with CD34, CD26, CD31, CD45 and HLA-DR markers. Typical cell surface markers for mesenchymal cells: SH2, SH3 and SH4, adhesion molecules of CD29, CD44 and HLA-A, B, C. MSCs have a spindle and flat filamentous shape. The only difference between these two types of MSCs is the level of CD90 KN expression. Spindle-shaped MSCs express a lot of CD90 KN, while flat filamentous MSCs show no expression of this KN [22].
Cord blood stem cells have a different advantage than stem cells created from bone marrow: MDR stem cells are not damaged by disease and mutation. Therefore, this is a stem cell source that is being increasingly used in the world. In addition, compared to other stem cell sources, this is a readily available stem cell source and has HLA test results available in the community MDR bank, so the waiting time for transplantation will be significantly shortened, making it possible to take advantage of the golden time in treating patients.
The main disadvantage of this source of stem cells is that the number of stem cells is quite low, leading to slow graft growth and a high risk of infection. Currently, there are many research trials to overcome this problem.
1.2. Creating stem cells from umbilical cord blood
Maybe you are interested!
-
 Research on the application of the process of collecting, processing and preserving community umbilical cord blood stem cells - 19
Research on the application of the process of collecting, processing and preserving community umbilical cord blood stem cells - 19 -
 Factors Affecting the Results of Cord Blood Stem Cell Transplantation for the Treatment of Hematological Diseases
Factors Affecting the Results of Cord Blood Stem Cell Transplantation for the Treatment of Hematological Diseases -
 Sample Size Distribution for Information Collection and Assessment of the Level of Linkage of Seafood Processing Facilities
Sample Size Distribution for Information Collection and Assessment of the Level of Linkage of Seafood Processing Facilities -
 Poetry Collection "Going to Battle" and "Blood and Flowers"
Poetry Collection "Going to Battle" and "Blood and Flowers" -
 HIV Diagnostic Test Sample Collection Using Dried Blood Drop Kit
HIV Diagnostic Test Sample Collection Using Dried Blood Drop Kit
1.2.1. Procedure for collecting, processing and preserving umbilical cord blood
1.2.1.1. Cord blood collection
Umbilical cord blood is a special source of stem cells, utilizing a waste product from the reproductive process to transform into a valuable source of medicine for treatment [23]. Therefore, the collection, processing and preservation of this stem cell source also requires different processes compared to other stem cell sources. Worldwide, there are many studies to improve processes to increase the quantity and quality of MDR stem cells for clinical use. The first step of this process is the collection of blood from the umbilical cord and placenta of mothers and newborns.
Collection can be done at two times: before and after the placenta is born. Research results have shown that collection before the placenta is born, which is the time right after the umbilical cord is clamped and cut while the placenta is still in the uterus, will help to collect the most optimal amount of TBG [24]. The volume of MDR collected at facilities is usually a minimum standard of 50 ml.
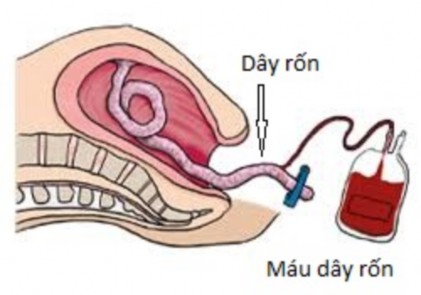
Figure 1.5. Cord blood collection before delivery
1.2.1.2. Umbilical cord blood processing
After collection, MDR samples will be transferred to MDR banks for processing. This is a very important step to (1) reduce the volume, (2) remove red blood cells, (3) purify TBG. The completed MDR has an average volume of about 25 ml.
The first MDR blood banks in the world such as the New York Blood Center applied manual volume reduction processes, using preparations that increase red blood cell sedimentation such as HES (hydroxylethyl starch) or Hetastarch (ethoxylated amylopectin) to remove plasma and red blood cells, reducing the hematocrit of the product [25]. However, manual processing often takes place in many steps,
open process and low stability due to dependence on technician skills. In 2001, Biosafe introduced the first generation of machines, Sepax, with fully automatic processing capabilities, specifically designed for cord blood processing and gradually becoming an international standard, used in most cord blood banks in the world [26]. Processing on an automatic system has a very important advantage of stability, a closed process, ensuring safety and limiting infection. However, compared to manual processing, automatic processing also has certain disadvantages such as residual red blood cells, especially high costs. Therefore, community cord blood banks must choose a processing method that is appropriate to their existing economic resources. In Japan, manual processing has been optimized to overcome these disadvantages and has become the method of choice for large-scale application at a more reasonable price than automatic technology. The National Institute of Hematology and Blood Transfusion sent staff to Japan to study the process and optimized it to suit Vietnamese conditions.
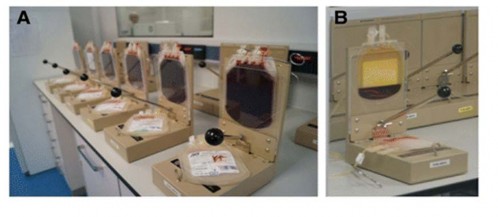
Figure 1.6. Manual processing of umbilical cord blood
1.2.1.3. Preservation of umbilical cord blood stem cells
After being processed to reduce the volume and remove excess components, the cord blood will be mixed with a preservation solution to protect the cells in the frozen environment. The success of stem cell banking depends on the preservation of stem cells.
The biggest risk to cells during freezing and thawing is the change in liquid-solid state, creating crystals during the freezing process that damage cells. When the cooling speed is fast, ice crystals are created inside the cells, inside the organelles, tearing the membrane and cell structure, causing cells to die quickly. When the cooling speed is slow, ice crystals will be created outside the cells, increasing osmotic pressure due to water being drawn into the ice crystals. Currently, people have used cell protection agents at freezing temperatures. There are 2 types of protection agents: cell-penetrating agents (DMSO, glycerol) and non-penetrating agents (HES).
After collection (and processing if necessary), the TBG mass will be stored in an environment with a temperature of 2-6ºC before being used fresh or frozen. The storage time will not exceed 72 hours.
After processing, the hematopoietic stem cell mass will be mixed with a preservative before being cooled and stored frozen at -196ºC. 10% DMSO solution is the most suitable preservative for hematopoietic stem cell mass. Before freezing the hematopoietic stem cell mass, it is necessary to lower the temperature according to a predetermined program to bring the stem cell mass down to -60ºC, then the stem cell mass is transferred to a nitrogen tank in the vapor or liquid phase to maintain the storage temperature at -150ºC to -196ºC. Under these conditions, the hematopoietic stem cell mass can be stored for a very long time, possibly decades, while still ensuring the quantity and quality of the stem cell mass.
1.2.2. Types of cord blood banks
Knudtzon's studies from 1974 showed that MDR contained hematopoietic stem cells and could be stored for a long time, paving the way for their application in treatment [27]. In 1988, in Paris, the first successful MDR stem cell transplant for a Fanconi patient. Results of MDR stem cell transplant in the following years
is the driving force for a series of TBG MDR banks in France, the US, Germany, Japan... to be born and spread throughout the world with different types.
MDR stem cell banks include: (1) community MDR banks, which store MDR stem cell units for use in an unrelated recipient; (2) private MDR banks, which store MDR for future use by the donor or a relative; (3) hybrid banks, which provide both types of services [28].
1.2.2.1. Community cord blood stem cell bank
As a bank used by anyone in the community, MDR stem cells are owned by the bank, and the source of stem cells is donated by volunteers to the bank. The first community MDR stem cell bank was established in 1991 in New York, USA. After that, this banking model was replicated worldwide because of its outstanding benefits. Unlike personal MDR storage banks, these banks often take MDR from volunteer mothers to donate for use in cases of patients in need of transplantation, so the efficiency of use is higher. MDR samples are collected according to predetermined standards for processing to create quality stem cell units. Thanks to the establishment of predetermined standards, the quality of stem cell units (shown in the collected volume, average number of stem cells, average number of CD34 cells) is higher and more stable than MDR from personal storage banks. This is the most suitable and effective form of MDR storage for cases where patients need stem cell transplantation but do not have a suitable donor in their family. In Europe, this model is relatively popular, accounting for a much larger proportion than personal MDR storage banks. Some countries such as Germany and Italy only allow the establishment of this bank, completely discouraging or even prohibiting the establishment of personal MDR storage banks. Because stem cell units in the community MDR bank can be selected for transplantation.
Anyone should require that in addition to the general safety of the samples, the number of TBGs must be large enough to be used for the subjects. In addition, HLA component testing is a mandatory requirement as well as blood grouping in blood transfusion to choose the most suitable unit for the patient [29].
In the world, Japan is one of the pioneering countries in MDR stem cell transplantation with the first transplant performed in 1995. Japan has 11 community MDR stem cell banks. Currently, they have been centralized and merged into 8 MDR banks for community storage with a total regular storage of about
32,000 units [30].
1.2.2.2. Personal TBG MDR storage bank
This is a bank where the donor or biological owner of the stem cell sample needs to deposit, it is not owned by the bank. At the request of the payer, it will collect, process, preserve and use. The personal MDR stem cell bank stores MDR stem cell units for preventive purposes, meaning that in the future if the donor or a relative develops a disease that can be treated with autologous stem cell transplantation, the MDR stem cell units can be used with the donor's permission and under the guidance of a physician. The units in the personal MDR stem cell bank remain the property of the child, under the guardianship of the parents and are not available to the public. However, they can be made available to other family members, so most personal MDR stem cell banks are banks for personal or family use [31].
This TBG bank is also growing strongly, currently the Cord Blood Registry in the US is the largest bank in the world with more than 500,000 samples. The Via Cord Bank in the US in 2013 had 300,000 samples stored. However, this type has some major disadvantages such as: the number of TBG is not high and unstable due to mandatory collection, only for personal use.
The use rate is usually quite low for the depositor or his or her family members. The chance of using MDR for the treatment of hematopoietic disorders before the age of 20 is low and is estimated to vary from 1/2,700 to 1/20,000 [ 32 ]. If the depositor does not have a need to use it, the collection, processing and storage becomes a huge waste of effort and cost. However, in some countries such as Italy this banking is prohibited.
1.2.2.3. Combined bank of community and individual storage.
This type is not common but has many different forms. Some banks divide MDR stem cells into two separate parts, one part is stored for use in autologous or allogeneic transplantation, the other part is used for community use. In others, units are initially stored for individuals and then transferred to community use [33]. Or parents may donate MDR to community banks but retain ownership for a specified period of time sufficient to meet the child's final needs [34].
1.2.3. Searching for cord blood stem cells for transplantation
The main purpose of establishing an MDR stem cell bank is to find a suitable MDR stem cell unit for patients in need of transplantation. The process of finding an MDR stem cell unit begins with the patient's HLA analysis. The patient is sent information including: diagnosis, HLA results, weight, blood type to the bank to evaluate compatibility. Therefore, issues of HLA, stem cell dose, blood type, gender and patient's medical conditions are particularly important issues affecting the success of an MDR stem cell transplant.
1.2.3.1. Select a suitable HLA (Human Leucocyte Antigen) sample
Human Leucocyte Antigen (HLA) or Major Histocompatibility Complex (MHC) plays a very important role in tissue and organ transplantation in general and stem cell transplantation in particular. HLA was first known because of its association with the phenomenon of
transplant rejection. This protein plays an important role in the body's immune organization as well as the "communication" mechanisms between cells. HLA is created by a group of genes that encode proteins that present antigens on the cell surface of most vertebrates.
The HLA system consists of a complex series of genes located on chromosome 6 and their molecular products are involved in immune regulation and cell differentiation. HLA can induce an immune responseby recognizing modified peptides that recognize foreign HLA molecules throughtheir polymorphisms. HLA incompatibility is associated with failure ofgraft, delayed immune reconstitution, graft-versus-host disease (GVHD), andmortality rate [ 35].
There are three classical loci in HLA class I: HLA-A, -B and -Cw and five loci in class II: HLA-DR, -DQ, -DP, -DM and -DO. The HLA system is highly polymorphic. The diversity of class I and II genes can be analyzed by serological methods or molecular methods at the DNA level by different methods such as specific primers (SSP) and specific probes (SSO). HLA class I and II matching is important in transplantation especially kidney and bone marrow transplantation. Allografts induce both humoral and cellular immune responses in the recipient which leads to graft rejection and graft versus host disease (GVHD).
The purpose of allogeneic hematopoietic stem cell transplantation for patients with hematological malignancies is to restore normal hematopoiesis and increase the graft-versus-tumor effect. The success of allogeneic hematopoietic stem cell transplantation in general and allogeneic MDR stem cell transplantation in particular depends on the degree of compatibility of HLA class I and class II alleles between the donor and recipient. There have been many studies on the necessary level of HLA compatibility between the donor and recipient, such as the Fred Hutchinson Cancer Research Center (FHCRC) and the National Marrow Donor Program (NMDP) have reported the importance of HLA matching.