quite high. To see the ratio of ingredients in crocodile meat can be compared with the composition of pork, in pork the average composition of substances is as follows: protein 13%, fat 20%, water 57% (according to Hoffman et al., 2000).
1.2.3 Other crocodile by-products
The two musk glands under the crocodile's jaw are used in the perfume industry because they give the perfume a distinctive and long-lasting scent. Some dried crocodile parts are used as traditional medicine in the East, but the market is still very limited. Crocodile bile is used to treat respiratory and digestive diseases. Some places sell crocodile oil made from its fat. Some people use the skull and entire bones of crocodiles to cook glue, make handicrafts, grind them into fertilizers, and feed animals.
1.3 Main nutritional components in crocodile meat
To date, although the consumption of crocodile meat has been growing, there is still not much information about the nutritional value of this meat. One of the few studies on the nutritional value of crocodile is the study of Hoffman et al. (2000). The research results from the tail, neck, body and leg meat of crocodile (Crocodylus niloticus) from 33
- 36 months of age are presented in Table 1-1 . showing no differences in protein and fat content among the body parts studied.
Table 1-1. Chemical composition of meat parts of Nile crocodile (g.kg -1 )
Tail | Foot | Close | Neck | Medium | |
Humidity | 701.7±18.96 | 733.9 ± 7.57 | 670.7 ± 22.38 | 759.5 ± 13.21 | 716.4 ± 36.77 |
Protein | 210.9 ± 8.09 | 224.0 ± 9.59 | 218.8 ± 7.02 | 229.4 ± 7.26 | 220.8 ± 10.09 |
Fat | 88.5±27.15 | 40.4 ± 11.63 | 91.1± 16.96 | 29.4±8.41 | 62.3 ± 32.17 |
Ash | 5.9 ± 1.55 | 3.6 ± 0.78 | 6.5 ± 2.00 | 4.5 ± 1.63 | 5.1 ± 1.85 |
Maybe you are interested!
-
 Product Orientation for Source Markets
Product Orientation for Source Markets -
 Goal of Improving Product Image and Quality
Goal of Improving Product Image and Quality -
 Accounting for production costs and product pricing of P&G Advertising and Trading Company Limited - 1
Accounting for production costs and product pricing of P&G Advertising and Trading Company Limited - 1 -
 Tourism Products and Product Systems of Travel Agencies
Tourism Products and Product Systems of Travel Agencies -
 Cat Ba National Park (Source: Internet, Accessed June 15, 2018)
Cat Ba National Park (Source: Internet, Accessed June 15, 2018)
(Hoffman et al., 2000)
In addition, crocodile meat also contains essential amino acids, the results are presented in Table 1-2.
Table 1-2 . Amino acid content of crocodile lean meat (g.kg -1 , on dry matter)
Amino acid content (g.kg -1 )
Threonine 3.291 ± 0.057
Serine 2.817 ± 0.066
Glycine 4.056 ± 0.239
Alanine 4.533 ± 0.086
Valine 3.471 ± 0.087
Methionine 2.060 ± 0.045
Isoleucine 3.557 ± 0.113
Leucine 6.430 ± 0.133
Tyrosine 2.597 ± 0.076
Phenylalanine 2.913 ± 0.081
Histidine 2.147 ± 0.073
Lysine 6.971 ± 0.152
Arginine 6.346 ± 1.469
(Hoffman et al., 2000)
1.4 Overview of bones
1.4.1 Bone structure
The main components of bone are mainly collagen fibers and inorganic bone minerals in the form of small crystals. In the bone structure (bone in living organisms), there is 10% to 20% water. Its dry weight, about 60 - 70% is bone mineral. Most of the remaining part is collagen, besides that, bone also contains a small amount of other substances such as non-protein and inorganic salts (according to Szpak, 2011). The composition of minerals is similar to hydroxyapatite (calcium phosphate hydrate). However, in hydroxyapatite, the Ca:P ratio is 5:3 (1.67), the bone mineral has a Ca:P ratio ranging from 1.37 - 1.87. This is because the mineral components in bone are much more complex and contain other ions such as silicon, carbonate and zinc. Compared with mammalian collagen, fish collagen in general, is characterized by a relatively high ratio
serine and glycine, and low proportions of hydroxyproline and proline (Omokanwaye et al., 2010; Szpak, 2011). Crocodile bone is very hard white Figure 1.1.

Figure 1.1. Crocodile bones with marrow, fat and meat removed (Source: Hoa Ca Crocodile Company Limited, 2014)
Bones play a role in supporting the body and performing many other functions in the animal body. Bones are characterized by high hardness, resilience, growth and self-repair. Considered a masterpiece of nature, bones are a type of tissue with a complex structure composed of inorganic substances and a network of organic substances that create mechanical strength and firm compression for bones.
1.4.2 Some applications of crocodile bones
Currently in Vietnam, Hoa Ca Crocodile Company Limited is a company that cooks crocodile bone extract as a dietary supplement and has sold it on the market. In the program 'Beautiful Diamond' of Hoa Ca Crocodile Company Limited established in 2010, crocodile bone extract is used to support the treatment of osteogenesis imperfecta (brittle bones) in children and has shown very potential results. Initial results from the analysis of bone turnover markers in blood serum show that taking crocodile bone extract for a period of time can increase the collagen biosynthesis process in the bones of patients with osteogenesis imperfecta. This helps to significantly improve bone health, reduce pain and reduce the frequency of bone fractures in these patients (Ton That Hung et al., 2013). In addition, according to the records of the Institute of Traditional Medicine, Ho Chi Minh City. HCM, this product is also effective for some patients with bone and joint problems and this information needs to be more widely communicated to users. According to the study of Gungormus and Kaya (2002),
Type I collagen when applied to bone during surgery can speed up bone healing.
The current method used by Ca Sau Hoa Ca Co., Ltd. to extract the extract is the traditional cooking-condensing method. Accordingly, the whole bone is boiled at 100 C. After each day of cooking, the collagen liquid is removed and new water is added to the bone for the next cooking. This process is repeated 6 - 7 times, meaning the cooking time is about 7 days. The cooking pot has two floors, the first floor is used to cook the bone. The steam in this floor heats the upper floor where the collagen liquid is contained and thus the liquid is condensed. With such a high cooking temperature and long cooking time, the resulting product is dark brown and there may be other uncontrolled changes such as changes in smell as well as other quality characteristics. In fact, other types of bone extract from animals are also cooked according to the same folk experience. There has not been any specific scientific research (domestic or international) conducted to improve this traditional process.
Currently, the method of extracting collagen from animal bones or other parts such as skin, tendons ... is soaked with acetic acid and pepsin at room temperature (Wood et al., 2008, Mokrejs et al., 2009, Zhang et al., 2009, Chen et al., 2011). So far, there has been no research to prove whether the health benefits found in crocodile bone extract are due only to collagen or other components but at low levels. Therefore, the experiments were conducted to improve the traditional thermal method, not to follow the approach used elsewhere. The experiments were conducted to improve in the direction of reducing energy use, reducing the impact on the quality of the obtained product while ensuring or even increasing high recovery efficiency from bones.
The applicability of collagen extract also needs attention. According to Omokanwaye et al. (2010), current collagen extract products are thick (moisture content ~ 35%), difficult to dissolve in water and when dispersed in water, they form a highly turbid suspension. Such characteristics will limit the application of bone extract in other food products. Enzymatic hydrolysis of collagen can increase the absorbability of the product and according to some pre-tests, hydrolysis also increases the clarity of the solution.
Type I collagen in bone has a triple helix structure. This structure makes it difficult for collagen to be digested efficiently in the intestine when consumed. Some studies have shown that pepsin (a proteolytic enzyme in the stomach) cannot completely break down the triple helix structure of collagen (Kittiphattanabawon et al., 2010, Liu et al., 2012). Trypsin (one of the two main enzymes responsible for peptide hydrolysis in the small intestine – trypsin and chymotrypsin) also cannot completely hydrolyze type I collagen (Buckley et al., 2008).
1.5 Overview of collagen
1.5.1 Introduction to collagen
Collagen is the main protein component of connective tissues, accounting for about 25% - 35% of total protein in the animal body and is the main component of the extracellular matrix. Collagen is a fibrous protein and is found in connective tissues of the heart, blood vessels, skin, cornea, cartilage, horns, ligaments, and bones. Collagen is also found in the internal tissues of most organs. Collagen provides strength to tissues and organs and maintains their structural form. Collagen is very diverse in composition and structure (Bolboacă et al., 2007; Pataridis et al., 2009; Singh et al., 2011).
1.5.2 Collagen classification
Collagen exists in many parts of the body. There are 28 types of collagen found and published in scientific literature. Over 90% of collagen in the body is type I, II, III and IV (Shoulders et al., 2009).
Collagen I: found in skin, tendons, blood vessels, organs, bones. Collagen II: found in cartilage (main component of cartilage). Collagen III: found in muscle (main component of muscle). Collagen IV: main component of cell membranes.
1.5.3 Collagen structure
Collagen is a structural protein found in the extracellular matrix and connective tissue of animals. It is a protein found in the animal kingdom and is not found in plants and unicellular organisms. In invertebrates, collagen is present in the body wall.
and epidermis. Collagen accounts for 25-30% of the total protein of the mammalian body (Bolboacă et al., 2007). Collagen is present in the cornea, bones, blood vessels, cartilage, dentin, etc.
Collagen has a complex structure. Collagen (or Tropocollagen) is a fiber about 300nm long with a diameter of 1.5nm, formed by three polypeptide chains, the chains twist together to form the helical structure of collagen, the polypeptide chains are linked together in the chain by hydrogen bonds. Polypeptide chains are synthesized similarly to other proteins (According to Pataridis et al., 2009).
The amino acid composition may vary depending on the origin of collagen, but there are some common and unique properties for all collagens. Collagen does not contain cysteine and tryptophan, but contains a large amount of glycine (Gly) accounting for about 30%, proline (Pro) accounting for 12% and hydroxyproline (Hyp) accounting for about 12.8 - 14.7%. Collagen is one of the few proteins that contain hydroxylysine (Hyl), in addition, collagen also contains minerals accounting for 1%. Hyp is a characteristic amino acid of collagen that other proteins do not have and depending on the origin of collagen, the amount of Hyp is different (Le Trung Thien, 2016).
Recently, 28 types of collagen have been identified, consisting of 46 distinct polypeptide chains, all of which have a characteristic triple helix, but the length, size, and properties of the remaining part vary depending on the type of collagen. According to published studies , in bone there are only two main types of collagen, type I: [α1(I)] 2 α2(I) and a few types V: α1(V) α2(V), α3(V) (According to Wood et al. (2008)
Collagen types I, II, III, IV account for more than 90% of total collagen in the body. Collagen type I is found in skin, tendons, blood vessels, organs, and bones. Collagen type II is the main component of cartilage. Collagen type III is found in muscles. Collagen type IV is the main component of cell membranes (Silvipriya et al., 2015) (Figure 1.2).
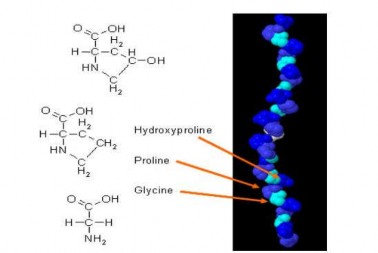
Figure 1.2 Structure of collagen
(Source: Silvipriya et al., 2015) Type I collagen is the most common in muscle tissue structure with molecular weight
150,000 units with special functional activities. There are 42 polypeptide chains found, which are encoded by 41 genes to form 28 different types of collagen. The structure of three polypeptide chains twisted in a spiral around a strand forms a tropocollagen triplet with an average length of 300 nm, diameter of 1.4 nm thanks to hydrogen bonds. Each polypeptide chain contains about 1,050 different amino acids, especially the sequential repetition of three amino acids Gly - X - Y (X is usually Pro, Y is Hyp). Because Gly occupies a large content and is distributed fully in the molecule, its repetition allows the polypeptide chains to be tightly linked together to form a spiral with small gaps in the concave part, each turn of the spiral contains 3.3 amino acid residues, height 2.9A°. About 4 - 8 tropocollagen molecules are linked together head-to-tail, forming collagen fibers (Gelse et al., 2003).
Collagen fibers are arranged in parallel longitudinally, linked together by cross-links to form fibers in a certain periodicity. They are arranged staggered about 67 nm apart and have a gap of 40 nm in adjacent molecules forming collagen fibers with diameters of 10 - 500 nm depending on the stage of development. Collagen fibers will establish collagen fiber bundles (Figure 1.2). In most body parts and especially bones. Collagen type I provides tensile strength and stiffness
in bone. It determines significant biomechanical properties related to load-bearing, tensile strength, torsional stiffness and especially after calcification (Gelse et al., 2003).
According to the study of Wood et al. (2008), collagen was extracted from the bones of the crocodile Alligator mississippiensis . The results showed that the collagen extracted from the bones of the above crocodile was type I collagen with molecular weights ([α1] 2 α2) of 359 kDa and 356 kDa, respectively, for the two extraction methods using acid and pepsin. The study also showed that if considering each α1 chain separately, the molecular weight was 124 kDa, the α2 chain was 111 kDa and the β chain was about 200 kDa. The amino acid composition of collagen is very rich with 18 types of amino acids (Table 1-3), especially glycine, proline, alanine and hydroxyproline.
Table 1-3. Free amino acid content in crocodile bone extract extracted by acetic acid and pepsin enzyme method
Amino acid
PP acetic acid | PP Enzyme pepsin | |
Hyp | 77.6 ± 1.2 | 86.6 ± 3.2 |
Asx | 40.1 ± 0.1 | 37.2 ± 1.8 |
Thr | 21.4 ± 0.5 | 22.0 ± 2.0 |
Ser | 45.2 ± 1.2 | 46.2 ± 1.0 |
Glx | 66.5 ± 1.0 | 65.8 ± 3.7 |
Pro | 116.5 ± 1.6 | 109.5 ± 1.5 |
Gly | 354.9 ± 10.5 | 367.2 ± 10.0 |
Ala | 134.4 ± 3.6 | 103.2 ± 4.2 |
Val | 15.2 ± 0.4 | 15.6 ± 1.2 |
Met | 8.5 ± 0.5 | 8.4 ± 0.4 |
Ile | 10.5 ± 0.3 | 10.8 ± 0.6 |
Leu | 20.4 ± 0.4 | 19.5 ± 0.3 |
Tyr | 1.4 ± 0.2 | 1.6 ± 0.1 |
Faction | 12.9 ± 0.3 | 12.2 ± 0.6 |
Hyl | 19.6 ± 0.3 | 19.1 ± 0.3 |
Lys | 33.3 ± 1.0 | 30.1 ± 1.1 |





