1.2.4.6 Hydrolysis method using dilute caustic solution
Using a dilute caustic solution combined with high temperature to hydrolyze raw materials, thereby breaking down the cell and tissue structure, breaking the bond between lipids and proteins, obtaining both oil and vitamins in a free state.
1.3 Quality indicators of fish oil (according to TCVN)
- The quality index of fish oil is evaluated based on Vietnamese standards for animal and vegetable fats and oils.
- Acid index according to TCVN 6127: 2010
Table 1.6 Acid index of some edible oils [7]
Sample
Refined canola oil | Fat | Sunflower seed oil rough | Extra virgin olive oil | Cold pressed wheat germ oil | Technical fatty acids | |
Acid index (mgKOH/g) | 0.080 | 0.381 | 1.39 | 5.48 | 7.48 | 128.1 |
Maybe you are interested!
- Soap index TCVN 6126:2007
Table 1.7 Saponification index of some edible oils [7]
Sample | ||||
Rape oil | Palm oil | Coconut oil | MCT Oil | |
Bar index room (mgKOH/g) | 190.2 | 199.5 | 256.8 | 334.1 |
- Iodine index according to TCVN 6122:2010
- Peroxide index according to TCVN 6121:2010
Peroxide value of some edible oils
A: Refined rapeseed/sunflower oil (1 : 1) G: Animal fat B: Olive oil (mixture of virgin and refined olive oil) H: Lard
C: Extra virgin olive oil I: Palm oil
D: Extra virgin olive oil J: Palm stearin
E: Canola oil K: Coconut oil
F: Lampante olive oil
This test was conducted by the German Institute for Standardization (DIN) in 2004-2005. The results were statistically analyzed according to TCVN 6910-1 (ISO 5725-1) and TCVN 6910-2 (ISO 5725-2) to give the data as shown in Table 1.8.
Table 1.8 Peroxide index of some edible oils [7]
Sample | |||||||||||
A | B | C | D | E | F | G | H | I | J | K | |
Peroxide index (meq/kg) | 1.63 | 3.21 | 8.34 | 12.04 | 19.02 | 26.92 | 1.6 | 3.67 | 2.99 | 4.77 | 0.55 |
Table 1.9 Quality indicators of crude tuna oil [12]
Crude Yellowfin Tuna oil specification
Product | Tuna oil |
Free Fatty Acid Index | Max 0.02% |
Acid Index | Max 4% |
Peroxide Index | Max 5 meq 0 2 /kg |
P-Anisidine Index | Max 40 |
Iodine Index | Min 175 g2/100g |
Unsaponifiable matter content | Max 2.5% |
DHA/EPA | Min 30% |
Impurities | 0.02% |
Humidity | Max 0.35% |
1.4 Changes in fish oil during storage
1.4.1 Hydrolysis of fish oil
The chemical properties of fats and oils are mainly due to the reaction of triglycerides, which has a significant impact on the change in product quality.
Under suitable conditions, grease is easily hydrolyzed according to the reaction C 3 H 5 (COOR) 3 + 3H 2 O → 3RCOOH + C 3 H 5 (OH) 3
If there is a certain amount of alkali (KOH, NaOH) present, after the hydrolysis reaction, the fatty acid will react
with alkalis to form alkali salts (soaps). RCOOH + NaOH → RCOONa + H 2 O
General equation:
C 3 H 5 (COOR) 3 +3NaOH → 3RCOONa + C 3 H 5 (OH) 3
1.4.2 Oxidation of fish oil
Fats contain many unsaturated fatty acids that are easily oxidized by atmospheric oxygen. The reaction occurs on the double bonds of carbon.
Oxidized lipids produce hydroperoxide compounds. These hydroperoxides continue to decompose to produce final products such as aldehyde compounds, acetone, alcohol, acid, etc., which make the oil have an unpleasant rancid smell. Rancidity is due to the following reactions.
Burning due to aldehyde production
- The oxidation phenomenon of aldehyde formation can occur in the presence or absence of enzymes, due to the reduction of fatty acids.
- When fatty acids turn into aldehydes, they have a very unpleasant rancid smell.
Burning due to ketone production
- This is the case of rancidity of fats with saturated fatty acids and hydrogen participating in the reaction.
The reaction is due to the oxidation of glycerin.
- The phenomenon of oxidation to form ketones is stimulated by some metals (such as lead, iron, manganese, copper)
- Glycerin is oxidized and gradually released into free form and then becomes epialdehyde (epihydrin aldehyde), making the fat smell rancid.
Rancidity due to the production of oxygen acid
- This is the case of oxidation of unsaturated fatty acids.
- “Active oxygen” attaches to the double bond of unsaturated fatty acids, forming peroxide, then oxygen acid, and finally decomposes into aldehyde.
- The reaction is not simultaneous, so the product contains acid, peroxide & aldehyde.
-Lead, cobalt, iron and especially copper act as catalysts for very fast oxidation reactions.
- In reality, rancid grease is difficult to distinguish in which form because acids, aldehydes, ketones, peroxides, etc. are all mixed in the product.
- Oxidized grease spreads from molecule to molecule and cannot be prevented.
- Oil and grease heated at high temperatures form acrolein C 3 H 4 O (Acrylaldehyde), an aldehyde.
toxic.
Chapter 2
RESEARCH OBJECTS AND METHODS
2.1 Research subjects
2.1.1 Tuna head oil
Fish oil is extracted from big-eye tuna heads collected at Thinh Hung - Suoi Dau Company Limited with each head weighing about 4 - 6.5 kg. The fish heads were frozen and then transported to the processing technology laboratory, the school's processing department. Here, the tuna heads were thawed, chopped and ground with a blender, then packaged.
into small packets of 200g. The raw material bags are stored in a freezer in the food technology laboratory at -18 o C ± 2 until use.
Tuna oil is extracted by hydrolysis with Flavourzyme enzyme under the following conditions: temperature 50 o C, natural pH of raw material, hydrolysis time: 2 hours, water/raw material ratio: 1:2. After the extraction process, fish oil is stored frozen at -18 o C ± 2.
2.1.2 Enzyme Flavourzyme
Flavourzyme is a protease derived from the Aspergillus oryzae fungus strain of Novozyme (Denmark) approved for use by FAO/WHO.

Flavourzyme enzyme works best at PH = 5÷7; the optimal temperature is about 50 o C.
2.1.3 Vitamin E (tocopherol)
Among the forms of vitamin E, α -tocopherol has the strongest biological activity and it is used as an antioxidant for lipids.
Molecular formula of -tocopherol R 1 =R 2 =R 3 =CH 3 The structural formula is shown in Figure 2.2
Figure 2.1 Vitamin E capsules
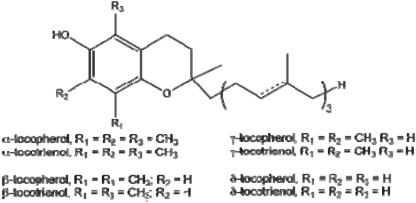
Figure 2.2 α-tocopherol
Vitamin E acts as an antioxidant and thus protects easily oxidized substances such as carotene, vitamin A, and unsaturated fatty acids. Therefore, vitamin E protects biological membranes containing unsaturated lipids.
Vitamin E is a colorless liquid, soluble in vegetable oils and organic solvents. Vitamin E is heat-resistant, can withstand 170 degrees Celsius, but is quickly destroyed by ultraviolet rays.
Vitamin E is obtained from SPM Joint Stock Company Lot 51, Road No. 2, Tan Tao Industrial Park, Binh Tan District, Ho Chi Minh City, Vietnam.
2.2 Research location
- Determination of chemical composition of tuna head and fish oil at the Biochemistry laboratory, processing department, Institute of Biotechnology and Environment, Nha Trang University.
- Hydrolysis of tuna head to remove oil at the food technology laboratory, processing faculty, Nha Trang University.
2.3 Research methods
2.3.1 Determination of chemical composition of tuna head
The process of determining the chemical composition of bigeye tuna head raw material is carried out according to the following diagram:
Bigeye tuna head
Grind
Determination of water, protein, lipid and ash content
Result



Figure 2.3 Diagram of determining the chemical composition of bigeye tuna
2.3.2 Process of separating tuna head oil by hydrolysis method





a. Process diagram:
Ingredient
Defrost
Hydrolysis
Enzyme inactivation
Dissociation
Bone
Centrifuge 1st time
Separate the parts
Fish oil
Hydrolysate
Centrifuge residue
Second centrifuge
Fish oil
Hydrolysate

Figure 2.4 Flowchart of fish oil separation process by hydrolysis method
b. Process description
Ingredients:
The raw material here is minced bigeye tuna head, and frozen at -18 o C ± 2
Defrosting:
The 200g bags of ingredients are removed from the freezer and thawed at 0-4 o C overnight in the refrigerator.
Hydrolysis:
After the ingredients have been thawed, we proceed with the hydrolysis process. Put all the fish heads that have been packed in bags into a glass cup, add water to the cup in a ratio of water: ingredients of 1:2. Use a glass rod to stir the mixture well. Raise the temperature of the mixture to 50 o C and add the enzyme. The hydrolysis process is carried out at 50 o C, the natural pH of the ingredients, the amount of enzyme added is 0.3% compared to the ingredients. Hydrolysis temperature
The sample is kept stable by a thermostatic bath. During the hydrolysis process, there is periodic stirring and the sample temperature must be constantly monitored, allowing only a temperature fluctuation of 0.5 o C.
Enzyme inactivation:
After the hydrolysis process is complete, remove the glass cup from the thermostatic bath and immediately place it in a pot of 95 o C water to inactivate the enzyme, the inactivation time is 15 minutes.
Separation:
Use a sieve to filter the mixture after hydrolysis, collect the liquid and remove the solid part (mainly bones).
First centrifuge:
Centrifuge the liquid part with a centrifuge at 5000 rpm for 30 minutes. After centrifugation, there is separation of 3 parts: lipid, hydrolysate, and centrifuged residue.
Hydrolyzed fish oil separation:
After centrifugation, we obtain 3 parts: fish oil in the top layer, hydrolyzed liquid in the middle and hydrolyzed residue in the bottom layer.

