c). Reaction due to α-carboxyl group
In addition to the common reactions to form esters, amides, salts... the -COOH group also has other typical reactions such as being reduced to amino alcohol compounds under the catalysis of NaBH4 .
R - NH 2 - CH – COOH R - NH 2 - CH – CH 2 OH
The -COOH group can form an aminoacyl-adenylate complex in the amino acid activation reaction to synthesize protein, or can eliminate CO2 ( found a lot in the amino acid degradation process ). Maybe you are interested! Division of common property between husband and wife according to the Law on Marriage and Family 2014 - 9
Division of common property between husband and wife according to the Law on Marriage and Family 2014 - 9 The Nature and Role of the Internal Control System
The Nature and Role of the Internal Control System Current Status of Cooperation Between School, Family, and Society in Educating Children Through Student Activities
Current Status of Cooperation Between School, Family, and Society in Educating Children Through Student Activities Student Survey Questionnaire
Student Survey Questionnaire The Nature of Commercial Banking Business
The Nature of Commercial Banking Business
d). Reaction due to α-amine group
Many reactions of the amino group are used to qualitatively and quantitatively identify amino acid properties such as:
To quantify the nitrogen of amino acids, people react with HNO 2 to release N 2 .
HOOC – CH – R
HOOC – CH – R
NH 2 + HNO 2
OH + N 2 + H 2 O
To quantify amino acids, people react with aldehydes to form Schiff bases.
To determine the N-terminal amino acid, people react with 2-4 dinitrofluorobenzene (Sanger reaction) or phenyliothiocyanate (Edman reaction).
3. PEPTID
3.1. Definition
Peptides are protein molecules consisting of two to dozens of amino acids, linked together by peptide bonds, with a molecular weight < 6000 daltons.
3.2. Structure
Peptides can be synthesized naturally or formed by protein degradation. In peptides, amino acids are linked together through peptide bonds (Figure 7.6).
In the peptide chain, at the end of the amino acid is the free α-amine group (α-NH 2 ) called the N-terminus and the end with the free α-carboxyl group (α-COOH) is called the C-terminus. The peptide bond forms the main skeleton, and the R group forms the side chain of the chain.
Thus, a peptide chain always has a free α-amino group and a free α-carboxyl group. With n amino acids, there are (n-1) peptide bonds formed and (n-1) water molecules eliminated.
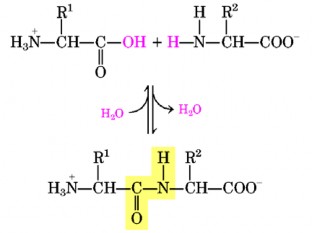
Figure 7.6. Peptide bond formation
3.3. How to name
Named according to the number of amino acids in the peptide chain: dipeptide (2 amino acids), tripeptide (3 amino acids), ...
Called by amino acid radical: starting from the N-terminal end of the first amino acid to the C-terminal end of the last amino acid.
Keep the name of the amino acid with the C-terminal end and replace the tails of other amino acids with the YL tail.
For example: Serine – Alanine – Methionine – Valine – Glycine Seryl – Alanyl – Methionyl – Valyl – Glycine
Call by name:
For example: Glutathione: tripeptide consisting of γ-glutamyl – cysteyl – glycine Glucagon: polypeptide consisting of 29 amino acids
Insulin: polypeptide of 51 amino acids
To indicate the composition and order of amino acids, use the letter symbol or a letter (if the peptide chain is long) of the amino acid written consecutively, from the N-terminal to the C-terminal, from left to right.
For example: Alanine – Valine – Leucine – Histidine – Proline
Ala – Val – Leu – His – Pro AVLHP
H 2 N – Ala – Val – Leu – His – Pro – COOH
3.4. Chemical properties
Peptide molecules have free -NH 2 and -COOH groups with chemical properties like amino acids, so electrophoresis, chromatography, etc. methods used to determine amino acids are also used to determine peptides.
In strong alkaline environment, peptide bonds react with
The R groups of peptides also give characteristic color reactions of the corresponding free amino acids. One of the most characteristic color reactions to determine peptide bonds is the Biuret color reaction . Biuret is the condensation product of 2 urea molecules with the release of 1 NH 3 molecule . This reaction does not occur with free amino acids and with dipeptides.
NH 2
NH 2
NH 2
O = C
O = C + O = C NH 3 + NH NH 2 NH 2 O = C
Urea
Urea
NH 2
Biuret
CuSO 4 forms a purple-red complex (Figure 3.11.), with maximum absorption at 540 nm. This reaction is widely used for protein quantification.
3.5. Common peptides in nature
In nature, there exist many types of peptides with important functions related to the body's vital activities: hormones, antibiotics, precursors of bacterial cells, etc. Besides, there are also peptides with unclear functions, and peptides that are unfinished hydrolysis products of proteins.
Some important peptides have many meanings for the life activities of living organisms.
object.
3.5.1. Glutathione and analogues
NH 2
CH 2 - SH
HOOC- CH - CH 2 - CH 2 -CO-NH- CH - CO-NH-CH 2 -COOH
Glutathione is a tripeptide γ-glutamyl-cysteyl-glycine with the structural formula:
In the structure of 76lutathione, the SH group of 76lutathione is the active group, so people often abbreviate 76lutathione as GSH. This is the reduced form of 76lutathione. In the active environment, 76lutathione can give up hydrogen (H) to form the oxidized form (GSSG) and vice versa, it can accept H to form the reduced form:
+ 2H
2 GSH GSSG
+ 2H
Glutathione is one of the most common intracellular peptides, acting as an oxidation-reduction system (hydrogen transport). It is widely distributed in tissues and organs such as: liver, kidney, spleen, heart, lungs, red blood cells, etc.
3.5.2. Growth hormones (HGH)
Human growth hormone (HGH) also known as STH (somatotropin hormone) is a polypeptide chain consisting of 191 amino acids with a molecular weight of 20,000. In the structure, there are two disulfide bridges formed between amino acids 53-165 and between amino acids 182-189. The biologically active chain consists of 134 amino acids.
HGH has an effect on general growth, stimulating cartilage formation rather than bone formation. It is also a metabolite, stimulating protein synthesis and is a hormone that causes hyperglycemia while stimulating lipid degradation to ensure energy needs in the body, causing an increase in free fatty acids in the plasma.
3.5.3. Insulin
Since 1953, Sanger (Nobel Prize 1958) researched, purified and completely determined the structure of the insulin molecule. Consisting of 51 amino acids, molecular weight 5,700, consisting of 2 polypeptide chains (chain A has 21 amino acids, chain B has 30 amino acids) connected by 2 disulfide bridges. In chain A, a disulfide bridge is also formed between the 6th amino acid and the 11th amino acid. The species-specific part only focuses on the 8th-9th-10th, 12th-14th amino acids of chain A and especially the 30th amino acid of chain B.
Insulin has the most profound effect among all pancreatic hormones, especially on the process of carbohydrate metabolism, has the effect of lowering blood sugar, stimulating the synthesis and inhibiting the process of glycogen degradation in muscle, liver and adipose tissue. In particular, insulin enhances the synthesis of fatty acids, proteins and stimulates glycolysis. The most important effect of insulin is to stimulate the penetration of glucose, some oses, amino acids in muscle and fat cells. Therefore, insulin reduces the amount of glucose in the blood. In addition, insulin also reduces gluconeogenesis by reducing the concentration of enzymes such as pyruvate carboxylase and fructose 1-6 diphosphatase.
3.6. Some other biologically active peptides
a) Oxytocin: is a peptide with 9 amino acids, acts on the smooth muscles of the uterus and mammary glands, causing contractions during childbirth and stimulating milk secretion during breastfeeding.
b) Vasopressin: is a 9-amino acid peptide, has antidiuretic effects, increases water reabsorption in the kidneys, and also causes vasoconstriction, thereby increasing blood pressure.
c) Glucagon: contains 29 amino acids, has the effect of increasing blood sugar.
d) Bradykinin: is a substance that causes smooth muscle hypotension, increases self-stimulation and self-defense reactions, low doses have analgesic effects.
e) Kallidine: agent that causes smooth muscle hypotension.
f) Antibiotic peptides: produced by bacteria or fungi: gramicillin, penicillin, tyrocidin, bacitracin.
4. PROTEIN
4.1. Definition
Proteins are macromolecules with a mass > 6000 daltons, consisting of hundreds or thousands of amino acids combined together to form a polypeptide chain.
4.2. Classification
Proteins can be classified in many ways: based on structure, solubility, shape, function, etc.
4.2.1. Classification by structure
- Pure protein (simple protein): is a protein that when hydrolyzed gives common amino acids, for example: albumin, globulin, protamine, histone, etc...
- Impurity protein (conjugated protein): includes pure protein and non-protein part (which is not protein in nature, can be glucid, lipid, nucleic acid, metal, etc...).
- Protein when partially hydrolyzed forms peptides, when completely hydrolyzed turns into amino acids.
4.2.2. Classification by shape
- Globular proteins: are proteins with length/width < 10 daltons; include enzyme proteins and serum proteins.
- Fibrous proteins: are proteins with length/width dimensions
> 10, including keratin of hair, nails and collagen of connective tissue.
4.3. Protein structure
- Primary structure: represents the arrangement of amino acids in a polypeptide chain, or multiple polypeptide chains, and the location of disulfide bonds.
- Secondary structure : represents the twisting of the polypeptide chain, typically the secondary structure of fibrous proteins, hydrogen bonds play an important role in secondary structure.
- Tertiary structure : represents the twisting and folding of the polypeptide chain, disulfide bonds play an important role in maintaining tertiary structure.
- Quaternary structure : represents the combination of many polypeptide chains with tertiary structure in a protein molecule. For example: hemoglobin has quaternary structure.
4.4. Properties of proteins
4.4.1. Physical and chemical properties
a) Solubility
Different types of proteins have the ability to dissolve easily in certain solvents, such as: albumin is easily soluble in water, globulin is easily soluble in dilute salt, glutelin is only soluble in dilute alkaline or acid solutions, etc.
b) Hydration
In an aqueous environment, proteins combine with water to swell into a colloidal form (hydration), water molecules adhere to hydrophilic groups in protein molecules such as
-NH 2 , -COOH... The water layer surrounding protein molecules is one of the factors that stabilize the structure, preventing protein molecules from sticking together to form precipitates.
c) Viscosity of protein solution
Different proteins have different viscosities when dissolved in solution. This property is used to determine the molecular weight of the protein (the higher the viscosity, the higher the molecular weight).
d) Dielectric constant of protein solution
When neutral organic solvents such as ethanol and acetone are added to aqueous protein solutions, the solubility of the protein decreases and the protein precipitates due to a decrease in the hydration level of the ionized groups of the protein, and the protein molecules combine to form a precipitate. Thus, the dielectric constant of the solvent prevents the electrostatic force between the charged groups of the protein and water.
e) Electrolytic properties of proteins
Like amino acids, proteins are amphoteric electrolytes because the molecule contains many strong polar groups (R groups) of amino acids.
The charge state of these groups depends on the pH of the environment. At a certain pH where the sum of the (+) and (-) charges of the protein molecule is zero, the protein molecule does not move in the electric field, this is called pH i (isoelectric point ) of the protein.
In an environment with pH = pH i of the protein, proteins easily clump together, so this property is used to determine the pH i of the protein as well as to precipitate the protein. On the other hand, due to the difference in pH i between proteins, the pH of the environment can be adjusted to separate the proteins from their mixture.
f) Salt precipitation of protein solution
Neutral salts have a subtle effect on the solubility of globular proteins, low concentrations increase the solubility. As the salt concentration increases, the solubility decreases and at very high salt concentrations, the protein can precipitate completely.
Different proteins precipitate at different concentrations of neutral salt. This property is used to extract and separate proteins from a mixture. This is called the salt precipitation method.
g) Denaturation
When the bonds (except peptide bonds) in the protein are broken, the protein structure changes, the protein is denatured. Denaturation reduces solubility and loses biological activity. When denatured, the protein structure is reversed, the hydrophobic groups turn outward, the hydrophilic groups turn inward, hydration is reduced, and proteins easily combine with each other. This property is applied in urine protein testing.
Denaturing agents:
Physics : temperature, ultraviolet rays, high pressure, ...
Chemistry : strong acids (a. trichloroacetic, a. nitric, ...), heavy metal salts (HgCl 2 ), strong alkalis, alcohols, ...
Distinguish between 2 types of denaturation:
Reversible : denatured protein can return to its original state (reversion). Trypsin solution is denatured when heated at pH = 3 and is converted after cooling for a certain period of time.
Irreversible : denatured protein cannot return to its original state, like boiling a chicken egg.
4.4.2. Chemical reactions of proteins
Proteins have chemical reactions similar to amino acids and peptides: reactions of -COOH, -NH 2 groups , R groups and color reactions characteristic of peptide bonds such as the biuret reaction.
In addition, there are a number of other characteristic color reactions that are important in detecting proteins and other amino acid residues in the polypeptide chain.
a) Reaction with Folin-Ciocateau reagent
Folin-Ciocateau reagent contains phosphomolybdic acid and phosphovolframic acid. These substances increase the sensitivity of the biuret reaction, and on the other hand react with Tyr and Trp radicals in protein molecules. These radicals participate in the formation of a blue complex.
b) Other characteristic color reactions
Used to detect amino acids and proteins in solution based on the presence of chemical functional groups in protein molecules.
Xanthproteic reaction: amino acid radicals Tyr, Trp, Phe in protein react with concentrated HNO 3 to form yellow color and after adding alkali, it turns orange.
Pauli reaction: Tyr and His radicals react with diazobenzosulfonic acid to form cherry red color.
Millon reaction: Tyr radical reacts with mercury nitrate in concentrated HNO 3 to form an earthy brown precipitate.
Saccaguichi reaction: Arg group reacts with alkaline solution of α-naphthol and hypobromite to give cherry red color.
Adamkievich reaction: Trp group reacts with glyoxylic acid and concentrated H 2 SO 4 to form a red-violet ring at the interface.
4.5. Biological functions of Protein
Protein participates in all life activities in living organisms. In addition to being the main component in the structure of cells and tissues, protein also has many rich functions.
Other factors determine the basic characteristics of life such as the transmission of genetic information, the metabolism of substances by 81hormones, antibodies against diseases, 81hormones that transmit signals in cells, etc.
4.5.1. Shaping function
In addition to proteins that have structural functions such as virus shells and cell membranes, there are also proteins that are often fibrous, such as: sclerotin found in the outer shell of insects; fibroin of silkworms and spiders; collagen and elastin of connective tissue and bone tissue. Collagen ensures the strength and flexibility of connective tissue.
4.5.2. Catalytic function
Most reactions in the body are catalyzed by specific proteins called enzymes. Currently, about 3,500 different enzymes are known, many of which have been purified, crystallized, and structurally studied.
4.5.3. Protection function
Proteins participate in the immune system, especially many types of proteins perform separate functions that create specific and non-specific immune effects. The most commonly mentioned immune proteins are antibodies, complements...
In addition, protein also participates in the blood clotting process to prevent blood loss for the body or some species can produce toxins that are protein in nature such as snake venom enzymes, lectins, etc. that have the ability to destroy enemies to protect the body.
4.5.4. Transport function
In the body, there are proteins that have the task of transporting such as: hemoglobin, myoglobin, hemocyanin transporting O 2 , CO 2 and H + throughout the tissues and organs in the body; lipoprotein transporting lipids, ceruloplasmin transporting Cu in the blood...
4.5.5. Motor function
Many proteins are responsible for muscle contraction (myosin, actin), and chromosome translocation during cell division.
4.5.6. Storage and nutritional functions
Reserve proteins are an important source of nutrition for tissue and embryo development. Reserve proteins such as casein of milk, ovalbumin of eggs, ferritin of spleen (iron storage), etc.
4.5.7. Nerve signal transmission function
Proteins involved in the transmission of nerve signals to specific stimuli such as the visual pigment rhodopsin in the retina.
4.5.8. Air conditioning function
Regulates metabolism through its effects on the genetic information system. Hormones and specific enzyme inhibitory proteins all have regulatory functions.
