.6) | 2 | 75.5 | 3.16 (m) |
2 | 36.6 | 1.54 (m)/2.05 (m) | 3 | 78.0 | 3.20 (m) |
3 | 78.5 | 3.82 (m) | 4 | 71.8 | 3.22 (m) |
4 | 83.8 | 3.24 (m) | 5 | 77.8 | 3.31 (m) |
5 | 69.8 | 3.79 (m) | 6 | 63.0 | 3.63 (dd, 6.0, 11.8) |
3.89 (d, 11.8)
a measured in methanol-d 4 , b 100 MHz, c 400 MHz
The HMBC interactions between benzoyl H-2/H-6 (δ H 8.12) and C-1 (δ C 131.8)/C-4 (δ C 134.2)/C-7 (δ C 167.8); between H-12 (δ H 4.68) and benzoyl C-7 (δ C 167.8) suggested the benzoyl group attached to the C-12 position of the pregnane backbone. The sugar chain was confirmed to be similar to gymnepregoside R (GS6) by comparison of 1 H, 13 C NMR spectral data as well as HMBC and COSY interactions . Therefore, the structure of





