to adjust, supplement.
- The couple discusses, agrees and submits photos of their notebooks/products to the teacher via the assignment section on Teams, receives feedback from the teacher to continue editing.
- Draw the KWL table in the TH notebook, self-evaluate the goals achieved after the online TH and fill in the corresponding information in columns K, W.
Product:
- Student notebooks are presented according to the following structure:
LESSON NAME :.................................................
Day:..................
Questions/ corrections, additions add/note (2) | Content of answers to TH orientation questions (1) |
Mind map/keywords (3): | |
Maybe you are interested!
-
 Lesson plan for developing students' capacity in Technology subject for grades 11-20
Lesson plan for developing students' capacity in Technology subject for grades 11-20 -
 Lesson plan for developing students' capacity in Biology 12-14
Lesson plan for developing students' capacity in Biology 12-14 -
 Knowledge. Through this lesson, students must consolidate the following knowledge:
Knowledge. Through this lesson, students must consolidate the following knowledge: -
 Assessing Students' Level of Lesson Preparation Before Coming to Class
Assessing Students' Level of Lesson Preparation Before Coming to Class -
 Objective Test Questions Used in Teaching the Lesson "Equation of a Straight Line"
Objective Test Questions Used in Teaching the Lesson "Equation of a Straight Line"
- Contents of columns K, W of KWL table:
K (Known/Achieved
after online TH)
W (Wanted) further discussion) | L (What is achieved after the lesson) study) and proof | |
What did you do well and what did not? How can you improve your bad work? ................................................................ ................................................................ .......................... Satisfaction level:
| ||
Implementing organization:
Online activities on Teams:
Teachers deliver e-lessons on Teams classroom groups with requirements (such as content items).
Students perform homework tasks, discuss questions with teachers and classmates to edit/add in their homework notebooks; agree and take photos of their homework notebooks, and submit them to teachers in the corresponding homework section on Teams.
Teachers provide feedback on the content of the TH notebook and remind students. Organize online meetings with students to continue answering questions and guiding students to make adjustments (if necessary).
Students/pairs of students self-assess for the first time according to the teacher's request.
Activity 3: Practice and apply (1 period in class + online at home)
Objectives: (4), (5), (7), (8). Students accurately and systematically acquire knowledge and cooperate to practice and apply learned knowledge.
Content: Students work in groups to conduct experiments, solve chemistry exercises, and participate in learning games.
Students solve practical exercises (individually or in pairs) at home and submit them via MS Teams.
Products: Experimental results reports and solutions to exercises; game participation results; answers to practical exercises.
Implementing organization:
Live class activities:
Activity 3.1: Conduct experiments and solve chemistry exercises (33 minutes)
Teachers organize cooperative teaching by stations: divide the class into 8 groups (2 clusters, each cluster has 4 groups), disseminate tasks in the learning sheets at the stations.
STATION 1 WORKSHEET
Identify organic substances X, Y, Z, T and write the chemical reactions of the following transformation diagrams, clearly stating the reaction conditions (if any):
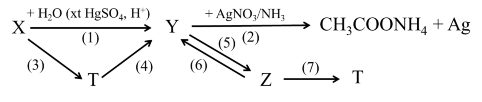
STATION 2 WORKSHEET
Write the structural formula and name the aldehydes with molecular formulas C 4 H 8 O and C 5 H 10 O, respectively.
STATION 3 WORKSHEET
Perform the following experiment : Take about 1 ml of 1% AgNO 3 solution into a clean test tube , slowly add 5% NH 3 solution drop by drop while shaking well until the precipitate is completely dissolved, then stop. Add 1 ml of ethanal to the test tube (drop around the wall), then gently heat the test tube over an alcohol lamp flame or soak it in a cup of hot water (60 - 70 0 C) for a few minutes.
State the phenomenon and write the chemical equation of the reaction that occurs.
STATION 4 WORKSHEET
1. Let 1.97 grams of formalin solution react with AgNO 3 solution in NH 3 (enough) to obtain 10.8 grams of Ag precipitate. The % concentration of formalin aldehyde in formalin is
A. 25.5%. B. 40.5%. C. 38.1%. D. 36.7%.
2. Let 10.9 grams of a mixture of two consecutive aldehydes in the homologous series of saturated, monofunctional, open-chain aldehydes react with an excess of AgNO 3 solution in NH 3 to obtain 43.2 grams of silver precipitate. What is the molecular formula of the two aldehydes?
A. C 2 H 5 CHO, C 3 H 7 CHO. B. C 3 H 7 CHO, C 4 H 9 CHO.
C.CH 3 CHO, C 2 H 5 CHO. D. HCHO, CH 3 CHO. Students learn about the tasks at the stations, ask questions (if any).
The teacher answers students' questions, organizes students to choose a starting station, work in groups (5 minutes) and write down the results on A4 paper . When the time is up, move to a new station. At the last station, the group of students writes down the results on A0 paper to prepare the report.
Any group that completes the main station's mission before the allotted time may move on to the additional station with the following mission:
SUPPLEMENTARY STATION WORKSHEET
By chemical methods, distinguish the following chemicals: formalin, ethyl alcohol, phenol solution. Write the chemical equations of the reactions that occur.
The teacher asks representatives of student groups to present the results at the stations. Other groups comment and ask questions to the presenting group.
Teachers review and summarize lesson knowledge.
Activity 3.2. Participate in the Chemistry Puzzle game (12 minutes)
The teacher forms 8 teams, introduces the game, and disseminates the requirements and gameplay to the students.
How to play: Each team is given a set of triangular puzzle pieces (cut apart) within a certain period of time (about 5-7 minutes). The teams must start by putting together the puzzle piece with the word "ANDEHIT" so that the pieces put together must have 2 opposite sides representing 2 pieces of information that are related to each other (or 1 question and 1 answer). The team that puts together correctly and fastest will win. When the time is up, the organization will cross-mark the groups according to the answers given by the teacher, count the number of pairs of correctly put together edges (each correct pair gets 1 point) and determine the winning team according to the number of points earned by each team.
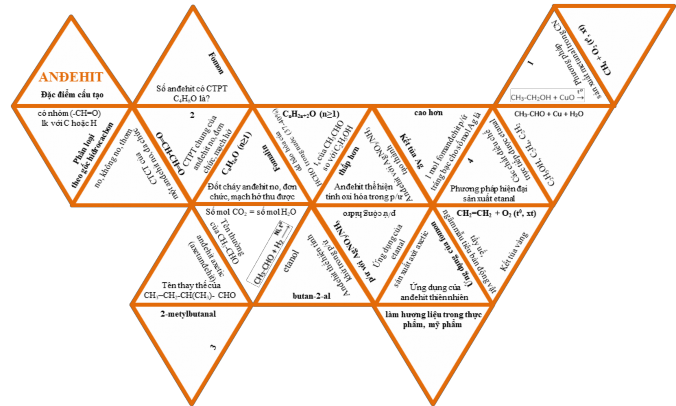
Students listen and discuss questions about the rules of the game.
The teacher answers questions and begins the game. The teacher uses commands to start the game, monitors and supervises the team's playing activities.
Students participate in the game and report the results.
The teacher gives the answers, corrects the matching results of the teams, deepens the key knowledge that needs to be remembered, then evaluates the results and awards (if any), comments on the spirit and attitude of the students. After that, the teacher disseminates the learning tasks of the next lesson (if necessary).
Online activities on Teams:
Teachers assign students to do at least 1 of the following 2 exercises (not required for all students). Requires creative presentation and clarification of: (1) the problem to be solved; (2) relevant known content; (3) solution steps and solutions.
Students/pairs of students solve practical exercises and send results to the teacher via the corresponding exercise on Teams.
Teacher comments, evaluates, summarizes and announces answers.
Exercise 1: Formalin is an aqueous solution of formaldehyde. A saturated solution of formaldehyde in water with a concentration of 37-40% is called formalin.
a. Why are human or animal corpses used as specimens for research often soaked in formalin?
of consumers?
b. Recently, many noodle and pho production and trading establishments have been discovered by authorities to be using formaldehyde in the production process. Please tell us what is the purpose of formaldehyde used in noodle and pho production? How does the abuse of formaldehyde in noodle, pho and food production affect health?
(Internet source) |
Instruct
a. Because formaldehyde (in formalin) denatures protein, turning protein into an elastic, non-rotting substance.
Formaldehyde is also toxic to bacteria (antiseptic) and thus kills bacteria that cause decomposition and putrefaction of human and animal corpses used as specimens.
b. Formaldehyde inhibits the activity, destroys bacteria, fungi and some microorganisms that spoil rice noodles, and denatures proteins (combining with proteins to form stable compounds that do not rot), thereby extending the shelf life, making rice noodles white, shiny, chewy and difficult to sour. This is different from rice noodles, which are usually opaque white or ivory white in color from rice flour, chewy and quickly sour.
Abusing formaldehyde in preserving vermicelli, pho and food not only reduces nutritional value but can also be harmful to health. Regular use of foods containing large amounts of formaldehyde (in formaldehyde) exceeding the permitted level for a long time can be dangerous. Acute reactions cause symptoms: severe abdominal pain, vomiting, vomiting blood, hematuria, kidney pain, cell necrosis, etc. When formaldehyde enters the digestive tract, it slows down and disrupts digestion; causes false fullness; gastritis, colitis, and intestinal inflammation; slows down the body's metabolism and nutritional metabolism; formaldehyde is converted into formic acid, which increases blood acidity, reduces white blood cells, and damages endocrine organs (stomach, liver, pancreas, kidneys). High levels of formaldehyde can cause death. In addition, commercial formaldehyde products can also contain many impurities, especially heavy metals, which can cause cancer in humans.
like why so?
Exercise 2:(Internet source) |
b. In some highland areas, people also hang meat (usually buffalo, beef, pork, etc.) on the kitchen ceiling to smoke, creating a specialty dish called "smoked meat" with a unique flavor. Please tell me why smoked meat can be preserved for a long time and has a unique flavor? Does frequent and frequent use of smoked meat products cause any harm to human health?
Instruct
a. Kitchen smoke contains a small amount of formaldehyde HCHO. HCHO has the ability to disinfect, kill microorganisms, molds, and prevent termites, so it makes baskets, trays, winnowing trays, etc. more durable.
b. Smoke contains many components including alcohols, phenols, aldehydes, carboxylic acids... (about 200 substances). These compounds (especially formaldehyde and acetic acid) are selectively absorbed and penetrate into the meat, strongly absorbed in meat with high humidity, inhibiting and destroying microorganisms that cause spoilage within a certain period of time, helping to preserve the meat longer. At the same time, kitchen smoke also dries and penetrates the natural compounds in the smoke into the meat, helping to create characteristic color and flavor.
Using smoked meat in moderation to diversify nutrition is less harmful because the product contains some harmful polycyclic aromatic hydrocarbons, phenols and aldehydes, but the amount remaining on the product is small and the body has biochemical or chemical reactions to reduce or eliminate their toxicity. However, if used regularly in large quantities, it will also directly affect the liver, kidneys, stomach, intestines, etc., increasing the risk of cancer. In addition, smoked meat also contains saturated fats that increase cholesterol levels, cause unhealthy weight gain, and increase the risk of cardiovascular disease and stroke.
Activity 4: Assess learning outcomes (at home)
Objective: Students can self-evaluate the results after the lesson and draw learning experiences for themselves.
Content: Students are required to self-evaluate what they have achieved after the lesson, draw lessons and complete the KWL table.
Product: Content of column L; good and bad things and how to improve.
Implementing organization:
Teachers ask students to do practice exercises, self-assess and learn from experience.
Students complete, take a photo of the KWL board and submit it to the teacher via the assignment on Teams. The teacher summarizes and announces the results of the student's learning assessment.
Teachers ask students to build a learning portfolio.
Students build a profile, save evidence and submit it to the teacher (if necessary).
Appendix 5.4. Lesson plan for lesson 45: Carboxylic acids (lesson 2)
LESSON 45: CARBOXYLIC ACIDS (lesson 2)
A. Objective
1. Chemical capacity
(1) Describe the chemical properties of carboxylic acids: Shows weak acidity (reacts with indicators, bases, basic oxides, salts of weaker acids, highly active metals), reacts with alcohol to form esters.
(2) Perform experiments on the acidity of acetic acid (or citric acid); reaction of acetic acid with ethyl alcohol (preparation of ethyl acetate); describe phenomena and explain.
(3) Describe the method of preparing carboxylic acid (preparing acetic acid by vinegar fermentation, oxidation of acetic aldehyde/alkane, starting from methanol).
(4) Describe the applications of some common carboxylic acids.
(5) Distinguish specific acids from alcohols and phenols by chemical methods.
(6) Calculate the mass or concentration of the acid solution in the reaction and the yield of the reaction to form the ester.
2. General capacity
Developing general and special competencies of students through organizing learning activities according to the BL model with the following manifestations:
- Students identify the objectives and content of the lesson (session 2).
- Plan a lesson in collaboration with another student.
- Access the internet, watch electronic lectures, and actively discuss to complete the requirements corresponding to the lecture.
- Apply knowledge to solve learning and practical problems assigned directly by teachers in class or online at home.
- Cooperate and support other students in performing tasks directly in class and online at home.
- Present and defend your own and your group's learning results during discussion.
- Evaluate results after online testing and after the entire lesson learning process.
- Identify good and bad work and suggest ways to improve in the next learning phase.
3. Quality
- Responsibility: Have a cooperative, sharing and responsible attitude in assigned tasks.
- Honesty: Be consistent between the content of the report and the results of the experiments, objectively evaluate the results of your own experiments and those of your classmates.
B. Teaching aids and learning materials
- Classes on Teams, e-lectures, worksheets, chemistry Bingo games!.
- Computer, projector, A0 paper , markers, magnets.
C. Learning activities
Activity 1: Identify learning tasks (5 minutes in class)
Objective: Students identify the learning tasks, recognize the lesson objectives and make a learning plan.
Content: Students are required to choose a "learning partner", study the lesson objectives and make a lesson plan.
Products: Lesson objectives (section A.1), student's lesson plan.
Implementing organization:
Direct activities in class: Teachers introduce learning tasks, requirements and assessment criteria for tasks to students.










